Recherches actuelles
La recherche de Guido ORI se concentre principalement sur l’application de techniques de modélisation atomique avancées (MD/MC, FPMD (BOMD, CPMD), MLIP), visant à une véritable capacité prédictive quantitative des structures, des liaisons chimiques, ainsi que des propriétés dynamiques et électroniques pour une large gamme de matériaux (verres, matériaux poreux et hybrides), dans le but d’une compréhension fondamentale et d’applications technologiques. Il a approfondi son expertise sur les vitrocéramiques multifonctionnelles à l’Imperial College de Londres dans le cadre du réseau d’excellence financé par l’UE sur les matériaux multifonctionnels basés sur la connaissance. Il a également travaillé sur les matériaux hybrides au laboratoire MACS de l’Université de Montpellier et au laboratoire international conjoint UMI CNRS-MIT « MultiScale Materials Science for Energy and Environment » au Massachusetts Institute of Technology.
Parcours universitaire
- 2024.06.19 Habilitation à diriger des Recherches (HDR) entitled: “Quantitative predictive modeling of complex disordered and hybrid materials for memory and energy applications“, Strasbourg
- 2014.12-present CNRS Researcher (CNCR), IPCMS UMR7504, Strasbourg
- 2012.09-2014.11 Visiting Postdoctoral Researcher at MSE2 UMI CNRS-MIT, Cambridge (USA)
- 2012.04-2013.08 Postdoctoral Researcher, MACS-ICG Laboratory, Montpellier (FRA)
- 2011.05-2011.12 Postdoctoral Researcher (CDD), IPCMS, Strasbourg (FRA)
- 2010.01-2011.04 Postdoctoral Researcher, University of Modena and Reggio Emilia (ITA)
- 2008.04-2008.07 Ph.D. research collaboration, Imperial College of London EU-NoE-KMM- (UK)
- 2007.01-2010.03 Ph.D. in Multiscale Modelling, Depart. of Materials and Envir. Eng., Univ. of Modena and RE (ITA)
Publications
1839302
NZSFH59F
2025
1
surface-science-reports
50
creator
asc
year
7054
https://www.ipcms.unistra.fr/plugins/zotpress/
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3Afalse%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%22GYNJLGE3%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Shuaib%20et%20al.%22%2C%22parsedDate%22%3A%222025%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EF.%20Shuaib%2C%20G.%20Ori%2C%20P.%20Thomas%2C%20O.%20Masson%2C%20A.%20Bouzid%2C%20Multikernel%20similarity-based%20clustering%20of%20amorphous%20systems%20and%20machine-learned%20interatomic%20potentials%20by%20active%20learning%2C%20Journal%20of%20the%20American%20Ceramic%20Society%20108%20%282025%29%20e20128.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1111%5C%2Fjace.20128%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1111%5C%2Fjace.20128%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Multikernel%20similarity-based%20clustering%20of%20amorphous%20systems%20and%20machine-learned%20interatomic%20potentials%20by%20active%20learning%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Firas%22%2C%22lastName%22%3A%22Shuaib%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Philippe%22%2C%22lastName%22%3A%22Thomas%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Olivier%22%2C%22lastName%22%3A%22Masson%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%5D%2C%22abstractNote%22%3A%22We%20present%20a%20hybrid%20similarity%20kernel%20that%20exemplifies%20the%20integration%20of%20short-%20and%20long-range%20descriptors%20via%20the%20use%20of%20an%20average%20kernel%20approach.%20This%20technique%20allows%20for%20a%20direct%20measure%20of%20the%20similarity%20between%20amorphous%20configurations%2C%20and%20when%20combined%20with%20an%20active%20learning%20%28AL%29%20spectral%20clustering%20approach%2C%20it%20leads%20to%20a%20classification%20of%20the%20amorphous%20configurations%20into%20uncorrelated%20clusters.%20Subsequently%2C%20a%20minimum%20size%20database%20is%20built%20by%20considering%20a%20small%20fraction%20of%20configurations%20belonging%20to%20each%20cluster%20and%20a%20machine%20learning%20interatomic%20potential%20%28MLIP%29%2C%20within%20the%20Gaussian%20approximation%20scheme%2C%20is%20fitted%20by%20relying%20on%20a%20Bayesian%20optimization%20of%20the%20potential%20hyperparameters.%20This%20step%20is%20embedded%20within%20an%20AL%20loop%20that%20allows%20to%20sequentially%20increase%20the%20size%20of%20the%20learning%20database%20whenever%20the%20MLIP%20fails%20to%20meet%20a%20predefined%20energy%20convergence%20threshold.%20As%20such%2C%20MLIP%20are%20fitted%20in%20an%20almost%20fully%20automatized%20fashion.%20This%20approach%20is%20tested%20on%20two%20diverse%20amorphous%20systems%20that%20were%20previously%20generated%20using%20first-principles%20molecular%20dynamics.%20Accurate%20potentials%20with%20less%20than%202%20meV%5C%2Fatom%20root%20mean%20square%20energy%20error%20compared%20to%20the%20reference%20data%20are%20obtained.%20This%20accuracy%20is%20achieved%20with%20only%20175%20configurations%20sampling%20the%20studied%20systems%20at%20various%20temperatures.%20The%20robustness%20of%20these%20potentials%20is%20then%20confirmed%20by%20producing%20models%20with%20several%20thousands%20of%20atoms%20featuring%20a%20good%20agreement%20with%20reference%20ab%20initio%20and%20experimental%20data.%22%2C%22date%22%3A%222025%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1111%5C%2Fjace.20128%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1111%5C%2Fjace.20128%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220002-7820%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T14%3A24%3A12Z%22%7D%7D%2C%7B%22key%22%3A%224B3B2525%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Shuaib%20et%20al.%22%2C%22parsedDate%22%3A%222025%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EF.%20Shuaib%2C%20A.%20Bouzid%2C%20R.%20Piotrowski%2C%20G.%20Delaizir%2C%20P.-M.%20Geffroy%2C%20D.%20Hamani%2C%20R.%20Raghvender%2C%20S.D.W.%20Wendji%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20G.%20Ori%2C%20P.%20Thomas%2C%20O.%20Masson%2C%20Atomic%20scale%20structure%20and%20dynamical%20properties%20of%20%28TeO2%291-x-%28Na2O%29x%20glasses%20through%20first-principles%20modeling%20and%20XRD%20measurements.%2C%20Physical%20Chemistry%20Chemical%20Physics%2027%20%282025%29%2017884%26%23x2013%3B17899.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd5cp01916h%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd5cp01916h%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Atomic%20scale%20structure%20and%20dynamical%20properties%20of%20%28TeO2%291-x-%28Na2O%29x%20glasses%20through%20first-principles%20modeling%20and%20XRD%20measurements.%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Firas%22%2C%22lastName%22%3A%22Shuaib%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Remi%22%2C%22lastName%22%3A%22Piotrowski%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Gaelle%22%2C%22lastName%22%3A%22Delaizir%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Pierre-Marie%22%2C%22lastName%22%3A%22Geffroy%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22David%22%2C%22lastName%22%3A%22Hamani%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Raghvender%22%2C%22lastName%22%3A%22Raghvender%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Steve%20Dave%20Wansi%22%2C%22lastName%22%3A%22Wendji%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Philippe%22%2C%22lastName%22%3A%22Thomas%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Olivier%22%2C%22lastName%22%3A%22Masson%22%7D%5D%2C%22abstractNote%22%3A%22We%20resort%20to%20first-principles%20molecular%20dynamics%2C%20in%20synergy%20with%20experiments%2C%20to%20study%20structural%20evolution%20and%20Na%2B%20cation%20diffusion%20inside%20%28TeO2%291-x-%28Na2O%29x%20%28x%20%3D%200.10-0.40%29%20glasses.%20Experimental%20and%20modeling%20results%20show%20a%20fair%20quantitative%20agreement%20in%20terms%20of%20total%20X-ray%20structure%20factors%20and%20pair%20distribution%20functions%2C%20thereby%20setting%20the%20ground%20for%20a%20comprehensive%20analysis%20of%20the%20glassy%20matrix%20evolution.%20We%20find%20that%20the%20structure%20of%20%28TeO2%291-x-%28Na2O%29x%20glasses%20deviates%20drastically%20from%20that%20of%20pure%20TeO2%20glass.%20Specifically%2C%20increasing%20the%20Na2O%20concentration%20leads%20to%20a%20reduction%20of%20the%20coordination%20number%20of%20Te%20atoms%2C%20reflecting%20the%20occurrence%20of%20a%20structural%20depolymerization%20upon%20introduction%20of%20the%20Na2O%20modifier%20oxide.%20The%20depolymerization%20phenomenon%20is%20ascribed%20to%20the%20transformation%20of%20Te-O-Te%20bridges%20into%20terminal%20Te-O%20non%20bridging%20oxygen%20atoms%20%28NBO%29.%20Consequently%2C%20the%20concentration%20of%20NBO%20increases%20in%20these%20systems%20as%20the%20concentration%20of%20the%20modifier%20increases%2C%20accompanied%20by%20a%20concomitant%20reduction%20in%20the%20coordination%20number%20of%20Na%20atoms.%20The%20structure%20factors%20results%20show%20a%20prominent%20peak%20at%201.4%20A%20that%20becomes%20more%20and%20more%20pronounced%20as%20the%20Na2O%20concentration%20increases.%20The%20occurrence%20of%20this%20first%20sharp%20diffraction%20peak%20is%20attributed%20to%20the%20growth%20of%20Na-rich%20channels%20inside%20the%20amorphous%20network%2C%20acting%20as%20preferential%20routes%20for%20alkali-ion%20conduction%20inside%20the%20relatively%20stable%20Te-O%20matrix.%20These%20channels%20enhance%20the%20ion%20mobility.%22%2C%22date%22%3A%222025%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fd5cp01916h%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fd5cp01916h%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221463-9084%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T14%3A24%3A08Z%22%7D%7D%2C%7B%22key%22%3A%22FPJK89HZ%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Wansi%20Wendji%20et%20al.%22%2C%22parsedDate%22%3A%222025%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ES.D.%20Wansi%20Wendji%2C%20R.%20Piotrowski%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20C.%20Tug%26%23xE8%3Bne%2C%20F.%20Shuaib%2C%20D.%20Hamani%2C%20P.%20-m.%20Geffroy%2C%20P.%20Thomas%2C%20A.%20Bouzid%2C%20O.%20Masson%2C%20G.%20Delaizir%2C%20G.%20Ori%2C%20Enhanced%20structural%20description%20of%20sodium%20vanadium%20phosphate%20glasses%3A%20A%20combined%20experimental%20and%20molecular%20dynamics%20study%2C%20Journal%20of%20Non-Crystalline%20Solids%20655%20%282025%29%20123420.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2025.123420%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2025.123420%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Enhanced%20structural%20description%20of%20sodium%20vanadium%20phosphate%20glasses%3A%20A%20combined%20experimental%20and%20molecular%20dynamics%20study%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22S.%20D.%22%2C%22lastName%22%3A%22Wansi%20Wendji%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22R.%22%2C%22lastName%22%3A%22Piotrowski%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Christine%22%2C%22lastName%22%3A%22Tug%5Cu00e8ne%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22F.%22%2C%22lastName%22%3A%22Shuaib%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22D.%22%2C%22lastName%22%3A%22Hamani%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22P.%20-m.%22%2C%22lastName%22%3A%22Geffroy%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22P.%22%2C%22lastName%22%3A%22Thomas%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A.%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22O.%22%2C%22lastName%22%3A%22Masson%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22G.%22%2C%22lastName%22%3A%22Delaizir%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%5D%2C%22abstractNote%22%3A%22Structural%20and%20bonding%20insights%20into%20sodium%20vanadium%20phosphate%20%28NVP%29%20glasses%20are%20crucial%20for%20optimizing%20their%20performance%20as%20cathode%20materials%20in%20sodium-ion%20batteries.%20This%20study%20quantitatively%20assesses%20the%20structural%20features%20and%20bonding%20characteristics%20of%20two%20NVP%20glass%20compositions%3A%20%28Na2O%29alpha-%28VxOy%29%281-2%20alpha%29-%28P2O5%29alpha%20with%20alpha%20%3D%200.375%2C%200.285.%20We%20combine%20experimental%20characterization%20%28differential%20scanning%20calorimetry%2C%20X-ray%20diffraction%20and%20X-ray%20photoelectron%20spectroscopy%2C%29%20and%20atomistic%20modeling%20%28classical%20molecular%20dynamics%20%28CMD%29%2C%20and%20Born-Oppenheimer%20molecular%20dynamics%20%28BOMD%29%29.%20This%20work%20provides%20a%20quantitative%20analysis%20of%20the%20different%20VO%20units%20in%20the%20two%20NVP%20glass%20models%2C%20superseding%20previous%20knowledge%20based%20largely%20on%20CMD%20simulations.%20Our%20results%20show%20that%20the%20account%20of%20the%20electronic%20structure%2C%20inherent%20in%20BOMD%20simulations%20is%20essential%20for%20capturing%20the%20V-O%20bonding%20environment.%20This%20includes%20the%20splitting%20of%20the%20V-O%20peak%20in%20the%20pair%20distribution%20function%20due%20to%20both%20short%20V%3DO%20and%20longer%20V-O%20bonds%2C%20a%20higher%20degree%20of%20polymerization%20in%20the%20phosphate%20network%20and%20amore%20significant%20role%20for%20V5%2B%20as%20a%20network%20former.%22%2C%22date%22%3A%222025%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.jnoncrysol.2025.123420%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2025.123420%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220022-3093%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T14%3A25%3A02Z%22%7D%7D%2C%7B%22key%22%3A%223F6RLGTU%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Wendji%20et%20al.%22%2C%22parsedDate%22%3A%222025%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ES.%20Wendji%2C%20R.%20Piotrowski%2C%20A.%20Familiari%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20C.%20Tugene%2C%20F.%20Shuaib%2C%20D.%20Hamani%2C%20P.%20Geffroy%2C%20P.%20Thomas%2C%20A.%20Pedone%2C%20A.%20Bouzid%2C%20O.%20Masson%2C%20G.%20Delaizir%2C%20G.%20Ori%2C%20Structure%2C%20bonding%20and%20ionic%20mobility%20in%20Na-V-P-O%20glasses%20for%20energy%20storage%20applications%2C%20Chemical%20Communications%2061%20%282025%29%2010993%26%23x2013%3B10996.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd5cc00443h%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd5cc00443h%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Structure%2C%20bonding%20and%20ionic%20mobility%20in%20Na-V-P-O%20glasses%20for%20energy%20storage%20applications%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22SDW%22%2C%22lastName%22%3A%22Wendji%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22R%22%2C%22lastName%22%3A%22Piotrowski%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A%22%2C%22lastName%22%3A%22Familiari%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22C%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22C%22%2C%22lastName%22%3A%22Tugene%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22F%22%2C%22lastName%22%3A%22Shuaib%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22D%22%2C%22lastName%22%3A%22Hamani%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22PM%22%2C%22lastName%22%3A%22Geffroy%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22P%22%2C%22lastName%22%3A%22Thomas%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A%22%2C%22lastName%22%3A%22Pedone%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22O%22%2C%22lastName%22%3A%22Masson%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22G%22%2C%22lastName%22%3A%22Delaizir%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%5D%2C%22abstractNote%22%3A%22Na-V-P-O%20glasses%20are%20promising%20materials%20for%20sodium%20ion%20batteries%2C%20and%20yet%20a%20thorough%20understanding%20of%20their%20atomic%20scale%20behavior%20has%20so%20far%20been%20elusive.%20In%20this%20work%20we%20leverage%20structural%20and%20electrochemical%20experiments%20with%20first-principles%20and%20large-scale%20machine%20learning-accelerated%20molecular%20dynamics%20to%20elucidate%20quantitatively%20the%20interplay%20among%20structure%2C%20bonding%2C%20and%20ion%20mobility%20on%20space%20and%20time%20scales%20of%20unprecedented%20extensions.%20We%20unravel%20the%20existence%20of%20a%20broad%20V%20coordination%20distribution%20together%20with%20heterogeneous%20Na-ion%20mobility%20featuring%20percolation%20channels.%20Our%20results%20are%20instrumental%20in%20the%20search%20for%20NVP%20glass%20optimization%20for%20electrochemical%20applications.%22%2C%22date%22%3A%222025%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fd5cc00443h%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fd5cc00443h%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221359-7345%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-07T08%3A02%3A51Z%22%7D%7D%5D%7D
[1]
F. Shuaib, G. Ori, P. Thomas, O. Masson, A. Bouzid, Multikernel similarity-based clustering of amorphous systems and machine-learned interatomic potentials by active learning, Journal of the American Ceramic Society 108 (2025) e20128.
https://doi.org/10.1111/jace.20128.
[1]
F. Shuaib, A. Bouzid, R. Piotrowski, G. Delaizir, P.-M. Geffroy, D. Hamani, R. Raghvender, S.D.W. Wendji, C. Massobrio, M. Boero, G. Ori, P. Thomas, O. Masson, Atomic scale structure and dynamical properties of (TeO2)1-x-(Na2O)x glasses through first-principles modeling and XRD measurements., Physical Chemistry Chemical Physics 27 (2025) 17884–17899.
https://doi.org/10.1039/d5cp01916h.
[1]
S.D. Wansi Wendji, R. Piotrowski, C. Massobrio, M. Boero, C. Tugène, F. Shuaib, D. Hamani, P. -m. Geffroy, P. Thomas, A. Bouzid, O. Masson, G. Delaizir, G. Ori, Enhanced structural description of sodium vanadium phosphate glasses: A combined experimental and molecular dynamics study, Journal of Non-Crystalline Solids 655 (2025) 123420.
https://doi.org/10.1016/j.jnoncrysol.2025.123420.
[1]
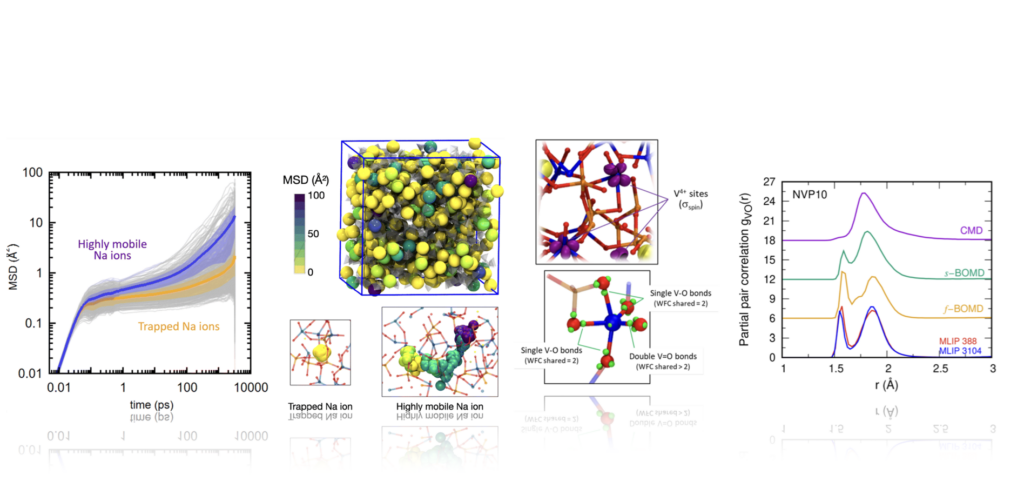
S. Wendji, R. Piotrowski, A. Familiari, C. Massobrio, M. Boero, C. Tugene, F. Shuaib, D. Hamani, P. Geffroy, P. Thomas, A. Pedone, A. Bouzid, O. Masson, G. Delaizir, G. Ori, Structure, bonding and ionic mobility in Na-V-P-O glasses for energy storage applications, Chemical Communications 61 (2025) 10993–10996.
https://doi.org/10.1039/d5cc00443h.
1839302
NZSFH59F
2024
1
surface-science-reports
50
creator
asc
year
7054
https://www.ipcms.unistra.fr/plugins/zotpress/
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3Afalse%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%22MGVK9RHI%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Barbalinardo%20et%20al.%22%2C%22parsedDate%22%3A%222024%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Barbalinardo%2C%20G.%20Ori%2C%20L.%20Lungaro%2C%20G.%20Caio%2C%20A.%20Migliori%2C%20D.%20Gentili%2C%20Direct%20Cationization%20of%20Citrate-Coated%20Gold%20and%20Silver%20Nanoparticles%2C%20Journal%20of%20Physical%20Chemistry%20C%20128%20%282024%29%2016220%26%23x2013%3B16226.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpcc.4c04931%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpcc.4c04931%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Direct%20Cationization%20of%20Citrate-Coated%20Gold%20and%20Silver%20Nanoparticles%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Marianna%22%2C%22lastName%22%3A%22Barbalinardo%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Lisa%22%2C%22lastName%22%3A%22Lungaro%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Giacomo%22%2C%22lastName%22%3A%22Caio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Andrea%22%2C%22lastName%22%3A%22Migliori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Denis%22%2C%22lastName%22%3A%22Gentili%22%7D%5D%2C%22abstractNote%22%3A%22Nanoparticles%20have%20emerged%20as%20promising%20materials%20for%20a%20wide%20range%20of%20applications%2C%20including%20biomedicine%2C%20energy%2C%20and%20electronics.%20However%2C%20controlling%20their%20surface%20chemistry%20is%20essential%20to%20fully%20harnessing%20their%20potential%2C%20as%20it%20affects%20their%20physicochemical%20properties%2C%20stability%2C%20and%20interactions%20with%20biological%20systems.%20Surface%20functionalization%20is%20a%20key%20process%20enabling%20the%20adaptation%20of%20nanoparticle%20properties%20to%20specific%20applications.%20While%20introducing%20ligands%20during%20nanoparticle%20synthesis%20may%20not%20always%20be%20feasible%2C%20ligand%20exchange%20offers%20versatility%20in%20controlling%20surface%20chemistry.%20However%2C%20the%20direct%20replacement%20of%20negatively%20charged%20citrate%20on%20gold%20and%20silver%20nanoparticles%20with%20its%20positive%20counterparts%20often%20leads%20to%20particle%20aggregation.%20Here%2C%20we%20present%20a%20straightforward%20one-step%20ligand%20exchange%20method%20to%20functionalize%20citrate-coated%20gold%20and%20silver%20nanoparticles%20with%20cationic%20ligands.%20By%20controlling%20citrate%20molecule%20protonation%2C%20we%20prevent%20nanoparticle%20aggregation%2C%20enabling%20successful%20displacement%20with%20positively%20charged%20alkanethiol%20ligands.%20Dynamic%20light%20scattering%2C%20zeta-potential%20measurement%2C%20and%20transmission%20electron%20microscopy%20alongside%20theoretical%20models%20provide%20comprehensive%20insights%20into%20the%20mechanism%20and%20dynamics%20of%20ligand%20exchange.%20Furthermore%2C%20we%20demonstrate%20the%20impact%20of%20surface%20functionalization%20of%20nanoparticles%20on%20the%20cytotoxic%20activity%20of%20nanoparticles%20in%20model%20cell%20lines%2C%20underscoring%20the%20significance%20of%20the%20surface%20chemistry%20of%20nanoparticles%20for%20their%20biomedical%20applications.%22%2C%22date%22%3A%222024%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Facs.jpcc.4c04931%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Facs.jpcc.4c04931%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221932-7447%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T15%3A47%3A07Z%22%7D%7D%2C%7B%22key%22%3A%22ADNYDDQV%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Bel-Hadj%20et%20al.%22%2C%22parsedDate%22%3A%222024%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EI.%20Bel-Hadj%2C%20M.%20Guerboub%2C%20A.%20Lambrecht%2C%20G.%20Ori%2C%20C.%20Massobrio%2C%20E.%20Martin%2C%20Thermal%20conductivity%20of%20crystalline%20Ge2Sb2Te5%3A%20lattice%20contribution%20and%20size%20effects%20in%20the%20cubic%20phase%20quantified%20by%20approach-to-equilibrium%20molecular%20dynamics%2C%20Journal%20of%20Physics%20D-Applied%20Physics%2057%20%282024%29%20235303.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1088%5C%2F1361-6463%5C%2Fad316b%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1088%5C%2F1361-6463%5C%2Fad316b%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Thermal%20conductivity%20of%20crystalline%20Ge2Sb2Te5%3A%20lattice%20contribution%20and%20size%20effects%20in%20the%20cubic%20phase%20quantified%20by%20approach-to-equilibrium%20molecular%20dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ibrahim%22%2C%22lastName%22%3A%22Bel-Hadj%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mohammed%22%2C%22lastName%22%3A%22Guerboub%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Achille%22%2C%22lastName%22%3A%22Lambrecht%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%5D%2C%22abstractNote%22%3A%22Approach-to-equilibrium%20molecular%20dynamics%20simulations%20are%20carried%20out%20for%20the%20cubic%20phase%20of%20crystalline%20Ge2Sb2Te5%2C%20using%20interatomic%20forces%20derived%20from%20a%20machine%20learning%20interatomic%20potential%20%28MLIP%29%20trained%20with%20ab%20initio%20calculations.%20The%20use%20of%20this%20MLIP%20potential%20significantly%20reduces%20the%20computational%20burden%2C%20allowing%20for%20the%20study%20of%20systems%20over%2070%20nm%20in%20length.%20Above%2020%20nm%2C%20the%20thermal%20conductivity%20of%20the%20lattice%20plateaus%20at%200.37%20%2B%5C%2F-%200.01%20W%20K-1%20m%28-1%29%20in%20agreement%20with%20measurements%20reported%20in%20the%20literature.%20However%2C%20below%2020%20nm%2C%20size%20effects%20lead%20to%20a%20reduction%20in%20thermal%20conductivity%20which%20is%20systematically%20calculated.%22%2C%22date%22%3A%222024%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1088%5C%2F1361-6463%5C%2Fad316b%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1088%5C%2F1361-6463%5C%2Fad316b%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220022-3727%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22PVWR7FJK%22%5D%2C%22dateModified%22%3A%222026-01-08T15%3A47%3A15Z%22%7D%7D%2C%7B%22key%22%3A%22VDSXEDFS%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Lambrecht%20et%20al.%22%2C%22parsedDate%22%3A%222024%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EA.%20Lambrecht%2C%20G.%20Ori%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20E.%20Martin%2C%20ADynMat%20Consortium%2C%20Assessing%20the%20thermal%20conductivity%20of%20amorphous%20SiN%20by%20approach-to-equilibrium%20molecular%20dynamics%2C%20Journal%20of%20Chemical%20Physics%20160%20%282024%29%20094505.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F5.0193566%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F5.0193566%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Assessing%20the%20thermal%20conductivity%20of%20amorphous%20SiN%20by%20approach-to-equilibrium%20molecular%20dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Achille%22%2C%22lastName%22%3A%22Lambrecht%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22name%22%3A%22ADynMat%20Consortium%22%7D%5D%2C%22abstractNote%22%3A%22First-principles%20molecular%20dynamics%20combined%20with%20the%20approach-to-equilibrium%20molecular%20dynamics%20methodology%20is%20employed%20to%20calculate%20the%20thermal%20conductivity%20of%20non-stoichiometric%20amorphous%20SiN.%20This%20is%20achieved%20by%20implementing%20thermal%20transients%20in%20five%20distinct%20models%20of%20different%20sizes%20along%20the%20direction%20of%20the%20heat%20transport.%20Such%20models%20have%20identical%20structural%20features%20and%20are%20representative%20of%20the%20same%20material%2C%20thereby%20allowing%20for%20a%20reliable%20analysis%20of%20thermal%20conductivity%20trends%20as%20a%20function%20of%20the%20relevant%20cell%20dimension.%20In%20line%20with%20the%20known%20physical%20law%20of%20heat%20propagation%20at%20short%20scale%2C%20the%20thermal%20conductivity%20increases%20in%20size%20with%20the%20direction%20of%20heat%20transport.%20The%20observed%20behavior%20is%20rationalized%20accounting%20for%20previous%20results%20on%20crystalline%20and%20amorphous%20materials%2C%20thus%20providing%20a%20unified%20description%20holding%20for%20a%20large%20class%20of%20materials%20and%20spanning%20a%20wide%20range%20of%20heat%20propagation%20lengths.%22%2C%22date%22%3A%222024%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1063%5C%2F5.0193566%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1063%5C%2F5.0193566%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220021-9606%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T15%3A51%3A59Z%22%7D%7D%2C%7B%22key%22%3A%22D47GXXZ5%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Massobrio%20et%20al.%22%2C%22parsedDate%22%3A%222024%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EC.%20Massobrio%2C%20I.A.%20Essomba%2C%20M.%20Boero%2C%20C.%20Diarra%2C%20M.%20Guerboub%2C%20K.%20Ishisone%2C%20A.%20Lambrecht%2C%20E.%20Martin%2C%20I.%20Morrot-Woisard%2C%20G.%20Ori%2C%20C.%20Tugene%2C%20S.D.%20Wansi%20Wendji%2C%20On%20the%20Actual%20Difference%20between%20the%20Nos%26%23xE9%3B%20and%20the%20Nos%26%23xE9%3B-Hoover%20Thermostats%3A%20A%20Critical%20Review%20of%20Canonical%20Temperature%20Control%20by%20Molecular%20Dynamics%2C%20Physica%20Status%20Solidi%20B-Basic%20Solid%20State%20Physics%20261%20%282024%29%202300209.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Fpssb.202300209%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Fpssb.202300209%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22On%20the%20Actual%20Difference%20between%20the%20Nos%5Cu00e9%20and%20the%20Nos%5Cu00e9-Hoover%20Thermostats%3A%20A%20Critical%20Review%20of%20Canonical%20Temperature%20Control%20by%20Molecular%20Dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Irene%20Amiehe%22%2C%22lastName%22%3A%22Essomba%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Cheick%22%2C%22lastName%22%3A%22Diarra%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mohammed%22%2C%22lastName%22%3A%22Guerboub%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kana%22%2C%22lastName%22%3A%22Ishisone%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Achille%22%2C%22lastName%22%3A%22Lambrecht%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Icare%22%2C%22lastName%22%3A%22Morrot-Woisard%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Christine%22%2C%22lastName%22%3A%22Tugene%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Steve%20Dave%22%2C%22lastName%22%3A%22Wansi%20Wendji%22%7D%5D%2C%22abstractNote%22%3A%22This%20article%20has%20to%20be%20intended%20both%20as%20a%20review%20and%20as%20an%20historical%20tribute%20to%20the%20ideas%20developed%20almost%2040%20years%20ago%20by%20S.%20Nose%2C%20establishing%20the%20theoretical%20foundations%20of%20the%20implementation%20and%20use%20of%20thermostats%20in%20molecular%20dynamics%20%28MDs%29.%20The%20original%20motivation%20of%20this%20work%20is%20enriched%20by%20an%20observation%20related%20to%20the%20connection%20between%20the%20Nose%20seminal%20expression%20of%20temperature%20control%20and%20the%20extension%20proposed%20in%201985%20by%20W.%20G.%20Hoover%2C%20known%20as%20the%20Nose-Hoover%20thermostat.%20By%20carefully%20rederiving%20the%20equations%20of%20motion%20in%20both%20formalisms%2C%20it%20appears%20that%20all%20features%20of%20Nose-Hoover%20framework%20%28replacement%20of%20the%20Nose%20variables%20by%20a%20single%20friction%20coefficient%20in%20the%20equations%20of%20motion%29%20are%20already%20built%20in%20the%20Nose%20approach.%20Therefore%2C%20one%20is%20able%20to%20work%20directly%20within%20the%20Nose%20formalism%20with%20the%20addition%20of%20a%20single%20variable%20only%2C%20by%20greatly%20extending%20its%20general%20impact%20and%20simplicity%20and%20somewhat%20making%20redundant%20the%20Nose-Hoover%20extension.%20Having%20been%20implicitly%20%28and%20somewhat%20inadvertently%29%20put%20to%20good%20use%20by%20a%20multitude%20of%20users%20over%20the%20past%2040%20years%20%28in%20the%20context%20of%20classical%20and%20first-principles%20MDs%29%2C%20this%20finding%20does%20not%20need%20any%20specific%20application%20to%20be%20assessed.This%20paper%20is%20both%20a%20review%20and%20historical%20tribute%20to%20the%20ideas%20developed%20almost%2040%20years%20ago%20by%20S.%20Nose%20establishing%20the%20theoretical%20foundations%20of%20implementation%20and%20use%20of%20thermostats%20in%20molecular%20dynamics.%20By%20carefully%20rederiving%20the%20equations%20of%20motion%20in%20both%20formalisms%2C%20it%20appears%20that%20all%20features%20of%20Nose-Hoover%20framework%20%28replacement%20of%20the%20Nose%20variables%20by%20a%20single-friction%20coefficient%20in%20the%20equations%20of%20motion%29%20are%20already%20built%20in%20the%20Nose%20approach.image%20%28c%29%202023%20WILEY-VCH%20GmbH%22%2C%22date%22%3A%222024%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1002%5C%2Fpssb.202300209%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1002%5C%2Fpssb.202300209%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220370-1972%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T15%3A52%3A23Z%22%7D%7D%2C%7B%22key%22%3A%228ABA5LXH%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Morrot-Woisard%20et%20al.%22%2C%22parsedDate%22%3A%222024%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EI.%20Morrot-Woisard%2C%20E.K.%20Nguyen%2C%20N.%20Vukadinovic%2C%20M.%20Boero%2C%20Structural%2C%20electronic%20and%20dielectric%20properties%20of%20carbon%20nanotubes%20interacting%20with%20Co%20nanoclusters%2C%20Carbon%20Trends%2017%20%282024%29%20100410.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cartre.2024.100410%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cartre.2024.100410%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Structural%2C%20electronic%20and%20dielectric%20properties%20of%20carbon%20nanotubes%20interacting%20with%20Co%20nanoclusters%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Icare%22%2C%22lastName%22%3A%22Morrot-Woisard%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Emile%20K.%22%2C%22lastName%22%3A%22Nguyen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Nicolas%22%2C%22lastName%22%3A%22Vukadinovic%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22A%20new%20frontier%20in%20tuning%20the%20electromagnetic%20response%20of%20carbon-based%20materials%2C%20particularly%20carbon%20nanotubes%2C%20consists%20in%20adding%20metallic%20clusters%20or%20nanoparticles%20at%20their%20external%20surface.%20This%20allows%20to%20change%20optical%2C%20dielectric%20and%20magnetic%20properties%20with%20potential%20applications%20in%20electronics%20and%20telecommunications%20for%20aeronautics.%20By%20resorting%20to%20first%20principles%20dynamical%20simulations%2C%20we%20provide%20a%20microscopic%20picture%20of%20the%20interaction%20at%20finite%20temperature%20between%20a%20carbon%20nanotube%20and%20Co%20aggregates%20mimicking%20the%20experimental%20coverage.%20The%20electronic%20structure%20evolution%20provides%20the%20absorption%20spectrum%20and%20the%20dielectric%20function%20for%20comparison%20with%20experiments%20and%20guidelines%20for%20tuning%20these%20composite%20systems.%22%2C%22date%22%3A%222024%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.cartre.2024.100410%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.cartre.2024.100410%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222667-0569%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222025-12-23T10%3A46%3A06Z%22%7D%7D%2C%7B%22key%22%3A%22VUBMQHCL%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Wansi%20Wendji%20et%20al.%22%2C%22parsedDate%22%3A%222024%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ES.D.%20Wansi%20Wendji%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20C.%20Tug%26%23xE8%3Bne%2C%20E.%20Levchenko%2C%20F.%20Shuaib%2C%20R.%20Piotrowski%2C%20D.%20Hamani%2C%20G.%20Delaizir%2C%20P.-M.%20Geffroy%2C%20P.%20Thomas%2C%20O.%20Masson%2C%20A.%20Bouzid%2C%20G.%20Ori%2C%20Quantitative%20assessment%20of%20the%20structure%20and%20bonding%20properties%20of%2050VxOy-50P2O5%20glass%20by%20classical%20and%20Born%26%23x2013%3BOppenheimer%20molecular%20dynamics%2C%20Journal%20of%20Non-Crystalline%20Solids%20634%20%282024%29%20122967.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2Fhttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2024.122967%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2Fhttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2024.122967%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Quantitative%20assessment%20of%20the%20structure%20and%20bonding%20properties%20of%2050VxOy-50P2O5%20glass%20by%20classical%20and%20Born%5Cu2013Oppenheimer%20molecular%20dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22S.%20D.%22%2C%22lastName%22%3A%22Wansi%20Wendji%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Christine%22%2C%22lastName%22%3A%22Tug%5Cu00e8ne%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22E.%22%2C%22lastName%22%3A%22Levchenko%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22F.%22%2C%22lastName%22%3A%22Shuaib%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22R.%22%2C%22lastName%22%3A%22Piotrowski%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22D.%22%2C%22lastName%22%3A%22Hamani%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22G.%22%2C%22lastName%22%3A%22Delaizir%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22P.-M.%22%2C%22lastName%22%3A%22Geffroy%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22P.%22%2C%22lastName%22%3A%22Thomas%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22O.%22%2C%22lastName%22%3A%22Masson%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A.%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%5D%2C%22abstractNote%22%3A%22The%20structure%20and%20bonding%20of%20VxOy%5Cu2013P2O5%20%28VP50%29%20glass%20is%20studied%20by%20classical%20and%20Born%5Cu2013Oppenheimer%20molecular%20dynamics%20%28CMD%20and%20BOMD%29.%20By%20testing%20the%20performance%20of%20three%20different%20empirical%20CMD%20force%20fields%20sets%20and%20through%20a%20thorough%20comparison%20with%20experimental%20data%2C%20our%20study%20showcases%20the%20effectiveness%20of%20BOMD%20in%20addressing%20the%20shortcomings%20of%20CMD%20and%20achieves%20a%20significantly%20improved%20and%20superior%20quantitative%20description%20of%20VP50%20glass%5Cu2019%20structures.%20BOMD%20allows%20us%20to%20achieve%20an%20unprecedented%20agreement%20with%20experimental%20data%20in%20terms%20of%20both%20reciprocal%20space%20%28neutron%20and%20X-ray%20structure%20factors%29%20and%20real%20space%20%28total%20pair%20correlation%20functions%29%20properties.%20The%20key%20improvement%20is%20ascribed%20to%20a%20better%20description%20of%20the%20local%20electronic%20and%20bonding%20environment%20around%20both%20P%20and%2C%20especially%2C%20V%20sites%20that%20cannot%20be%20obtained%20by%20empirical%20force%20fields.%20A%20clear%20signature%20of%20single%20and%20double%20bonds%20is%20found%20with%20BOMD%20together%20with%20an%20overall%20better%20description%20of%20VOn%20coordinating%20polyhedra%20distribution%20that%20constitute%20the%20network%20of%20this%20glass.%20Our%20study%20is%20enriched%20by%20a%20thorough%20analysis%20of%20bond%20angle%20distributions%20around%20VOn%20units%2C%20order%20and%20connectivity%20parameters%20and%20local%20bonding%20features%20based%20on%20Wannier%20functions%20formalism.%20As%20a%20byproduct%20of%20this%20work%2C%20we%20assessed%20which%20CMD%20scheme%20aligns%20more%20closely%20with%20the%20BOMD%20data%20and%20experimental%20findings.%20Our%20analysis%20revealed%20that%20including%20three-body%20potential%20parameters%20associated%20with%20the%20local%20environment%20of%20V%20sites%20significantly%20improves%20the%20performance%20of%20CMD.%20However%2C%20while%20enhanced%2C%20CMD%20still%20falls%20short%20of%20achieving%20the%20full%20accuracy%20demonstrated%20by%20BOMD%20in%20describing%20VP50%20glass.%20All%20together%2C%20the%20results%20found%20define%20the%20computational%20grounds%20for%20a%20deep%20understating%20of%20VP%20amorphous%20glasses%20in%20conjunctions%20with%20depicting%20the%20necessary%20requirements%20for%20the%20development%20of%20interatomic%20potentials%20aiming%20to%20a%20quantitative%20comprehension%20and%20design%20of%20VP-based%20amorphous%20materials.%22%2C%22date%22%3A%222024%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%22https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2024.122967%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.sciencedirect.com%5C%2Fscience%5C%2Farticle%5C%2Fpii%5C%2FS0022309324001480%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220022-3093%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T15%3A55%3A05Z%22%7D%7D%5D%7D
[1]
M. Barbalinardo, G. Ori, L. Lungaro, G. Caio, A. Migliori, D. Gentili, Direct Cationization of Citrate-Coated Gold and Silver Nanoparticles, Journal of Physical Chemistry C 128 (2024) 16220–16226.
https://doi.org/10.1021/acs.jpcc.4c04931.
[1]
I. Bel-Hadj, M. Guerboub, A. Lambrecht, G. Ori, C. Massobrio, E. Martin, Thermal conductivity of crystalline Ge2Sb2Te5: lattice contribution and size effects in the cubic phase quantified by approach-to-equilibrium molecular dynamics, Journal of Physics D-Applied Physics 57 (2024) 235303.
https://doi.org/10.1088/1361-6463/ad316b.
[1]
A. Lambrecht, G. Ori, C. Massobrio, M. Boero, E. Martin, ADynMat Consortium, Assessing the thermal conductivity of amorphous SiN by approach-to-equilibrium molecular dynamics, Journal of Chemical Physics 160 (2024) 094505.
https://doi.org/10.1063/5.0193566.
[1]
C. Massobrio, I.A. Essomba, M. Boero, C. Diarra, M. Guerboub, K. Ishisone, A. Lambrecht, E. Martin, I. Morrot-Woisard, G. Ori, C. Tugene, S.D. Wansi Wendji, On the Actual Difference between the Nosé and the Nosé-Hoover Thermostats: A Critical Review of Canonical Temperature Control by Molecular Dynamics, Physica Status Solidi B-Basic Solid State Physics 261 (2024) 2300209.
https://doi.org/10.1002/pssb.202300209.
[1]
I. Morrot-Woisard, E.K. Nguyen, N. Vukadinovic, M. Boero, Structural, electronic and dielectric properties of carbon nanotubes interacting with Co nanoclusters, Carbon Trends 17 (2024) 100410.
https://doi.org/10.1016/j.cartre.2024.100410.
[1]
S.D. Wansi Wendji, C. Massobrio, M. Boero, C. Tugène, E. Levchenko, F. Shuaib, R. Piotrowski, D. Hamani, G. Delaizir, P.-M. Geffroy, P. Thomas, O. Masson, A. Bouzid, G. Ori, Quantitative assessment of the structure and bonding properties of 50VxOy-50P2O5 glass by classical and Born–Oppenheimer molecular dynamics, Journal of Non-Crystalline Solids 634 (2024) 122967.
https://doi.org/https://doi.org/10.1016/j.jnoncrysol.2024.122967.
1839302
NZSFH59F
2023
1
surface-science-reports
50
creator
asc
year
7054
https://www.ipcms.unistra.fr/plugins/zotpress/
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3Afalse%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%22QX92KDSE%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Diarra%20et%20al.%22%2C%22parsedDate%22%3A%222023%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EC.O.%20Diarra%2C%20M.%20Boero%2C%20E.%20Steveler%2C%20T.%20Heiser%2C%20E.%20Martin%2C%20Exciton%20diffusion%20in%20poly%283-hexylthiophene%29%20by%20first-principles%20molecular%20dynamics.%2C%20Physical%20Chemistry%20Chemical%20Physics%2025%20%282023%29%2015539%26%23x2013%3B15546.%20%3Ca%20class%3D%27zp-ItemURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd3cp00533j%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd3cp00533j%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Exciton%20diffusion%20in%20poly%283-hexylthiophene%29%20by%20first-principles%20molecular%20dynamics.%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Cheick%20Oumar%22%2C%22lastName%22%3A%22Diarra%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Emilie%22%2C%22lastName%22%3A%22Steveler%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Thomas%22%2C%22lastName%22%3A%22Heiser%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%5D%2C%22abstractNote%22%3A%22Poly%283-hexylthiophene%29%20%28P3HT%29%20is%20a%20polymer%20used%20in%20organic%20solar%20cells%20as%20a%20light%20absorber%20and%20an%20electron%20donor.%20Photogenerated%20excitons%20diffuse%20and%20dissociate%20into%20free%20charge%20carriers%20provided%20they%20reach%20the%20absorber%20boundaries.%20The%20device%20efficiency%20is%20therefore%20dependent%20on%20the%20exciton%20diffusion.%20Although%20measurements%20can%20be%20performed%20for%20example%20by%20time-resolved%20photoluminescence%2C%20a%20quantitative%20modeling%20is%20highly%20desirable%20to%20get%20an%20insight%20into%20the%20relationship%20between%20the%20atomic%20structure%20at%20finite%20temperature%20and%20the%20diffusion%20coefficient%20of%20the%20exciton.%20This%20is%20the%20objective%20of%20the%20present%20work%2C%20achieved%20by%20resorting%20to%20first-principles%20molecular%20dynamics%20in%20combination%20with%20the%20restricted%20open-shell%20approach%20to%20model%20the%20singlet%20excited%20state.%20The%20maximally%20localized%20Wannier%20functions%20and%20their%20centers%20are%20used%20to%20monitor%20and%20localize%20the%20electron%20and%20the%20hole%20along%20the%20dynamics.%20The%20resulting%20diffusion%20coefficient%20is%20in%20close%20agreement%20with%20available%20measurements.%22%2C%22date%22%3A%222023%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fd3cp00533j%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd3cp00533j%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T15%3A57%3A12Z%22%7D%7D%2C%7B%22key%22%3A%22P3ISC2FL%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Guerboub%20et%20al.%22%2C%22parsedDate%22%3A%222023%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Guerboub%2C%20S.D.%20Wansi%20Wendji%2C%20C.%20Massobrio%2C%20A.%20Bouzid%2C%20M.%20Boero%2C%20G.%20Ori%2C%20E.%20Martin%2C%20Impact%20of%20the%20local%20atomic%20structure%20on%20the%20thermal%20conductivity%20of%20amorphous%20Ge2Sb2Te5.%2C%20Journal%20of%20Chemical%20Physics%20158%20%282023%29%20084504.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F5.0139590%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F5.0139590%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Impact%20of%20the%20local%20atomic%20structure%20on%20the%20thermal%20conductivity%20of%20amorphous%20Ge2Sb2Te5.%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mohammed%22%2C%22lastName%22%3A%22Guerboub%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Steve%20Dave%22%2C%22lastName%22%3A%22Wansi%20Wendji%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%5D%2C%22abstractNote%22%3A%22Thermal%20properties%20are%20expected%20to%20be%20sensitive%20to%20the%20network%20topology%2C%20and%20however%2C%20no%20clearcut%20information%20is%20available%20on%20how%20the%20thermal%20conductivity%20of%20amorphous%20systems%20is%20affected%20by%20details%20of%20the%20atomic%20structure.%20To%20address%20this%20issue%2C%20we%20use%20as%20a%20target%20system%20a%20phase-change%20amorphous%20material%20%28i.e.%2C%20Ge2Sb2Te5%29%20simulated%20by%20first-principles%20molecular%20dynamics%20combined%20with%20the%20approach-to-equilibrium%20molecular%20dynamics%20technique%20to%20access%20the%20thermal%20conductivity.%20Within%20the%20density-functional%20theory%2C%20we%20employed%20two%20models%20sharing%20the%20same%20exchange-correlation%20functional%20but%20differing%20in%20the%20pseudopotential%20%28PP%29%20implementation%20%5Bnamely%2C%20Trouiller-Martins%20%28TM%29%20and%20Goedecker%2C%20Teter%2C%20and%20Hutter%20%28GTH%29%5D.%20They%20are%20both%20compatible%20with%20experimental%20data%2C%20and%20however%2C%20the%20TM%20PP%20construction%20results%20in%20a%20Ge%20tetrahedral%20environment%20largely%20predominant%20over%20the%20octahedral%20one%2C%20although%20the%20proportion%20of%20tetrahedra%20is%20considerably%20smaller%20when%20the%20GTH%20PP%20is%20used.%20We%20show%20that%20the%20difference%20in%20the%20local%20structure%20between%20TM%20and%20GTH%20models%20impacts%20the%20vibrational%20density%20of%20states%20while%20the%20thermal%20conductivity%20does%20not%20feature%20any%20appreciable%20sensitivity%20to%20such%20details.%20This%20behavior%20is%20rationalized%20in%20terms%20of%20extended%20vibrational%20modes.%22%2C%22date%22%3A%222023%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1063%5C%2F5.0139590%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1063%5C%2F5.0139590%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221089-7690%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T15%3A59%3A10Z%22%7D%7D%2C%7B%22key%22%3A%22I6R7GWXU%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Lawes%20et%20al.%22%2C%22parsedDate%22%3A%222023%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EP.%20Lawes%2C%20M.%20Boero%2C%20R.%20Barhoumi%2C%20S.%20Klyatskaya%2C%20M.%20Ruben%2C%20J.-P.%20Bucher%2C%20Hierarchical%20Self-Assembly%20and%20Conformation%20of%20Tb%20Double-Decker%20Molecular%20Magnets%3A%20Experiment%20and%20Molecular%20Dynamics%2C%20Nanomaterials%2013%20%282023%29%202232.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3390%5C%2Fnano13152232%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3390%5C%2Fnano13152232%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Hierarchical%20Self-Assembly%20and%20Conformation%20of%20Tb%20Double-Decker%20Molecular%20Magnets%3A%20Experiment%20and%20Molecular%20Dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Patrick%22%2C%22lastName%22%3A%22Lawes%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Rabei%22%2C%22lastName%22%3A%22Barhoumi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Svetlana%22%2C%22lastName%22%3A%22Klyatskaya%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mario%22%2C%22lastName%22%3A%22Ruben%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jean-Pierre%22%2C%22lastName%22%3A%22Bucher%22%7D%5D%2C%22abstractNote%22%3A%22Nanostructures%2C%20fabricated%20by%20locating%20molecular%20building%20blocks%20in%20well-defined%20positions%2C%20for%20example%2C%20on%20a%20lattice%2C%20are%20ideal%20platforms%20for%20studying%20atomic-scale%20quantum%20effects.%20In%20this%20context%2C%20STM%20data%20obtained%20from%20self-assembled%20Bis%28phthalocyaninato%29%20Terbium%20%28III%29%20%28TbPc2%29%20single-molecule%20magnets%20on%20various%20substrates%20have%20raised%20questions%20about%20the%20conformation%20of%20the%20TbPc2%20molecules%20within%20the%20lattice.%20In%20order%20to%20address%20this%20issue%2C%20molecular%20dynamics%20simulations%20were%20carried%20out%20on%20a%202D%20assembly%20of%20TbPc2%20molecules.%20The%20calculations%20are%20in%20excellent%20agreement%20with%20the%20experiment%2C%20and%20thus%20improve%20our%20understanding%20of%20the%20self-assembly%20process.%20In%20particular%2C%20the%20calculated%20electron%20density%20of%20the%20molecular%20assembly%20compares%20well%20with%20STM%20contrast%20of%20self-assembled%20TbPc2%20on%20Au%28111%29%2C%20simultaneously%20providing%20the%20conformation%20of%20the%20two%20Pc%20ligands%20of%20the%20individual%20double-decker%20molecule.%20This%20approach%20proves%20valuable%20in%20the%20identification%20of%20the%20STM%20contrast%20of%20LnPc%282%29%20layers%20and%20could%20be%20used%20in%20similar%20cases%20where%20it%20is%20difficult%20to%20interpret%20the%20STM%20images%20of%20an%20assembly%20of%20molecular%20complexes.%22%2C%22date%22%3A%222023%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.3390%5C%2Fnano13152232%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.3390%5C%2Fnano13152232%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222079-4991%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222025-12-23T09%3A43%3A54Z%22%7D%7D%2C%7B%22key%22%3A%22U3DAY9IH%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Pham%20et%20al.%22%2C%22parsedDate%22%3A%222023%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ET.-L.%20Pham%2C%20W.I.%20Choi%2C%20A.%20Shafique%2C%20H.J.%20Kim%2C%20M.%20Shim%2C%20K.%20Min%2C%20W.-J.%20Son%2C%20I.%20Jang%2C%20D.S.%20Kim%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20G.%20Ori%2C%20H.S.%20Lee%2C%20Y.-H.%20Shin%2C%20Structural-Stability%20Study%20of%20Antiperovskite%20Na3OCl%20for%20Na-Rich%20Solid%20Electrolyte%2C%20Physical%20Review%20Applied%2019%20%282023%29%20034004.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevApplied.19.034004%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevApplied.19.034004%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Structural-Stability%20Study%20of%20Antiperovskite%20Na3OCl%20for%20Na-Rich%20Solid%20Electrolyte%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tan%20-Lien%22%2C%22lastName%22%3A%22Pham%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Woon%20Ih%22%2C%22lastName%22%3A%22Choi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Aamir%22%2C%22lastName%22%3A%22Shafique%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hye%20Jung%22%2C%22lastName%22%3A%22Kim%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Munbo%22%2C%22lastName%22%3A%22Shim%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kyoungmin%22%2C%22lastName%22%3A%22Min%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Won-Joon%22%2C%22lastName%22%3A%22Son%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Inkook%22%2C%22lastName%22%3A%22Jang%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Dae%20Sin%22%2C%22lastName%22%3A%22Kim%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hyo%20Sug%22%2C%22lastName%22%3A%22Lee%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Young%20-Han%22%2C%22lastName%22%3A%22Shin%22%7D%5D%2C%22abstractNote%22%3A%22The%20structural%20phase%20transition%20of%20the%20high-symmetry%20cubic%20phase%20of%20antiperovskite%20Na3OCl%20is%20investigated%20by%20computing%20the%20phonon%20band%20structures%20of%2014%20different%20polymorphs%20with%20distinct%20types%20of%20ONa6%20octahedral%20tilting.%20The%20resulting%20P-T%20phase%20diagram%20shows%20that%2C%20at%20high%20temperature%20and%20low%20pressure%2C%20the%20high-symmetry%20cubic%20structure%20with%20Pm3%20over%20bar%20m%20symmetry%20is%20the%20most%20stable%20phase.%20At%20low%20temperature%20and%20high%20pressure%2C%20on%20the%20other%20hand%2C%20the%20monoclinic%20structure%20with%20P21%5C%2Fm%20symmetry%20becomes%20the%20most%20stable%20phase.%20In%20between%20those%20two%2C%20there%20is%20a%20region%20in%20the%20phase%20diagram%20where%20the%20orthorhombic%20structure%20with%20Bmmb%20symmetry%20is%20the%20most%20stable%20phase.%20To%20improve%20upon%20the%20quasiharmonic%20results%2C%20we%20do%20additional%20calculations%20in%20the%20framework%20of%20the%20self-consistent%20phonon%20%28SCP%29%20theory%2C%20including%20lattice%20anharmonicity%20by%20using%20cubic%20and%20quartic%20interatomic%20force%20constants%20%28IFCs%29.%20This%20is%20particularly%20important%20for%20the%20highsymmetry%20cubic%20phase.%20We%20find%20that%20by%20decreasing%20the%20temperature%2C%20the%20frequency%20of%20the%20soft%20phonon%20at%20the%20M%20and%20R%20symmetry%20points%20gradually%20shifts%20to%20lower%20values.%20From%20these%20results%2C%20we%20can%20infer%20that%20a%20phase%20transition%20occurs%20around%20166-195%20K%20upon%20soft-mode%20condensation.%20Due%20to%20the%20proximity%20of%20the%20soft-mode%20frequencies%20at%20both%20symmetry%20points%20R%20and%20M%2C%20we%20expect%20a%20cubic-to-orthorhombic%20phase%20transition%20to%20be%20realized%20via%20simultaneous%20condensation%20of%20the%20two%20octahedral%20tilting%20modes.%22%2C%22date%22%3A%222023%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevApplied.19.034004%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevApplied.19.034004%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222331-7019%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A02%3A29Z%22%7D%7D%2C%7B%22key%22%3A%22B8KXKHXS%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Pham%20et%20al.%22%2C%22parsedDate%22%3A%222023%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ET.-L.%20Pham%2C%20M.%20Guerboub%2C%20A.%20Bouzid%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20Y.-H.%20Shin%2C%20G.%20Ori%2C%20Unveiling%20the%20structure%20and%20ion%20dynamics%20of%20amorphous%20Na%3Csub%3E3-%3Ci%3Ex%3C%5C%2Fi%3E%3C%5C%2Fsub%3EOH%3Csub%3E%3Ci%3Ex%3C%5C%2Fi%3E%3C%5C%2Fsub%3ECl%20antiperovskite%20electrolytes%20by%20first-principles%20molecular%20dynamics%2C%20Journal%20of%20Materials%20Chemistry%20A%2011%20%282023%29%2022922%26%23x2013%3B22940.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd3ta01373a%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd3ta01373a%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Unveiling%20the%20structure%20and%20ion%20dynamics%20of%20amorphous%20Na%3Csub%3E3-%3Ci%3Ex%3C%5C%2Fi%3E%3C%5C%2Fsub%3EOH%3Csub%3E%3Ci%3Ex%3C%5C%2Fi%3E%3C%5C%2Fsub%3ECl%20antiperovskite%20electrolytes%20by%20first-principles%20molecular%20dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tan-Lien%22%2C%22lastName%22%3A%22Pham%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mohammed%22%2C%22lastName%22%3A%22Guerboub%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Young-Han%22%2C%22lastName%22%3A%22Shin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%5D%2C%22abstractNote%22%3A%22Sodium%20oxyhalide%20and%20hydroxyhalide%20antiperovskites%20are%20promising%20solid-state%20electrolytes%20%28SSEs%29%20because%20of%20their%20low%20melting%20point%20and%20rapid%20synthesis%20and%2C%20as%20such%2C%20they%20are%20becoming%20competitive%20with%20respect%20to%20other%20systems.%20While%20the%20structure%20and%20the%20mechanism%20underlying%20the%20ion%20dynamics%20are%20increasingly%20well%20understood%20in%20crystalline%20antiperovskites%2C%20their%20amorphous%20counterpart%20lacks%20precise%20structural%20characterization%2C%20hampering%20any%20conclusive%20insight%20into%20their%20properties.%20In%20this%20work%2C%20we%20resort%20to%20first-principles%20molecular%20dynamics%20within%20the%20Car-Parrinello%20scheme%20to%20assess%20the%20structure%20and%20ion%20dynamics%20of%20amorphous%20Na3-xOHxCl%20%28with%20x%20%3D%200%2C%200.5%2C%20and%201%29%20antiperovskites%20at%20a%20quantitative%20level.%20We%20obtain%20a%20detailed%20structural%20description%20of%20these%20amorphous%20systems%2C%20unveiling%20the%20mechanism%20inherent%20to%20the%20dynamics%20of%20Na%20ions%2C%20the%20role%20of%20H%20atoms%2C%20and%20the%20resulting%20ionic%20conductivity.%20Our%20results%20demonstrate%20that%20the%20structure%20of%20amorphous%20Na3OCl%20significantly%20differs%20from%20its%20crystal%20phase%2C%20showing%20very%20limited%20intermediate-range%20order%20and%20a%20short-range%20order%20mainly%20driven%20by%20four-fold%20Na%20atoms.%20Our%20results%20reveal%20that%20there%20is%20no%20evidence%20of%20phase%20separation%20in%20the%20amorphous%20Na3-xOHxCl%2C%20unlike%20the%20previous%20conjectured%20model%20of%20glassy%20Li3OCl.%20The%20amorphous%20structure%20of%20Na3OCl%20features%20remarkable%20Na%20ion%20dynamics%20and%20ionic%20conductivity%2C%20rivaling%20that%20of%20defective%20crystalline%20phases%20and%20highlighting%20its%20potential%20as%20a%20promising%20solid-state%20electrolyte.%20In%20hydroxylated%20models%2C%20the%20presence%20of%20hydroxyl%20OH-%20anions%20plays%20a%20crucial%20role%20in%20the%20mobility%20of%20Na%20ions.%20This%20is%20facilitated%20by%20the%20rapid%20rotation%20of%20O-H%20bonds%20and%20paddlewheel-type%20mechanisms%2C%20leading%20to%20enhanced%20ion%20mobility%20in%20the%20amorphous%20Na3-xOHxCl%20systems.%20This%20work%20provides%20unprecedented%20physical%20and%20chemical%20insight%20into%20the%20interplay%20between%20the%20structure%2C%20bonding%2C%20and%20ion%20transport%20in%20amorphous%20sodium-rich%20oxyhalide%20and%20hydroxyhalide%20antiperovskites%2C%20paving%20the%20way%20to%20their%20practical%20realization%20in%20next-generation%20SSEs.%22%2C%22date%22%3A%222023%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fd3ta01373a%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fd3ta01373a%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222050-7488%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A02%3A32Z%22%7D%7D%2C%7B%22key%22%3A%22Z4RWP5MK%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Sato%20et%20al.%22%2C%22parsedDate%22%3A%222023%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EA.%20Sato%2C%20M.%20Shoji%2C%20N.%20Watanabe%2C%20M.%20Boero%2C%20Y.%20Shigeta%2C%20M.%20Umemura%2C%20Origin%20of%20Homochirality%20in%20Amino%20Acids%20Induced%20by%20Lyman-alpha%20Irradiation%20in%20the%20Early%20Stage%20of%20the%20Milky%20Way.%2C%20Astrobiology%2023%20%282023%29%201019%26%23x2013%3B1026.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1089%5C%2Fast.2022.0140%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1089%5C%2Fast.2022.0140%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Origin%20of%20Homochirality%20in%20Amino%20Acids%20Induced%20by%20Lyman-alpha%20Irradiation%20in%20the%20Early%20Stage%20of%20the%20Milky%20Way.%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Akimasa%22%2C%22lastName%22%3A%22Sato%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mitsuo%22%2C%22lastName%22%3A%22Shoji%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Natsuki%22%2C%22lastName%22%3A%22Watanabe%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yasuteru%22%2C%22lastName%22%3A%22Shigeta%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masayuki%22%2C%22lastName%22%3A%22Umemura%22%7D%5D%2C%22abstractNote%22%3A%22The%20enantiomeric%20excess%20%28ee%29%20of%20l-form%20amino%20acids%20found%20in%20the%20Murchison%20meteorite%20poses%20some%20issues%20about%20the%20cosmic%20origin%20of%20their%20chirality.%20Circular%20dichroism%20%28CD%29%20spectra%20of%20amino%20acids%20in%20the%20far-ultraviolet%20%28FUV%29%20at%20around%206.8eV%20%28182nm%29%20indicate%20that%20the%20circularly%20polarized%20light%20can%20induce%20ee%20through%20photochemical%20reactions.%20Here%2C%20we%20resort%20to%20ab%20initio%20calculations%20to%20extract%20the%20CD%20spectra%20up%20to%20the%20vacuum-ultraviolet%20%28VUV%29%20region%20%2811eV%29%2C%20and%20we%20propose%20a%20novel%20equation%20to%20compute%20the%20ee%20applicable%20to%20a%20wider%20range%20of%20light%20frequency%20than%20what%20is%20available%20to%20date.%20This%20allows%20us%20to%20show%20that%20the%20strength%20of%20the%20induced%20ee%20%28%7Cee%7C%29%20in%20the%2010eV%20VUV%20region%20is%20comparable%20to%20the%20one%20in%20the%206.8eV%20FUV%20region.%20This%20feature%20is%20common%20for%20some%20key%20amino%20acids%20%28alanine%2C%202-aminobutyric%20acid%2C%20and%20valine%29.%20In%20space%2C%20intense%20Lyman-alpha%20%28Lyalpha%29%20light%20of%2010.2eV%20is%20emitted%20from%20star%20forming%20regions.%20This%20study%20provides%20a%20theoretical%20basis%20that%20Lyalpha%20emitter%20from%20an%20early%20starburst%20in%20the%20Milky%20Way%20plays%20a%20crucial%20role%20in%20initiating%20the%20ee%20of%20amino%20acids.%22%2C%22date%22%3A%222023%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1089%5C%2Fast.2022.0140%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1089%5C%2Fast.2022.0140%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A03%3A06Z%22%7D%7D%2C%7B%22key%22%3A%22QW7J5BUL%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Shoji%20et%20al.%22%2C%22parsedDate%22%3A%222023%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Shoji%2C%20Y.%20Kitazawa%2C%20A.%20Sato%2C%20N.%20Watanabe%2C%20M.%20Boero%2C%20Y.%20Shigeta%2C%20M.%20Umemura%2C%20Enantiomeric%20Excesses%20of%20Aminonitrile%20Precursors%20Determine%20the%20Homochirality%20of%20Amino%20Acids.%2C%20Journal%20of%20Physical%20Chemistry%20Letters%2014%20%282023%29%203243%26%23x2013%3B3248.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpclett.2c03862%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpclett.2c03862%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Enantiomeric%20Excesses%20of%20Aminonitrile%20Precursors%20Determine%20the%20Homochirality%20of%20Amino%20Acids.%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mitsuo%22%2C%22lastName%22%3A%22Shoji%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yuya%22%2C%22lastName%22%3A%22Kitazawa%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Akimasa%22%2C%22lastName%22%3A%22Sato%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Natsuki%22%2C%22lastName%22%3A%22Watanabe%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yasuteru%22%2C%22lastName%22%3A%22Shigeta%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masayuki%22%2C%22lastName%22%3A%22Umemura%22%7D%5D%2C%22abstractNote%22%3A%22High%20enantiomeric%20excesses%20%28ee%27s%29%20of%20l-amino%20acids%2C%20including%20non-proteinogenic%20amino%20acid%20isovaline%20%28Iva%29%2C%20were%20discovered%20in%20the%20Murchison%20meteorite%2C%20but%20the%20detailed%20molecular%20mechanism%20responsible%20for%20the%20observed%20ee%20of%20amino%20acids%20remains%20elusive%20and%20inconsistent%2C%20because%20Iva%20has%20an%20inverted%20circular%20dichroism%20%28CD%29%20spectrum%20with%20respect%20to%20alpha-H%20amino%20acids%2C%20e.g.%2C%20alanine.%20To%20address%20this%20issue%2C%20we%20resort%20to%20accurate%20ab%20initio%20calculations%20for%20amino%20acids%20and%20their%20precursors%20in%20the%20Strecker%20synthesis.%20We%20evaluated%20their%20photolysis-induced%20ee%20in%20the%20range%205-11%20eV%20including%20the%20Lyman%20alpha%20emission%20line%20%28Lyalpha%29%2C%20the%20typical%20intensive%2010.2%20eV%20radiation%20ascribed%20to%20the%20early%20phase%20of%20galactic%20evolution.%20We%20show%20that%20only%20the%20aminonitrile%20precursors%20are%20characterized%20by%20positive%20ee%20in%20the%20Lyalpha%20region%2C%20explaining%20why%20right-handed%20circularly%20polarized%20Lyalpha%20%28R-CP-Lyalpha%29%20induces%20homologous%20l-amino%20acids.%20This%20study%20shows%20that%20the%20homochirality%20of%20amino%20acids%20is%20produced%20at%20the%20aminonitrile%20precursors%20stage.%22%2C%22date%22%3A%222023%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Facs.jpclett.2c03862%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Facs.jpclett.2c03862%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221948-7185%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A03%3A20Z%22%7D%7D%2C%7B%22key%22%3A%22CNFNPCS9%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Watanabe%20et%20al.%22%2C%22parsedDate%22%3A%222023%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EN.%20Watanabe%2C%20Y.%20Hori%2C%20M.%20Shoji%2C%20M.%20Boero%2C%20Y.%20Shigeta%2C%20Organocatalytic-racemization%20reaction%20elucidation%20of%20aspartic%20acid%20by%20density%20functional%20theory.%2C%20Chirality%20%282023%29%201%26%23x2013%3B7.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Fchir.23573%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Fchir.23573%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Organocatalytic-racemization%20reaction%20elucidation%20of%20aspartic%20acid%20by%20density%20functional%20theory.%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Natsuki%22%2C%22lastName%22%3A%22Watanabe%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yuta%22%2C%22lastName%22%3A%22Hori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mitsuo%22%2C%22lastName%22%3A%22Shoji%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yasuteru%22%2C%22lastName%22%3A%22Shigeta%22%7D%5D%2C%22abstractNote%22%3A%22Aldehydes%20and%20carboxylic%20acids%20are%20widely%20used%20as%20catalysts%20for%20efficient%20racemization%20process%20of%20amino%20acids.%20However%2C%20the%20detailed%20reaction%20mechanism%20remains%20unclear.%20This%20work%20aims%20to%20clarify%20the%20racemization%20mechanism%20of%20aspartic%20acid%20%28Asp%29%20catalyzed%20by%20salicylaldehyde%20and%20acetic%20acid%20by%20using%20computational%20approaches.%20Density%20functional%20theory%20was%20used%20to%20obtain%20the%20structures%20and%20relative%20energies%20of%2010%20intermediates%20and%20five%20transition%20states%2C%20thus%20characterizing%20the%20main%20stages%20of%20the%20reaction.%20The%20calculated%20energy%20diagram%20shows%20that%20the%20dehydration%20step%20has%20the%20highest%20energy%20barrier%2C%20followed%20by%20the%20reaction%20step%20to%20change%20the%20chirality%20of%20Asp%2C%20which%20is%20a%20crucial%20process%20for%20racemization.%20In%20the%20dehydration%20reaction%2C%20water%20molecules%20can%20induce%20a%20remarkable%20decrease%20in%20the%20required%20energy.%22%2C%22date%22%3A%222023%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1002%5C%2Fchir.23573%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1002%5C%2Fchir.23573%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221520-636X%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A04%3A21Z%22%7D%7D%2C%7B%22key%22%3A%22EB2FVKNU%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Watanabe%20et%20al.%22%2C%22parsedDate%22%3A%222023%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EN.%20Watanabe%2C%20M.%20Shoji%2C%20K.%20Miyagawa%2C%20Y.%20Hori%2C%20M.%20Boero%2C%20M.%20Umemura%2C%20Y.%20Shigeta%2C%20Enantioselective%20amino%20acid%20interactions%20in%20solution%2C%20Physical%20Chemistry%20Chemical%20Physics%2025%20%282023%29%2015023%26%23x2013%3B15029.%20%3Ca%20class%3D%27zp-ItemURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd3cp00278k%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd3cp00278k%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Enantioselective%20amino%20acid%20interactions%20in%20solution%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Natsuki%22%2C%22lastName%22%3A%22Watanabe%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mitsuo%22%2C%22lastName%22%3A%22Shoji%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Koichi%22%2C%22lastName%22%3A%22Miyagawa%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yuta%22%2C%22lastName%22%3A%22Hori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masayuki%22%2C%22lastName%22%3A%22Umemura%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yasuteru%22%2C%22lastName%22%3A%22Shigeta%22%7D%5D%2C%22abstractNote%22%3A%22Enantiomeric%20excesses%20%28ee%29%20of%20l-amino%20acids%20in%20meteorites%20are%20higher%20than%2010%25%2C%20especially%20for%20isovaline%20%28Iva%29.%20This%20suggests%20the%20existence%20of%20some%20kind%20of%20triggering%20mechanism%20responsible%20for%20the%20amplification%20of%20the%20ee%20from%20an%20initial%20small%20value.%20Here%2C%20we%20investigate%20the%20dimeric%20molecular%20interactions%20of%20alanine%20%28Ala%29%20and%20Iva%20in%20solution%20as%20an%20initial%20nucleation%20step%20of%20crystals%20at%20an%20accurate%20first-principles%20level.%20We%20find%20that%20the%20dimeric%20interaction%20of%20Iva%20is%20more%20chirality-dependent%20than%20that%20of%20Ala%2C%20thus%20providing%20a%20clear%20molecular-level%20insight%20into%20the%20enantioselectivity%20of%20amino%20acids%20in%20solution.%22%2C%22date%22%3A%222023%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fd3cp00278k%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd3cp00278k%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221463-9076%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A04%3A24Z%22%7D%7D%5D%7D
[1]
C.O. Diarra, M. Boero, E. Steveler, T. Heiser, E. Martin, Exciton diffusion in poly(3-hexylthiophene) by first-principles molecular dynamics., Physical Chemistry Chemical Physics 25 (2023) 15539–15546.
https://doi.org/10.1039/d3cp00533j.
[1]
M. Guerboub, S.D. Wansi Wendji, C. Massobrio, A. Bouzid, M. Boero, G. Ori, E. Martin, Impact of the local atomic structure on the thermal conductivity of amorphous Ge2Sb2Te5., Journal of Chemical Physics 158 (2023) 084504.
https://doi.org/10.1063/5.0139590.
[1]
P. Lawes, M. Boero, R. Barhoumi, S. Klyatskaya, M. Ruben, J.-P. Bucher, Hierarchical Self-Assembly and Conformation of Tb Double-Decker Molecular Magnets: Experiment and Molecular Dynamics, Nanomaterials 13 (2023) 2232.
https://doi.org/10.3390/nano13152232.
[1]
T.-L. Pham, W.I. Choi, A. Shafique, H.J. Kim, M. Shim, K. Min, W.-J. Son, I. Jang, D.S. Kim, M. Boero, C. Massobrio, G. Ori, H.S. Lee, Y.-H. Shin, Structural-Stability Study of Antiperovskite Na3OCl for Na-Rich Solid Electrolyte, Physical Review Applied 19 (2023) 034004.
https://doi.org/10.1103/PhysRevApplied.19.034004.
[1]
T.-L. Pham, M. Guerboub, A. Bouzid, M. Boero, C. Massobrio, Y.-H. Shin, G. Ori, Unveiling the structure and ion dynamics of amorphous Na
3-xOH
xCl antiperovskite electrolytes by first-principles molecular dynamics, Journal of Materials Chemistry A 11 (2023) 22922–22940.
https://doi.org/10.1039/d3ta01373a.
[1]
A. Sato, M. Shoji, N. Watanabe, M. Boero, Y. Shigeta, M. Umemura, Origin of Homochirality in Amino Acids Induced by Lyman-alpha Irradiation in the Early Stage of the Milky Way., Astrobiology 23 (2023) 1019–1026.
https://doi.org/10.1089/ast.2022.0140.
[1]
M. Shoji, Y. Kitazawa, A. Sato, N. Watanabe, M. Boero, Y. Shigeta, M. Umemura, Enantiomeric Excesses of Aminonitrile Precursors Determine the Homochirality of Amino Acids., Journal of Physical Chemistry Letters 14 (2023) 3243–3248.
https://doi.org/10.1021/acs.jpclett.2c03862.
[1]
N. Watanabe, Y. Hori, M. Shoji, M. Boero, Y. Shigeta, Organocatalytic-racemization reaction elucidation of aspartic acid by density functional theory., Chirality (2023) 1–7.
https://doi.org/10.1002/chir.23573.
[1]
N. Watanabe, M. Shoji, K. Miyagawa, Y. Hori, M. Boero, M. Umemura, Y. Shigeta, Enantioselective amino acid interactions in solution, Physical Chemistry Chemical Physics 25 (2023) 15023–15029.
https://doi.org/10.1039/d3cp00278k.
1839302
NZSFH59F
2022
1
surface-science-reports
50
creator
asc
year
7054
https://www.ipcms.unistra.fr/plugins/zotpress/
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3Afalse%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%228EWSYIKE%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Boero%20et%20al.%22%2C%22parsedDate%22%3A%222022%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Boero%2C%20K.M.%20Bui%2C%20K.%20Shiraishi%2C%20K.%20Ishisone%2C%20Y.%20Kangawa%2C%20A.%20Oshiyama%2C%20An%20atomistic%20insight%20into%20reactions%20and%20free-energy%20profiles%20of%20NH3%20and%20Ga%20on%20GaN%20surfaces%20during%20the%20epitaxial%20growth%2C%20Applied%20Surface%20Science%20599%20%282022%29%20153935.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.apsusc.2022.153935%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.apsusc.2022.153935%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22An%20atomistic%20insight%20into%20reactions%20and%20free-energy%20profiles%20of%20NH3%20and%20Ga%20on%20GaN%20surfaces%20during%20the%20epitaxial%20growth%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kieu%20My%22%2C%22lastName%22%3A%22Bui%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenji%22%2C%22lastName%22%3A%22Shiraishi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kana%22%2C%22lastName%22%3A%22Ishisone%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yoshihiro%22%2C%22lastName%22%3A%22Kangawa%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Atsushi%22%2C%22lastName%22%3A%22Oshiyama%22%7D%5D%2C%22abstractNote%22%3A%22Precursor%20molecules%20%28NH3%20and%20Ga%20compounds%29%20along%20with%20carrier%20gas%20%28H-2%20or%20N-2%29%20used%20to%20grow%20GaN%20structures%20bring%20a%20large%20amount%20of%20hydrogen%20atoms%20which%20affect%20the%20growing%20mechanism%20of%20GaN.%20This%20has%20a%20non-negligible%20effect%20of%20the%20chemistry%20and%20diffusivity%20of%20precursors%20and%20dissociation%20products.%20To%20encompass%20the%20experimentally%20difficulty%20in%20of%20unraveling%20such%20a%20complicated%20reaction%20mechanism%2C%20we%20resort%20to%20first%20principles%20molecular%20dynamics%20modeling%2C%20providing%20an%20atomistic%20insight%20into%20two%20major%20issues.%20The%20first%20one%20is%20the%20evolution%20of%20H%20atoms%20after%20the%20adsorption%20and%20dissociation%20of%20NH3%20on%20the%20growing%20GaN%20surface.%20The%20second%20issue%20is%20to%20shed%20light%20on%20the%20role%20of%20passivating%20hydrogen%20at%20growth%20conditions%20for%20a%20typical%20GaN%20Ga-rich%20%280001%29%20surface.%20In%20the%20first%20case%2C%20reaction%20pathways%20alternative%20to%20the%20product%20of%20molecular%20hydrogen%20%28H-2%29%20can%20be%20realized%2C%20depending%20on%20the%20initial%20conditions%20and%20morphology%20of%20the%20surface%2C%20resulting%20in%20an%20adsorption%20of%20H%20atoms%2C%20thus%20contributing%20to%20its%20hydrogenation.%20In%20the%20second%20one%2C%20instead%2C%20we%20show%20how%20the%20presence%20of%20passivating%20H%20atoms%20at%20the%20surface%2C%20corresponding%20to%20a%20relatively%20high%20degree%20of%20hydrogenation%2C%20contribute%20to%20limit%20the%20diffusivity%20of%20Ga%20adatoms%20at%20the%20typical%20growth%20temperatures.%22%2C%22date%22%3A%222022%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.apsusc.2022.153935%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.apsusc.2022.153935%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220169-4332%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A05%3A53Z%22%7D%7D%2C%7B%22key%22%3A%22WK85MYU6%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Boero%20et%20al.%22%2C%22parsedDate%22%3A%222022%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Boero%2C%20F.%20Imoto%2C%20A.%20Oshiyama%2C%20Atomistic%20insight%20into%20the%20initial%20stage%20of%20graphene%20formation%20on%20SiC%280001%29%20surfaces%2C%20Physical%20Review%20Materials%206%20%282022%29%20093403.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevMaterials.6.093403%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevMaterials.6.093403%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Atomistic%20insight%20into%20the%20initial%20stage%20of%20graphene%20formation%20on%20SiC%280001%29%20surfaces%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fumihiro%22%2C%22lastName%22%3A%22Imoto%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Atsushi%22%2C%22lastName%22%3A%22Oshiyama%22%7D%5D%2C%22abstractNote%22%3A%22We%20present%20an%20atomistic%20insight%20into%20the%20processes%20leading%20to%20the%20formation%20of%20graphene%20on%20SiC%280001%29%20surfaces%20by%20resorting%20to%20first-principles%20molecular%20dynamics%20empowered%20by%20free-energy%20sampling%20methods.%20Based%20on%20the%20experimental%20surface%2C%20consisting%20of%20terraces%20bordered%20by%20a%20sequence%20of%20steps%2C%20we%20find%20that%20Si%20atoms%20are%20dislodged%20from%20step%20edges%20and%20migrate%20toward%20more%20stable%20sites%20on%20the%20terrace%2C%20leaving%20behind%20C%20atoms%20carrying%20unsaturated%20chemical%20bonds.%20Our%20investigations%20reveal%20that%20subsequent%20Si%20atoms%20removal%20acts%20as%20a%20trigger%20to%20the%20formation%20of%20stable%20C-C%20bonds%20among%20these%20unsaturated%20C%20sites.%20This%20process%20eventually%20leads%20to%20the%20formation%20of%20C%20clusters%20which%20merge%20into%20larger%20structures%20with%20the%20typical%20pattern%20of%20graphene%20flakes.%20Specifically%2C%20a%20C-6%20ring%20formed%20during%20our%20simulations%2C%20assumes%20the%20typical%20hexagonal%20structure%20of%20graphene%2C%20becoming%20a%20precursor%20of%20larger%20graphene%20nanostructures.%20The%20characterization%20of%20the%20mechanisms%20and%20related%20free-energy%20landscapes%20provide%20an%20insight%20into%20the%20fundamental%20processes%20responsible%20for%20the%20realization%20of%20ordered%20C-based%20building%20blocks%20of%20graphene%20on%20the%20SiC%280001%29%20surface.%22%2C%22date%22%3A%222022%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevMaterials.6.093403%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevMaterials.6.093403%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222475-9953%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A05%3A57Z%22%7D%7D%2C%7B%22key%22%3A%22XBZW9XPW%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Burel%20et%20al.%22%2C%22parsedDate%22%3A%222022%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EC.%20Burel%2C%20O.%20Ibrahim%2C%20E.%20Marino%2C%20H.%20Bharti%2C%20C.B.%20Murray%2C%20B.%20Donnio%2C%20Z.%20Fakhraai%2C%20R.%20Dreyfus%2C%20Tunable%20Plasmonic%20Microcapsules%20with%20Embedded%20Noble%20Metal%20Nanoparticles%20for%20Optical%20Microsensing%2C%20ACS%20Applied%20Nano%20Materials%205%20%282022%29%202828%26%23x2013%3B2838.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facsanm.1c04542%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facsanm.1c04542%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Tunable%20Plasmonic%20Microcapsules%20with%20Embedded%20Noble%20Metal%20Nanoparticles%20for%20Optical%20Microsensing%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Celine%22%2C%22lastName%22%3A%22Burel%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Omar%22%2C%22lastName%22%3A%22Ibrahim%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Emanuele%22%2C%22lastName%22%3A%22Marino%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Harshit%22%2C%22lastName%22%3A%22Bharti%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Christopher%20B.%22%2C%22lastName%22%3A%22Murray%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Bertrand%22%2C%22lastName%22%3A%22Donnio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Zahra%22%2C%22lastName%22%3A%22Fakhraai%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Remi%22%2C%22lastName%22%3A%22Dreyfus%22%7D%5D%2C%22abstractNote%22%3A%22We%20report%20a%20comprehensive%20investigation%20of%20the%20synthetic%20conditions%20leading%20to%20the%20formation%20of%20tunable%20plasmonic%20microcapsules%20%28MCs%29%20made%20of%20a%20hydrophobic%20liquid%20core%20encapsulated%20into%20a%20hard%20silica%20shell%20embedding%20plasmonic%20metallic%20nanoparticles%20%28NPs%29.%20The%20distinctive%20and%20remarkable%20features%20of%20the%20prepared%20MCs%20are%20the%20inert%20nanometer-thin%20silica%20shell%20and%20the%20small%20plasmonic%20NPs%20embedded%20in%20it%2C%20which%20confer%20interesting%20optical%20absorbance%20properties.%20We%20tie%20the%20mechanical%20robustness%20of%20the%20MCs%20to%20the%20thickness%20of%20their%20silica%20shell.%20We%20show%20that%20several%20oils%20can%20be%20used%20for%20the%20synthesis%20of%20MCs%20and%20we%20evidence%20how%20the%20relative%20solubility%20of%20the%20silica%20precursor%20and%20the%20polarity%20of%20the%20oil%20phase%20influence%20the%20final%20MC%20characteristics.%20We%20also%20evidence%20the%20synthesis%20of%20%5Cu201cmonoflavor%5Cu201d%20or%20%5Cu201cmultiflavor%5Cu201d%20MCs%20with%2C%20respectively%2C%20a%20single%20type%20of%20NPs%20or%20a%20mixture%20of%20metallic%20NPs%2C%20respectively%2C%20embedded%20in%20the%20silica%20shell.%20Using%20experiments%20and%20simulations%2C%20we%20demonstrate%20that%20the%20optical%20response%20of%20the%20MCs%20can%20be%20finely%20tuned%20by%20choosing%20the%20right%20ratio%20between%20Ag%20and%20Au%20NPs%20initially%20suspended%20in%20the%20solution.%20Our%20heterogeneous%20hybrid%20MCs%20exhibit%20optical%20properties%20directly%20resulting%20from%20the%20choice%20of%20NP%20composition%20and%20shell%20thickness%2C%20making%20them%20of%20great%20interest%20not%20only%20for%20mechanical%20and%20chemical%20microsensing%20but%20also%20for%20applications%20in%20photothermal%20therapy%2C%20surface-enhanced%20Raman%20spectroscopy%20studies%2C%20microreactor%20vesicles%20for%20interfacial%20electrocatalysis%2C%20antimicrobial%20activity%2C%20and%20drug%20delivery.%20Our%20simple%20and%20versatile%20emulsion%20template%20method%20holds%20great%20promise%20for%20the%20tailored%20design%20of%20a%20generation%20of%20multifunctional%20MCs%20consisting%20of%20modular%20nanoscale%20building%20blocks.%22%2C%22date%22%3A%222022%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Facsanm.1c04542%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Facsanm.1c04542%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22IEGKATUQ%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A06%3A15Z%22%7D%7D%2C%7B%22key%22%3A%22NFVBLTLC%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Gentili%20and%20Ori%22%2C%22parsedDate%22%3A%222022%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ED.%20Gentili%2C%20G.%20Ori%2C%20Reversible%20assembly%20of%20nanoparticles%3A%20theory%2C%20strategies%20and%20computational%20simulations.%2C%20Nanoscale%2014%20%282022%29%2014385%26%23x2013%3B14432.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd2nr02640f%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd2nr02640f%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Reversible%20assembly%20of%20nanoparticles%3A%20theory%2C%20strategies%20and%20computational%20simulations.%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Denis%22%2C%22lastName%22%3A%22Gentili%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%5D%2C%22abstractNote%22%3A%22The%20significant%20advances%20in%20synthesis%20and%20functionalization%20have%20enabled%20the%20preparation%20of%20high-quality%20nanoparticles%20that%20have%20found%20a%20plethora%20of%20successful%20applications.%20The%20unique%20physicochemical%20properties%20of%20nanoparticles%20can%20be%20manipulated%20through%20the%20control%20of%20size%2C%20shape%2C%20composition%2C%20and%20surface%20chemistry%2C%20but%20their%20technological%20application%20possibilities%20can%20be%20further%20expanded%20by%20exploiting%20the%20properties%20that%20emerge%20from%20their%20assembly.%20The%20ability%20to%20control%20the%20assembly%20of%20nanoparticles%20not%20only%20is%20required%20for%20many%20real%20technological%20applications%2C%20but%20allows%20the%20combination%20of%20the%20intrinsic%20properties%20of%20nanoparticles%20and%20opens%20the%20way%20to%20the%20exploitation%20of%20their%20complex%20interplay%2C%20giving%20access%20to%20collective%20properties.%20Significant%20advances%20and%20knowledge%20gained%20over%20the%20past%20few%20decades%20on%20nanoparticle%20assembly%20have%20made%20it%20possible%20to%20implement%20a%20growing%20number%20of%20strategies%20for%20reversible%20assembly%20of%20nanoparticles.%20In%20addition%20to%20being%20of%20interest%20for%20basic%20studies%2C%20such%20advances%20further%20broaden%20the%20range%20of%20applications%20and%20the%20possibility%20of%20developing%20innovative%20devices%20using%20nanoparticles.%20This%20review%20focuses%20on%20the%20reversible%20assembly%20of%20nanoparticles%20and%20includes%20the%20theoretical%20aspects%20related%20to%20the%20concept%20of%20reversibility%2C%20an%20up-to-date%20assessment%20of%20the%20experimental%20approaches%20applied%20to%20this%20field%20and%20the%20advanced%20computational%20schemes%20that%20offer%20key%20insights%20into%20the%20assembly%20mechanisms.%20We%20aim%20to%20provide%20readers%20with%20a%20comprehensive%20guide%20to%20address%20the%20challenges%20in%20assembling%20reversible%20nanoparticles%20and%20promote%20their%20applications.%22%2C%22date%22%3A%222022%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fd2nr02640f%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fd2nr02640f%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222040-3372%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A08%3A01Z%22%7D%7D%2C%7B%22key%22%3A%22HE8NYSA3%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Ishisone%20et%20al.%22%2C%22parsedDate%22%3A%222022%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Ishisone%2C%20G.%20Ori%2C%20M.%20Boero%2C%20Structural%2C%20dynamical%2C%20and%20electronic%20properties%20of%20the%20ionic%20liquid%201-ethyl-3-methylimidazolium%20bis%28trifluoromethylsulfonyl%29imide.%2C%20Physical%20Chemistry%20Chemical%20Physics%26%23x202F%3B%3A%20PCCP%2024%20%282022%29%209597%26%23x2013%3B9607.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd2cp00741j%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd2cp00741j%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Structural%2C%20dynamical%2C%20and%20electronic%20properties%20of%20the%20ionic%20liquid%201-ethyl-3-methylimidazolium%20bis%28trifluoromethylsulfonyl%29imide.%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kana%22%2C%22lastName%22%3A%22Ishisone%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22We%20provide%20a%20microscopic%20insight%2C%20both%20structural%20and%20electronic%2C%20into%20the%20multifold%20interactions%20occurring%20in%20the%20ionic%20liquid%201-ethyl-3-methylimidazolium%20bis%28trifluoromethylsulfonyl%29imide%20%5BEMIM%5D%5BTFSI%5D%20currently%20targeted%20for%20applications%20in%20next-generation%20low-power%20electronics%20and%20optoelectronic%20devices.%20To%20date%2C%20practical%20applications%20have%20remained%20hampered%20by%20the%20lack%20of%20fundamental%20understanding%20of%20the%20interactions%20occurring%20both%20inside%20the%20IL%20and%20at%20the%20interface%20with%20the%20substrate.%20Our%20first%20principles%20dynamical%20simulations%20provide%20accurate%20insights%20into%20the%20nature%20of%20bonding%20and%20non-bonding%20interactions%2C%20dynamical%20conformational%20changes%20and%20induced%20dipole%20moments%2C%20along%20with%20their%20statistical%20distributions%2C%20of%20this%20ionic%20liquid%2C%20that%20have%20so%20far%20not%20been%20completely%20unraveled.%20The%20mobilities%20of%20the%20two%20ionic%20species%20are%20obtained%20by%20long-lasting%20dynamical%20simulations%20at%20finite%20temperature%2C%20allowing%20simultaneous%20monitoring%20and%20quantification%20of%20the%20isomerization%20occurring%20in%20the%20IL.%20Moreover%2C%20a%20thorough%20analysis%20of%20the%20electronic%20structure%20and%20partial%20charge%20distributions%20characterizing%20the%20two%20components%2C%20the%20cation%20and%20anion%2C%20allow%20rationalization%20of%20the%20nature%20of%20the%20electrostatic%20interactions%2C%20hydrogen%20bonding%20properties%20of%20the%20two%20ionic%20counterparts%2C%20and%20the%20infra-red%20and%20dielectric%20response%20of%20the%20system%2C%20especially%20in%20the%20low%20frequency%20range%2C%20for%20the%20full%20characterization%20of%20the%20IL.%22%2C%22date%22%3A%222022%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fd2cp00741j%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fd2cp00741j%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A08%3A49Z%22%7D%7D%2C%7B%22key%22%3A%22VCEXHJH9%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Lambrecht%20et%20al.%22%2C%22parsedDate%22%3A%222022%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EA.%20Lambrecht%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20G.%20Ori%2C%20E.%20Martin%2C%20Atomic%20structure%20of%20amorphous%20SiN%3A%20Combining%20Car-Parrinello%20and%20Born-Oppenheimer%20first-principles%20molecular%20dynamics%2C%20Computational%20Materials%20Science%20211%20%282022%29%20111555.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.commatsci.2022.111555%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.commatsci.2022.111555%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Atomic%20structure%20of%20amorphous%20SiN%3A%20Combining%20Car-Parrinello%20and%20Born-Oppenheimer%20first-principles%20molecular%20dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Achille%22%2C%22lastName%22%3A%22Lambrecht%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%5D%2C%22abstractNote%22%3A%22First-principles%20molecular%20dynamics%20is%20employed%20to%20describe%20the%20atomic%20structure%20of%20amorphous%20SiN%2C%20a%20non-stoichiometric%20compound%20belonging%20to%20the%20SixNy%20family.%20To%20produce%20the%20amorphous%20state%20via%20the%20cooling%20of%20the%20liquid%2C%20both%20the%20Car-Parrinello%20and%20the%20Born-Oppenheimer%20approaches%20are%20exploited%20to%20obtain%20a%20system%20featuring%20sizeable%20atomic%20mobility.%20At%20high%20temperatures%2C%20due%20to%20the%20peculiar%20electronic%20structure%20of%20SiN%2C%20exhibiting%20gap%20closing%20effects%2C%20the%20Car-Parrinello%20methodology%20could%20not%20be%20followed%20since%20non-adiabatic%20effects%20involving%20the%20ionic%20and%20electronic%20degrees%20of%20freedom%20do%20occur.%20This%20shortcoming%20was%20surmounted%20by%20resorting%20to%20the%20Born-Oppenheimer%20approach%20allowing%20to%20achieve%20significant%20ionic%20diffusion%20at%20T%20%3D%202500%20K.%20From%20this%20highly%20diffusive%20sample%2C%20an%20amorphous%20state%20at%20room%20temperature%20was%20obtained%20with%20a%20quenching%20rate%20of%2010%20K%5C%2Fps.%20Four%20different%20models%20were%20created%2C%20differing%20by%20their%20sizes%20and%20the%20thermal%20cycles.%20We%20found%20that%20the%20subnetwork%20of%20atoms%20N%20has%20the%20same%20environment%20than%20in%20the%20stoichiometric%20material%20Si3N4%20since%20N%20is%20mostly%20threefold%20coordinated%20with%20Si.%20Si%20atoms%20can%20also%20be%20found%20coordinated%20to%20four%20N%20atoms%20as%20in%20Si3N4%2C%20but%20a%20substantial%20fraction%20of%20them%20forms%20homopolar%20bonds%20with%20one%2C%20two%2C%20three%20and%20even%20four%20Si.%20Our%20results%20are%20not%20too%20dissimilar%20from%20former%20models%20available%20in%20the%20literature%20but%20they%20feature%20a%20higher%20statistical%20accuracy%20and%20refer%20more%20precisely%20to%20room%20temperature%20as%20the%20reference%20thermodynamical%20condition%20for%20the%20analysis%20of%20the%20structure%20in%20the%20amorphous%20state.%22%2C%22date%22%3A%222022%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.commatsci.2022.111555%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.commatsci.2022.111555%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220927-0256%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A09%3A58Z%22%7D%7D%2C%7B%22key%22%3A%22YKF4L3J6%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Martin%20et%20al.%22%2C%22parsedDate%22%3A%222022%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EE.%20Martin%2C%20G.%20Ori%2C%20T.-Q.%20Duong%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20Thermal%20conductivity%20of%20amorphous%20SiO2%20by%20first-principles%20molecular%20dynamics%2C%20Journal%20of%20Non-Crystalline%20Solids%20581%20%282022%29%20121434.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2022.121434%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2022.121434%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Thermal%20conductivity%20of%20amorphous%20SiO2%20by%20first-principles%20molecular%20dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Thuy-Quynh%22%2C%22lastName%22%3A%22Duong%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22The%20approach-to-equilibrium%20molecular%20dynamics%20%28AEMD%29%20methodology%20implemented%20within%20a%20first-principles%20molecular%20dynamics%20%28FPMD%29%20scheme%20is%20applied%20to%20amorphous%20SiO2.%20In%20this%20disordered%20material%2C%20measurements%20of%20the%20thermal%20conductivity%20indicate%20no%20reduction%20down%20to%2010%20nm.%20In%20view%20of%20these%20premises%2C%20we%20calculate%20the%20thermal%20conductivity%20of%20amorphous%20SiO2%20in%20the%20size%20range%20comprised%20between%202%20and%208%20nm%20via%20the%20AEMD%5C%2FFPMD%20approach.%20The%20thermal%20conductivity%20agrees%20with%20experiments%20for%20the%20largest%20sizes%20we%20considered%2C%20while%20it%20is%20strongly%20reduced%20for%20values%20not%20accessible%20to%20experimental%20resolution%20%28up%20to%2050%25%20for%202%20nm%29.%20This%20behavior%20is%20close%20to%20that%20found%20in%20amorphous%20chalcogenides%20GeTe4%20and%20Ge2Sb2Te5%20within%20the%20same%20AEMD%5C%2FFPMD%20approach.%20Taken%20together%2C%20these%20results%20show%20that%20the%20observed%20decrease%20of%20the%20thermal%20conductivity%20is%20a%20general%20feature%20of%20disordered%20networks%20and%20in%20any%20case%20cannot%20be%20taken%20as%20peculiar%20to%20a%20specific%20class%20of%20systems.%22%2C%22date%22%3A%222022%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.jnoncrysol.2022.121434%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2022.121434%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220022-3093%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A10%3A49Z%22%7D%7D%2C%7B%22key%22%3A%223FJRYELY%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Martin%20et%20al.%22%2C%22parsedDate%22%3A%222022%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EE.%20Martin%2C%20I.B.%20Amiehe%20Essomba%2C%20K.%20Ishisone%2C%20M.%20Boero%2C%20G.%20Ori%2C%20C.%20Massobrio%2C%20Impact%20of%20Dispersion%20Force%20Schemes%20on%20Liquid%20Systems%3A%20Comparing%20Efficiency%20and%20Drawbacks%20for%20Well-Targeted%20Test%20Cases%2C%20Molecules%2027%20%282022%29.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3390%5C%2Fmolecules27249034%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3390%5C%2Fmolecules27249034%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Impact%20of%20Dispersion%20Force%20Schemes%20on%20Liquid%20Systems%3A%20Comparing%20Efficiency%20and%20Drawbacks%20for%20Well-Targeted%20Test%20Cases%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Irene%20Berenger%22%2C%22lastName%22%3A%22Amiehe%20Essomba%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kana%22%2C%22lastName%22%3A%22Ishisone%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22First-principles%20molecular%20dynamics%20%28FPMD%29%20calculations%20were%20performed%20on%20liquid%20GeSe4%20with%20the%20aim%20of%20inferring%20the%20impact%20of%20dispersion%20%28van%20der%20Waals%2C%20vdW%29%20forces%20on%20the%20structural%20properties.%20Different%20expressions%20for%20the%20dispersion%20forces%20were%20employed%2C%20allowing%20us%20to%20draw%20conclusions%20on%20their%20performances%20in%20a%20comparative%20fashion.%20These%20results%20supersede%20previous%20FPMD%20calculations%20obtained%20in%20smaller%20systems%20and%20shorter%20time%20trajectories%20by%20providing%20data%20of%20unprecedented%20accuracy.%20We%20obtained%20a%20substantial%20agreement%20with%20experiments%20for%20the%20structure%20factor%20regardless%20of%20the%20vdW%20scheme%20employed.%20This%20objective%20was%20achieved%20by%20using%20%28in%20addition%20to%20FPMD%20with%20no%20dispersion%20forces%29%20a%20selection%20of%20vdW%20schemes%20available%20within%20density%20functional%20theory.%20The%20first%20two%20are%20due%20to%20Grimme%2C%20D2%20and%20D3%2C%20and%20the%20third%20one%20is%20devised%20within%20the%20so-called%20maximally%20localized%20Wannier%20functions%20approach%20%28MLWF%29.%20D3%20results%20feature%20a%20sizeable%20disagreement%20in%20real%20space%20with%20D2%20and%20MLWF%20in%20terms%20of%20the%20partial%20and%20total%20pair%20correlation%20functions%20as%20well%20as%20the%20coordination%20numbers.%20More%20strikingly%2C%20total%20and%20partial%20structure%20factors%20calculated%20with%20D3%20exhibit%20an%20unexpected%20sharp%20increase%20at%20low%20k.%20This%20peculiarity%20goes%20along%20with%20large%20void%20regions%20within%20the%20network%2C%20standing%20for%20a%20phase%20separation%20of%20indecipherable%20physical%20meaning.%20In%20view%20of%20these%20findings%2C%20further%20evidence%20of%20unconventional%20structural%20properties%20found%20by%20employing%20D3%20is%20presented%20by%20relying%20on%20results%20obtained%20for%20a%20complex%20ionic%20liquid%20supported%20on%20a%20solid%20surface.%20The%20novelty%20of%20our%20study%20is%20multifold%3A%20new%2C%20reliable%20FPMD%20data%20for%20a%20prototypical%20disordered%20network%20system%2C%20convincing%20agreement%20with%20experimental%20data%20and%20assessment%20of%20the%20impact%20of%20dispersion%20forces%2C%20with%20emphasis%20on%20the%20intriguing%20behavior%20of%20one%20specific%20recipe%20and%20the%20discovery%20of%20common%20structural%20features%20shared%20by%20drastically%20dissimilar%20physical%20systems%20when%20the%20D3%20vdW%20scheme%20is%20employed.%22%2C%22date%22%3A%222022%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.3390%5C%2Fmolecules27249034%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.3390%5C%2Fmolecules27249034%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A10%3A47Z%22%7D%7D%2C%7B%22key%22%3A%22U4HXPMZM%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Massobrio%22%2C%22parsedDate%22%3A%222022%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EC.%20Massobrio%2C%20The%20Structure%20of%20Amorphous%20Materials%20using%20Molecular%20Dynamics%2C%20IOP%20Publishing%2C%202022.%20%3Ca%20class%3D%27zp-ItemURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1088%5C%2F978-0-7503-2436-6%27%3Ehttps%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1088%5C%2F978-0-7503-2436-6%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22book%22%2C%22title%22%3A%22The%20Structure%20of%20Amorphous%20Materials%20using%20Molecular%20Dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22This%20reference%20text%20demonstrates%20how%20molecular%20dynamics%20can%20be%20used%20in%20practice%20to%20achieve%20a%20precise%20understanding%20of%20structural%20properties%20for%20systems%20devoid%20of%20any%20order%20beyond%20the%20first%20interatomic%20distances.%20The%20reader%20will%20learn%20the%20basic%20principles%20underlying%20molecular%20dynamics%20with%20a%20special%20emphasis%20on%20first-principles%20methodology.%20A%20roadmap%20of%20correct%20and%20efficient%20use%20is%20also%20provided%20using%20clear%20examples.%20The%20book%20concludes%20with%20a%20set%20of%20results%20that%20exemplify%20the%20level%20of%20accuracy%20and%20information%20inherent%20in%20%28first-principles%29%20molecular%20dynamics%20methodology%20when%20applied%20to%20amorphous%20and%20glassy%20materials.%20While%20the%20majority%20of%20systems%20studied%20are%20disordered%20chalcogenides%2C%20the%20ideas%2C%20concepts%20and%20methodologies%20involved%20are%20easily%20applicable%20to%20any%20system%2C%20providing%20a%20universal%20manual%20well-adapted%20to%20a%20wide%20range%20of%20practitioners%2C%20from%20graduate%20students%20to%20experienced%20researchers.Key%20features%5Cu2022%20Describes%20the%20structure%20of%20amorphous%20materials%20using%20molecular%20dynamics%20through%20research%20conducted%20by%20a%20single%20author%20over%20an%20extended%20period%20of%20time%5Cu2022%20Demonstrates%20how%20molecular%20dynamics%20can%20be%20used%20in%20practice%20to%20achieve%20a%20precise%20understanding%20of%20structural%20properties%20for%20systems%20devoid%20of%20any%20order%20beyond%20the%20first%20interatomic%20distances%5Cu2022%20Provides%20a%20roadmap%20of%20correct%20and%20efficient%20use%20using%20clear%20examples%5Cu2022%20Includes%20a%20set%20of%20results%20that%20exemplify%20the%20level%20of%20accuracy%20and%20information%20inherent%20in%20%28first-principles%29%20molecular%20dynamics%20methodology%20when%20applied%20to%20amorphous%20and%20glassy%20materials%5Cu2022%20Covers%20ideas%2C%20concepts%20and%20methodologies%20that%20are%20easily%20applicable%20to%20any%20system%5Cu2022%20Provides%20a%20universal%20manual%20well-adapted%20to%20a%20wide%20range%20of%20practitioners%2C%20from%20graduate%20students%20to%20experienced%20researchers%22%2C%22date%22%3A%222022%22%2C%22originalDate%22%3A%22%22%2C%22originalPublisher%22%3A%22%22%2C%22originalPlace%22%3A%22%22%2C%22format%22%3A%22%22%2C%22ISBN%22%3A%22978-0-7503-2436-6%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1088%5C%2F978-0-7503-2436-6%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A10%3A55Z%22%7D%7D%2C%7B%22key%22%3A%22LDTHFUGQ%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Omeis%20et%20al.%22%2C%22parsedDate%22%3A%222022%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EF.%20Omeis%2C%20Z.%20Boubegtiten-Fezoua%2C%20A.F.S.%20Seica%2C%20R.%20Bernard%2C%20M.H.%20Iqbal%2C%20N.%20Javahiraly%2C%20R.M.A.%20Vergauwe%2C%20H.%20Majjad%2C%20F.%20Boulmedais%2C%20D.%20Moss%2C%20P.%20Hellwig%2C%20Plasmonic%20Resonant%20Nanoantennas%20Induce%20Changes%20in%20the%20Shape%20and%20the%20Intensity%20of%20Infrared%20Spectra%20of%20Phospholipids%2C%20Molecules%2027%20%282022%29%2062.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3390%5C%2Fmolecules27010062%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3390%5C%2Fmolecules27010062%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Plasmonic%20Resonant%20Nanoantennas%20Induce%20Changes%20in%20the%20Shape%20and%20the%20Intensity%20of%20Infrared%20Spectra%20of%20Phospholipids%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fatima%22%2C%22lastName%22%3A%22Omeis%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Zahia%22%2C%22lastName%22%3A%22Boubegtiten-Fezoua%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ana%20Filipa%20Santos%22%2C%22lastName%22%3A%22Seica%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Romain%22%2C%22lastName%22%3A%22Bernard%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Muhammad%20Haseeb%22%2C%22lastName%22%3A%22Iqbal%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Nicolas%22%2C%22lastName%22%3A%22Javahiraly%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Robrecht%20M.%20A.%22%2C%22lastName%22%3A%22Vergauwe%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hicham%22%2C%22lastName%22%3A%22Majjad%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fouzia%22%2C%22lastName%22%3A%22Boulmedais%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22David%22%2C%22lastName%22%3A%22Moss%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Petra%22%2C%22lastName%22%3A%22Hellwig%22%7D%5D%2C%22abstractNote%22%3A%22Surface%20enhanced%20infrared%20absorption%20spectroscopic%20studies%20%28SEIRAS%29%20as%20a%20technique%20to%20study%20biological%20molecules%20in%20extremely%20low%20concentrations%20is%20greatly%20evolving.%20In%20order%20to%20use%20the%20technique%20for%20identification%20of%20the%20structure%20and%20interactions%20of%20such%20biological%20molecules%2C%20it%20is%20necessary%20to%20identify%20the%20effects%20of%20the%20plasmonic%20electric-field%20enhancement%20on%20the%20spectral%20signature.%20In%20this%20study%20the%20spectral%20properties%20of%201%2C2-Dipalmitoyl-sn-glycero-3%20phosphothioethanol%20%28DPPTE%29%20phospholipid%20immobilized%20on%20gold%20nanoantennas%2C%20specifically%20designed%20to%20enhance%20the%20vibrational%20fingerprints%20of%20lipid%20molecules%20were%20studied.%20An%20AFM%20study%20demonstrates%20an%20organization%20of%20the%20DPPTE%20phospholipid%20in%20bilayers%20on%20the%20nanoantenna%20structure.%20The%20spectral%20data%20were%20compared%20to%20SEIRAS%20active%20gold%20surfaces%20based%20on%20nanoparticles%2C%20plain%20gold%20and%20plain%20substrate%20%28Si%29%20for%20different%20temperatures.%20The%20shape%20of%20the%20infrared%20signals%2C%20the%20peak%20positions%20and%20their%20relative%20intensities%20were%20found%20to%20be%20sensitive%20to%20the%20type%20of%20surface%20and%20the%20presence%20of%20an%20enhancement.%20The%20strongest%20shifts%20in%20position%20and%20intensity%20were%20seen%20for%20the%20nanoantennas%2C%20and%20a%20smaller%20effect%20was%20seen%20for%20the%20DPPTE%20immobilized%20on%20gold%20nanoparticles.%20This%20information%20is%20crucial%20for%20interpretation%20of%20data%20obtained%20for%20biological%20molecules%20measured%20on%20such%20structures%2C%20for%20future%20application%20in%20nanodevices%20for%20biologically%20or%20medically%20relevant%20samples.%22%2C%22date%22%3A%222022%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.3390%5C%2Fmolecules27010062%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.3390%5C%2Fmolecules27010062%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221420-3049%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%225T5YGD4D%22%2C%22N8397DCZ%22%2C%22NZSFH59F%22%2C%22QK933HES%22%2C%22UVN4N32C%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A11%3A31Z%22%7D%7D%2C%7B%22key%22%3A%22LJEU23HD%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Payet%20et%20al.%22%2C%22parsedDate%22%3A%222022%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EF.%20Payet%2C%20C.%20Bouillet%2C%20F.%20Leroux%2C%20C.%20Leuvrey%2C%20P.%20Rabu%2C%20F.%20Schosseler%2C%20C.%20Taviot-Gu%26%23xE9%3Bho%2C%20G.%20Rogez%2C%20Fast%20and%20efficient%20shear-force%20assisted%20production%20of%20covalently%20functionalized%20oxide%20nanosheets%2C%20Journal%20of%20Colloid%20and%20Interface%20Science%20607%20%282022%29%20621%26%23x2013%3B632.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jcis.2021.08.213%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jcis.2021.08.213%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Fast%20and%20efficient%20shear-force%20assisted%20production%20of%20covalently%20functionalized%20oxide%20nanosheets%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fr%5Cu00e9d%5Cu00e9ric%22%2C%22lastName%22%3A%22Payet%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Corinne%22%2C%22lastName%22%3A%22Bouillet%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fabrice%22%2C%22lastName%22%3A%22Leroux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22C%5Cu00e9dric%22%2C%22lastName%22%3A%22Leuvrey%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Pierre%22%2C%22lastName%22%3A%22Rabu%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fran%5Cu00e7ois%22%2C%22lastName%22%3A%22Schosseler%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Christine%22%2C%22lastName%22%3A%22Taviot-Gu%5Cu00e9ho%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guillaume%22%2C%22lastName%22%3A%22Rogez%22%7D%5D%2C%22abstractNote%22%3A%22Hypothesis%5CnWhile%20controlled%20and%20efficient%20exfoliation%20of%20layered%20oxides%20often%20remains%20a%20time%20consuming%20challenge%2C%20the%20surface%20modification%20of%20inorganic%20nanosheets%20is%20of%20outmost%20importance%20for%20future%20applications.%20The%20functionalization%20of%20the%20bulk%20material%20prior%20to%20exfoliation%20should%20allow%20the%20application%20of%20tools%20developped%20for%20Van%20der%20Waals%20materials%20to%20directly%20produce%20functionalized%20oxide%20nanosheets.%5CnExperiments%5CnThe%20Aurivillius%20phase%20Bi2SrTa2O9%20is%20functionalized%20by%20a%20linear%20aliphatic%20phosphonic%20acid%20via%20microwave-assisted%20reactions.%20The%20structure%20of%20the%20hybrid%20material%20and%20the%20coordination%20of%20the%20phosphonate%20group%20is%20scrutinized%2C%20notably%20by%20Pair%20Distribution%20Function.%20This%20functionalized%20layered%20oxide%20is%20then%20exfoliated%20in%20one%20hour%20in%20organic%20solvent%2C%20using%20high%20shear%20force%20dispersion.%20The%20obtained%20nanosheets%20are%20characterized%20in%20suspension%20and%20as%20deposits%20to%20check%20their%20chemical%20integrity.%5CnFindings%5CnThe%20covalent%20functionalization%20decreases%20the%20electrostatic%20cohesion%20between%20the%20inorganic%20layers%20leading%20to%20an%20efficient%20exfoliation%20in%20short%20time%20under%20shearing.%20The%20functionalization%20of%20the%20bulk%20material%20is%20preserved%20on%20the%20nanosheets%20upon%20exfoliation%20and%20plays%20a%20major%20role%20to%20enable%20liquid-phase%20exfoliation%20and%20in%20the%20stability%20of%20the%20resulting%20suspensions.%20This%20strategy%20is%20very%20promising%20for%20the%20straighforward%20preparation%20of%20functionalized%20nanosheets%2C%20paving%20the%20way%20for%20versatile%20design%20of%20new%20%28multi%29functional%20hybrid%20nanosheets%20for%20various%20potential%20applications.%22%2C%22date%22%3A%222022%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.jcis.2021.08.213%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.sciencedirect.com%5C%2Fscience%5C%2Farticle%5C%2Fpii%5C%2FS0021979721014478%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220021-9797%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%226IWM732K%22%2C%22CF4ZI7HM%22%2C%22DEB5KWFS%22%2C%22M244N6AF%22%2C%22NZSFH59F%22%2C%22WJDNKBGA%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A11%3A56Z%22%7D%7D%2C%7B%22key%22%3A%22XBT537KI%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Peltier%20et%20al.%22%2C%22parsedDate%22%3A%222022%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EJ.-D.%20Peltier%2C%20B.%20Heinrich%2C%20B.%20Donnio%2C%20O.A.%20Ibraikulov%2C%20T.%20Heiser%2C%20N.%20Leclerc%2C%20J.%20Rault-Berthelot%2C%20C.%20Poriel%2C%20Dispiroacridine-indacenobisthiophene%20positional%20isomers%3A%20impact%20of%20the%20bridge%20on%20the%20physicochemical%20properties%2C%20Materials%20Chemistry%20Frontiers%206%20%282022%29%20225%26%23x2013%3B236.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd1qm01393a%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd1qm01393a%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Dispiroacridine-indacenobisthiophene%20positional%20isomers%3A%20impact%20of%20the%20bridge%20on%20the%20physicochemical%20properties%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jean-David%22%2C%22lastName%22%3A%22Peltier%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Benoit%22%2C%22lastName%22%3A%22Heinrich%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Bertrand%22%2C%22lastName%22%3A%22Donnio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Olzhas%20A.%22%2C%22lastName%22%3A%22Ibraikulov%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Thomas%22%2C%22lastName%22%3A%22Heiser%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Nicolas%22%2C%22lastName%22%3A%22Leclerc%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Joelle%22%2C%22lastName%22%3A%22Rault-Berthelot%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Cyril%22%2C%22lastName%22%3A%22Poriel%22%7D%5D%2C%22abstractNote%22%3A%22We%20report%20the%20influence%20of%20positional%20isomerism%20on%20the%20electronic%20%28electrochemical%20HOMO%5C%2FLUMO%20energy%20levels%29%2C%20photophysical%20and%20physical%20properties%20%28molecular%20organization%2C%20crystallinity%20and%20phase%20transitions%29%20and%20charge%20transport%20properties%20of%20dispiroacridine-indacenobisthiophene%20positional%20isomers.%20The%20isomers%20differ%20from%20the%20central%20indacenobisthiophene%20%28IDT%29%20core%2C%20which%20displays%20either%20para%20or%20meta%20linkages.%20We%20show%20that%20the%20spiro-connected%20phenylacridine%20bridges%20have%20a%20significant%20influence%20on%20all%20these%20properties%20and%20particularly%20on%20the%20charge%20transport%20mobility%20values%2C%20which%20were%20found%20to%20be%20higher%20in%20the%20meta%20isomer%20than%20in%20the%20para%20isomer.%20This%20finding%20is%20different%20to%20what%20was%20reported%20in%20the%20literature%20for%20the%20other%20couples%20of%20IDT-based%20isomers%20and%20shows%20the%20key%20role%20played%20by%20the%20spiro-connected%20fragments%20in%20these%20molecular%20systems.%22%2C%22date%22%3A%222022%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fd1qm01393a%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fd1qm01393a%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22IEGKATUQ%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A11%3A59Z%22%7D%7D%2C%7B%22key%22%3A%22L3T9W9A4%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Roulland%20et%20al.%22%2C%22parsedDate%22%3A%222022%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EF.%20Roulland%2C%20G.%20Roseau%2C%20A.P.%20Corredor%2C%20L.%20Wendling%2C%20G.%20Krieger%2C%20C.%20Lef%26%23xE8%3Bvre%2C%20M.%20Trassin%2C%20G.%20Pourroy%2C%20N.%20Viart%2C%20Promoting%20the%20magnetic%20exchanges%20in%20PLD%20deposited%20strained%20films%20of%20FeV2O4%20thin%20films%2C%20Materials%20Chemistry%20and%20Physics%20276%20%282022%29%20125360.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.matchemphys.2021.125360%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.matchemphys.2021.125360%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Promoting%20the%20magnetic%20exchanges%20in%20PLD%20deposited%20strained%20films%20of%20FeV2O4%20thin%20films%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fran%5Cu00e7ois%22%2C%22lastName%22%3A%22Roulland%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22G.%22%2C%22lastName%22%3A%22Roseau%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A.%20Pena%22%2C%22lastName%22%3A%22Corredor%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Laurianne%22%2C%22lastName%22%3A%22Wendling%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guillaume%22%2C%22lastName%22%3A%22Krieger%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Christophe%22%2C%22lastName%22%3A%22Lef%5Cu00e8vre%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Morgan%22%2C%22lastName%22%3A%22Trassin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Genevieve%22%2C%22lastName%22%3A%22Pourroy%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Nathalie%22%2C%22lastName%22%3A%22Viart%22%7D%5D%2C%22abstractNote%22%3A%22Transition%20metal%20complex%20oxides%20are%20remarkable%20for%20the%20richness%20of%20tunable%20physical%20properties%20their%20strongly%20correlated%20electrons%20allow.%20We%20have%20undertaken%20the%20pulsed%20laser%20deposition%20of%20epitaxial%20thin%20films%20of%20the%20magnetically%20frustrated%20FeV2O4%20spinel%20vanadate%20compound.%20A%20precise%20optimization%20of%20the%20oxygen%20pressure%20deposition%20conditions%20was%20necessary%20to%20avoid%20the%20presence%20of%20volatile%20over-oxidated%20vanadium%20while%20avoiding%20the%20presence%20of%20oxygen%20vacancies.%20We%20report%20the%20effect%20of%20the%20epitaxial%20compressive%20stress%20induced%20by%20the%20MgO%20substrates%20on%20the%20structural%20and%20magnetic%20properties%20of%20the%20films.%20The%20surprising%20films%20lattice%20parameters%20reduction%20in%20all%20directions%20lead%20to%20an%20unusual%20auxetic%20behaviour.%20The%20reduced%20volume%20of%20the%20cell%20results%20in%20a%20decrease%20of%20the%20V-V%20distances.%20This%20allows%20a%2050%20K%20increase%20of%20the%20magnetic%20transition%20temperature%2C%20which%20now%20reaches%20160%20K.%20The%20epitaxial%20strain%20tuning%20of%20the%20magnetic%20order%20in%20the%20iron-based%20vanadates%20open%20new%20degrees%20of%20freedom%20towards%20the%20integration%20of%20the%20films%20in%20oxide%20electronics.%22%2C%22date%22%3A%222022%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.matchemphys.2021.125360%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.matchemphys.2021.125360%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220254-0584%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22SB8Q592R%22%2C%22UBUT97QT%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A12%3A40Z%22%7D%7D%2C%7B%22key%22%3A%22H7Z6ZSYS%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Shoji%20et%20al.%22%2C%22parsedDate%22%3A%222022%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Shoji%2C%20N.%20Watanabe%2C%20Y.%20Hori%2C%20K.%20Furuya%2C%20M.%20Umemura%2C%20M.%20Boero%2C%20Y.%20Shigeta%2C%20Comprehensive%20Search%20of%20Stable%20Isomers%20of%20Alanine%20and%20Alanine%20Precursors%20in%20Prebiotic%20Syntheses%2C%20Astrobiology%2022%20%282022%29%201129%26%23x2013%3B1142.%20%3Ca%20class%3D%27zp-ItemURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1089%5C%2Fast.2022.0011%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1089%5C%2Fast.2022.0011%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Comprehensive%20Search%20of%20Stable%20Isomers%20of%20Alanine%20and%20Alanine%20Precursors%20in%20Prebiotic%20Syntheses%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mitsuo%22%2C%22lastName%22%3A%22Shoji%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Natsuki%22%2C%22lastName%22%3A%22Watanabe%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yuta%22%2C%22lastName%22%3A%22Hori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenji%22%2C%22lastName%22%3A%22Furuya%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masayuki%22%2C%22lastName%22%3A%22Umemura%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yasuteru%22%2C%22lastName%22%3A%22Shigeta%22%7D%5D%2C%22abstractNote%22%3A%22Enantiomeric%20excesses%20of%20l-amino%20acids%20have%20been%20detected%20in%20meteorites%3B%20however%2C%20their%20molecular%20mechanism%20and%20prebiotic%20syntheses%20are%20still%20a%20matter%20of%20debate.%20To%20elucidate%20the%20origin%20of%20homochirality%2C%20alanine%20and%20the%20chiral%20precursors%20formed%20in%20prebiotic%20processes%20were%20investigated%20with%20regard%20to%20their%20stabilities%20among%20their%20isomers%20by%20employing%20the%20minimum%20energy%20principle%2C%20namely%2C%20the%20abundancy%20of%20a%20molecule%20in%20the%20interstellar%20medium%20is%20directly%20correlated%20to%20the%20stability%20among%20isomers.%20To%20facilitate%20the%20search%20for%20possible%20isomers%2C%20we%20developed%20a%20new%20isomer%20search%20algorithm%2C%20the%20random%20connection%20method%2C%20and%20performed%20a%20thorough%20search%20for%20all%20the%20stable%20isomers%20within%20a%20given%20chemical%20formula.%20We%20found%20that%20alanine%20and%20most%20of%20its%20precursors%20are%20located%20at%20higher%20energy%20by%20more%20than%205.7%20kcal%20mol%5Cu22121%2C%20with%20respect%20to%20the%20most%20stable%20isomer%20that%20consists%20of%20a%20linear-chain%20structure%2C%20whereas%20only%20the%202-aminopropanenitrile%20is%20the%20most%20stable%20isomer%20among%20all%20others%20possible.%20The%20inherent%20stability%20of%20the%20%5Cu03b1-amino%20nitrile%20suggests%20that%20the%202-aminopropanenitrile%20is%20the%20dominant%20contribution%20in%20the%20formation%20of%20the%20common%20enantiomeric%20excess%20over%20%5Cu03b1-amino%20acids.%22%2C%22date%22%3A%222022%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1089%5C%2Fast.2022.0011%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1089%5C%2Fast.2022.0011%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A13%3A01Z%22%7D%7D%2C%7B%22key%22%3A%22FY9IGBBF%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Shoji%20et%20al.%22%2C%22parsedDate%22%3A%222022%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Shoji%2C%20T.%20Murakawa%2C%20S.%20Nakanishi%2C%20M.%20Boero%2C%20Y.%20Shigeta%2C%20H.%20Hayashi%2C%20T.%20Okajima%2C%20Molecular%20mechanism%20of%20a%20large%20conformational%20change%20of%20the%20quinone%20cofactor%20in%20the%20semiquinone%20intermediate%20of%20bacterial%20copper%20amine%20oxidase%2C%20Chemical%20Science%2013%20%282022%29%2010923%26%23x2013%3B10938.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd2sc01356h%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd2sc01356h%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Molecular%20mechanism%20of%20a%20large%20conformational%20change%20of%20the%20quinone%20cofactor%20in%20the%20semiquinone%20intermediate%20of%20bacterial%20copper%20amine%20oxidase%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mitsuo%22%2C%22lastName%22%3A%22Shoji%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Takeshi%22%2C%22lastName%22%3A%22Murakawa%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Shota%22%2C%22lastName%22%3A%22Nakanishi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yasuteru%22%2C%22lastName%22%3A%22Shigeta%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hideyuki%22%2C%22lastName%22%3A%22Hayashi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Toshihide%22%2C%22lastName%22%3A%22Okajima%22%7D%5D%2C%22abstractNote%22%3A%22Copper%20amine%20oxidase%20from%20Arthrobacter%20globiformis%20%28AGAO%29%20catalyses%20the%20oxidative%20deamination%20of%20primary%20amines%20via%20a%20large%20conformational%20change%20of%20a%20topaquinone%20%28TPQ%29%20cofactor%20during%20the%20semiquinone%20formation%20step.%20This%20conformational%20change%20of%20TPQ%20occurs%20in%20the%20presence%20of%20strong%20hydrogen%20bonds%20and%20neighboring%20bulky%20amino%20acids%2C%20especially%20the%20conserved%20Asn381%2C%20which%20restricts%20TPQ%20conformational%20changes%20over%20the%20catalytic%20cycle.%20Whether%20such%20a%20semiquinone%20intermediate%20is%20catalytically%20active%20or%20inert%20has%20been%20a%20matter%20of%20debate%20in%20copper%20amine%20oxidases.%20Here%2C%20we%20show%20that%20the%20reaction%20rate%20of%20the%20Asn381Ala%20mutant%20decreases%20160-fold%2C%20and%20the%20X-ray%20crystal%20structures%20of%20the%20mutant%20reveals%20a%20TPQ-flipped%20conformation%20in%20both%20the%20oxidized%20and%20reduced%20states%2C%20preceding%20semiquinone%20formation.%20Our%20hybrid%20quantum%20mechanics%5C%2Fmolecular%20mechanics%20%28QM%5C%2FMM%29%20simulations%20show%20that%20the%20TPQ%20conformational%20change%20is%20realized%20through%20the%20sequential%20steps%20of%20the%20TPQ%20ring-rotation%20and%20slide.%20We%20determine%20that%20the%20bulky%20side%20chain%20of%20Asn381%20hinders%20the%20undesired%20TPQ%20ring-rotation%20in%20the%20oxidized%20form%2C%20favoring%20the%20TPQ%20ring-rotation%20in%20reduced%20TPQ%20by%20a%20further%20stabilization%20leading%20to%20the%20TPQ%20semiquinone%20form.%20The%20acquired%20conformational%20flexibility%20of%20TPQ%20semiquinone%20promotes%20a%20high%20reactivity%20of%20Cu%28i%29%20to%20O-2%2C%20suggesting%20that%20the%20semiquinone%20form%20is%20catalytically%20active%20for%20the%20subsequent%20oxidative%20half-reaction%20in%20AGAO.%20The%20ingenious%20molecular%20mechanism%20exerted%20by%20TPQ%20to%20achieve%20the%20%5C%22state-specific%5C%22%20reaction%20sheds%20new%20light%20on%20a%20drastic%20environmental%20transformation%20around%20the%20catalytic%20center.%22%2C%22date%22%3A%222022%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fd2sc01356h%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fd2sc01356h%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222041-6520%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A12%3A58Z%22%7D%7D%2C%7B%22key%22%3A%22QPTL4E88%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Wang%20et%20al.%22%2C%22parsedDate%22%3A%222022%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EQ.%20Wang%2C%20S.%20Santos%2C%20C.A.%20Urbina-Blanco%2C%20W.%20Zhou%2C%20Y.%20Yang%2C%20M.%20Marinova%2C%20S.%20Heyte%2C%20T.-R.%20Joelle%2C%20O.%20Ersen%2C%20W.%20Baaziz%2C%20O.%20Safonova%20V.%2C%20M.%20Saeys%2C%20V.V.%20Ordomsky%2C%20Ru%28III%29%20single%20site%20solid%20micellar%20catalyst%20for%20selective%20aqueous%20phase%20hydrogenation%20of%20carbonyl%20groups%20in%20biomass-derived%20compounds%2C%20Applied%20Catalysis%20B-Environmental%20300%20%282022%29%20120730.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.apcatb.2021.120730%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.apcatb.2021.120730%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Ru%28III%29%20single%20site%20solid%20micellar%20catalyst%20for%20selective%20aqueous%20phase%20hydrogenation%20of%20carbonyl%20groups%20in%20biomass-derived%20compounds%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Qiyan%22%2C%22lastName%22%3A%22Wang%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sara%22%2C%22lastName%22%3A%22Santos%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Cesar%20A.%22%2C%22lastName%22%3A%22Urbina-Blanco%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Wenjuan%22%2C%22lastName%22%3A%22Zhou%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yong%22%2C%22lastName%22%3A%22Yang%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Maya%22%2C%22lastName%22%3A%22Marinova%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Svetlana%22%2C%22lastName%22%3A%22Heyte%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Thuriot-Roukos%22%2C%22lastName%22%3A%22Joelle%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ovidiu%22%2C%22lastName%22%3A%22Ersen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Walid%22%2C%22lastName%22%3A%22Baaziz%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Olga%2C%20V%22%2C%22lastName%22%3A%22Safonova%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mark%22%2C%22lastName%22%3A%22Saeys%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Vitaly%20V.%22%2C%22lastName%22%3A%22Ordomsky%22%7D%5D%2C%22abstractNote%22%3A%22Catalytic%20processes%20in%20water%20have%20a%20lower%20environmental%20impact%2C%20cost%2C%20and%20toxicity%20than%20in%20organic%20solvents.%20Considering%20the%20high%20content%20of%20water%20in%20biomass%2C%20it%20would%20be%20natural%20to%20use%20aqueous%20phase%20catalytic%20technology%20for%20the%20production%20of%20valuable%20products.%20However%2C%20in%20the%20aqueous%20phase%2C%20most%20metal-based%20catalysts%20suffer%20from%20low%20activity%2C%20low%20selectivity%20and%20deactivation%20due%20to%20metal%20oxidation%20and%20leaching.%20In%20this%20paper%2C%20we%20propose%20a%20solid%20micellar%20Ru%20catalyst%20%28Ru%28III%29%40MCM%29%20based%20on%20single-site%20Ru%28III%29%20species%20stabilized%20by%20cetyltrimethylammonium%20%28CTA%2B%29%20surfactant%20and%20immobilized%20in%20the%20walls%20of%20MCM-41%20for%20the%20selective%20aqueous%20phase%20hydrogenation%20of%20carbonyl%20groups.%20This%20catalyst%20demonstrates%20exceptional%20selectivity%2C%20activity%2C%20and%20stability%20in%20comparison%20with%20conventional%20metallic%20catalysts.%20DFT%20modeling%20suggests%20that%20the%20reaction%20proceeds%20via%20heterolytic%20dissociation%20of%20hydrogen%2C%20forming%20a%20Ru-Hydride%20species%2C%20and%20subsequent%20hydride%20transfer%20to%20the%20carbonyl%20group.%20Water%20plays%20a%20key%20role%20in%20avoiding%20product%20inhibition.%22%2C%22date%22%3A%222022%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.apcatb.2021.120730%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.apcatb.2021.120730%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220926-3373%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%226739WBV7%22%2C%22DEB5KWFS%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-01-08T16%3A13%3A42Z%22%7D%7D%5D%7D
[1]
M. Boero, K.M. Bui, K. Shiraishi, K. Ishisone, Y. Kangawa, A. Oshiyama, An atomistic insight into reactions and free-energy profiles of NH3 and Ga on GaN surfaces during the epitaxial growth, Applied Surface Science 599 (2022) 153935.
https://doi.org/10.1016/j.apsusc.2022.153935.
[1]
C. Burel, O. Ibrahim, E. Marino, H. Bharti, C.B. Murray, B. Donnio, Z. Fakhraai, R. Dreyfus, Tunable Plasmonic Microcapsules with Embedded Noble Metal Nanoparticles for Optical Microsensing, ACS Applied Nano Materials 5 (2022) 2828–2838.
https://doi.org/10.1021/acsanm.1c04542.
[1]
D. Gentili, G. Ori, Reversible assembly of nanoparticles: theory, strategies and computational simulations., Nanoscale 14 (2022) 14385–14432.
https://doi.org/10.1039/d2nr02640f.
[1]
K. Ishisone, G. Ori, M. Boero, Structural, dynamical, and electronic properties of the ionic liquid 1-ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide., Physical Chemistry Chemical Physics : PCCP 24 (2022) 9597–9607.
https://doi.org/10.1039/d2cp00741j.
[1]
A. Lambrecht, C. Massobrio, M. Boero, G. Ori, E. Martin, Atomic structure of amorphous SiN: Combining Car-Parrinello and Born-Oppenheimer first-principles molecular dynamics, Computational Materials Science 211 (2022) 111555.
https://doi.org/10.1016/j.commatsci.2022.111555.
[1]
E. Martin, I.B. Amiehe Essomba, K. Ishisone, M. Boero, G. Ori, C. Massobrio, Impact of Dispersion Force Schemes on Liquid Systems: Comparing Efficiency and Drawbacks for Well-Targeted Test Cases, Molecules 27 (2022).
https://doi.org/10.3390/molecules27249034.
[1]
F. Omeis, Z. Boubegtiten-Fezoua, A.F.S. Seica, R. Bernard, M.H. Iqbal, N. Javahiraly, R.M.A. Vergauwe, H. Majjad, F. Boulmedais, D. Moss, P. Hellwig, Plasmonic Resonant Nanoantennas Induce Changes in the Shape and the Intensity of Infrared Spectra of Phospholipids, Molecules 27 (2022) 62.
https://doi.org/10.3390/molecules27010062.
[1]
F. Payet, C. Bouillet, F. Leroux, C. Leuvrey, P. Rabu, F. Schosseler, C. Taviot-Guého, G. Rogez, Fast and efficient shear-force assisted production of covalently functionalized oxide nanosheets, Journal of Colloid and Interface Science 607 (2022) 621–632.
https://doi.org/10.1016/j.jcis.2021.08.213.
[1]
J.-D. Peltier, B. Heinrich, B. Donnio, O.A. Ibraikulov, T. Heiser, N. Leclerc, J. Rault-Berthelot, C. Poriel, Dispiroacridine-indacenobisthiophene positional isomers: impact of the bridge on the physicochemical properties, Materials Chemistry Frontiers 6 (2022) 225–236.
https://doi.org/10.1039/d1qm01393a.
[1]
F. Roulland, G. Roseau, A.P. Corredor, L. Wendling, G. Krieger, C. Lefèvre, M. Trassin, G. Pourroy, N. Viart, Promoting the magnetic exchanges in PLD deposited strained films of FeV2O4 thin films, Materials Chemistry and Physics 276 (2022) 125360.
https://doi.org/10.1016/j.matchemphys.2021.125360.
[1]
M. Shoji, N. Watanabe, Y. Hori, K. Furuya, M. Umemura, M. Boero, Y. Shigeta, Comprehensive Search of Stable Isomers of Alanine and Alanine Precursors in Prebiotic Syntheses, Astrobiology 22 (2022) 1129–1142.
https://doi.org/10.1089/ast.2022.0011.
[1]
M. Shoji, T. Murakawa, S. Nakanishi, M. Boero, Y. Shigeta, H. Hayashi, T. Okajima, Molecular mechanism of a large conformational change of the quinone cofactor in the semiquinone intermediate of bacterial copper amine oxidase, Chemical Science 13 (2022) 10923–10938.
https://doi.org/10.1039/d2sc01356h.
[1]
Q. Wang, S. Santos, C.A. Urbina-Blanco, W. Zhou, Y. Yang, M. Marinova, S. Heyte, T.-R. Joelle, O. Ersen, W. Baaziz, O. Safonova V., M. Saeys, V.V. Ordomsky, Ru(III) single site solid micellar catalyst for selective aqueous phase hydrogenation of carbonyl groups in biomass-derived compounds, Applied Catalysis B-Environmental 300 (2022) 120730.
https://doi.org/10.1016/j.apcatb.2021.120730.
1839302
NZSFH59F
2021
1
surface-science-reports
50
creator
asc
year
7054
https://www.ipcms.unistra.fr/plugins/zotpress/
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3Afalse%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%22QF25U968%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Bouzid%20et%20al.%22%2C%22parsedDate%22%3A%222021%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EA.%20Bouzid%2C%20T.-L.%20Pham%2C%20Z.%20Chaker%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20Y.-H.%20Shin%2C%20G.%20Ori%2C%20Quantitative%20assessment%20of%20the%20structure%20of%20Ge20Te73I7%20chalcohalide%20glass%20by%20first-principles%20molecular%20dynamics%2C%20Physical%20Review%20B%20103%20%282021%29%20094204.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.103.094204%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.103.094204%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Quantitative%20assessment%20of%20the%20structure%20of%20Ge20Te73I7%20chalcohalide%20glass%20by%20first-principles%20molecular%20dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tan-Lien%22%2C%22lastName%22%3A%22Pham%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ziyad%22%2C%22lastName%22%3A%22Chaker%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Young-Han%22%2C%22lastName%22%3A%22Shin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%5D%2C%22abstractNote%22%3A%22First-principles%20molecular%20dynamics%2C%20within%20the%20density%20functional%20theory%20framework%2C%20is%20employed%20to%20assess%20the%20structural%20properties%20of%20the%20chalcohalide%20glass%20Ge20Te73I7%20%28gGTI%29.%20The%20calculated%20total%20x-ray%20and%20neutron%20structure%20factors%20are%20in%20quantitative%20agreement%20with%20the%20experimental%20counterparts.%20Glassy%20gGTI%20features%20a%20predominantly%20Ge-centered%20tetrahedral%20network%20moderately%20altered%20by%20iodine%20atoms.%20Compared%20to%20glassy%20Ge20Te80%2C%20gGTI%20shows%20a%20substantial%20decrease%20in%20the%20number%20of%20Ge-Te%20bonds%20as%20the%20presence%20of%20I%20leads%20to%20a%20breaking%20of%20the%20Ge-Te%20connections%20and%20favors%20the%20formation%20of%20Ge-I%20terminations%20where%20iodine%20atoms%20are%20onefold.%20Furthermore%2C%20we%20remark%20a%20concomitant%20increase%20of%20mixed%20GeTe4-xIx%20tetrahedra%20and%20GeTe3-xIx%20trigonal%20pyramidal%20units.%20A%20minor%20yet%20not-negligible%20fraction%20of%20iodine%20atoms%20forms%20Te-I%20bonds.%20Breaking%20of%20Ge-Te%20connections%20and%20formation%20of%20terminal%20Ge%28Te%29-I%20bonds%20results%20in%20a%20reduced%20average%20coordination%20number%20associated%20with%20an%20increase%20of%20undercoordinated%20Ge%20at%20the%20expense%20of%20fourfold%20coordinated%20Ge%20atoms.%20The%20analysis%20of%20chains%20and%20rings%20statistics%20reveals%20the%20presence%20of%20both%20linear%20and%20cross-linked%20chains%20made%20of%20Te%20and%20five-members%20rings%20containing%20at%20least%20one%20Te-Te%20or%20one%20Ge-Ge%20homopolar%20bond.%22%2C%22date%22%3A%222021%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevB.103.094204%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevB.103.094204%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222469-9950%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-02-02T13%3A20%3A03Z%22%7D%7D%2C%7B%22key%22%3A%22WZ5G2SIY%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Duong%20et%20al.%22%2C%22parsedDate%22%3A%222021%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ET.-Q.%20Duong%2C%20A.%20Bouzid%2C%20C.%20Massobrio%2C%20G.%20Ori%2C%20M.%20Boero%2C%20E.%20Martin%2C%20First-principles%20thermal%20transport%20in%20amorphous%20Ge2Sb2Te5%20at%20the%20nanoscale%2C%20RSC%20Advances%2011%20%282021%29%2010747%26%23x2013%3B10752.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd0ra10408f%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd0ra10408f%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22First-principles%20thermal%20transport%20in%20amorphous%20Ge2Sb2Te5%20at%20the%20nanoscale%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Thuy-Quynh%22%2C%22lastName%22%3A%22Duong%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%5D%2C%22abstractNote%22%3A%22Achieving%20a%20precise%20understanding%20of%20nanoscale%20thermal%20transport%20in%20phase%20change%20materials%20%28PCMs%29%2C%20such%20as%20Ge2Sb2Te5%20%28GST%29%2C%20is%20the%20key%20of%20thermal%20management%20in%20nanoelectronics%2C%20photonic%20and%20neuromorphic%20applications%20using%20non-volatile%20memories.%20By%20resorting%20to%20a%20first-principles%20approach%20to%20calculate%20the%20thermal%20conductivity%20of%20amorphous%20GST%2C%20we%20found%20that%20size%20effects%20and%20heat%20transport%20via%20propagative%20modes%20persist%20well%20beyond%20extended%20range%20order%20distances%20typical%20of%20disordered%20network-forming%20materials.%20Values%20obtained%20are%20in%20quantitative%20agreement%20with%20the%20experimental%20data%2C%20by%20revealing%20a%20strong%20size%20dependence%20of%20the%20thermal%20conductivity%20down%20to%20the%201.7-10%20nm%20range%2C%20fully%20covering%20the%20scale%20of%20current%20PCMs-based%20devices.%20In%20particular%2C%20a%20reduction%20of%20thermal%20conductivity%20as%20large%20as%2075%25%20occurs%20for%20dimensions%20lying%20below%202%20nm.%20These%20results%20provide%20a%20quantitative%20description%20of%20the%20thermal%20properties%20of%20amorphous%20GST%20at%20the%20nanoscale%20and%20are%20expected%20to%20underpin%20the%20development%20of%20PCM-based%20device%20applications.%22%2C%22date%22%3A%222021%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fd0ra10408f%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fd0ra10408f%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222026-02-02T14%3A10%3A15Z%22%7D%7D%2C%7B%22key%22%3A%22W9TAUHKU%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Le%20Roux%20et%20al.%22%2C%22parsedDate%22%3A%222021%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ES.%20Le%20Roux%2C%20G.%20Ori%2C%20S.%20Bellemin-Laponnaz%2C%20M.%20Boero%2C%20Tridentate%20complexes%20of%20group%204%20bearing%20bis-aryloxide%20N-heterocyclic%20carbene%20ligand%3A%20Structure%2C%20spin%20density%20and%20charge%20states%2C%20Chemical%20Physics%20Letters%20781%20%282021%29%20138888.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cplett.2021.138888%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cplett.2021.138888%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Tridentate%20complexes%20of%20group%204%20bearing%20bis-aryloxide%20N-heterocyclic%20carbene%20ligand%3A%20Structure%2C%20spin%20density%20and%20charge%20states%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Stephane%22%2C%22lastName%22%3A%22Bellemin-Laponnaz%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22Metal%20carbene%20complexes%20represent%20an%20ubiquitous%20class%20of%20compounds%20in%20organomettalic%20chemistry%20able%20to%20trigger%20a%20wealth%20of%20catalytic%20reactions%20of%20both%20fundamental%20and%20industrial%20processes.%20By%20resorting%20to%20first%20principles%20approaches%2C%20we%20focus%20on%20the%20fundamental%20features%20of%20these%20complexes%20in%20which%20the%20metal%20center%20can%20be%20either%20Hf%2C%20Ti%20or%20Zr.%20These%20specific%20systems%20show%20interesting%20features%20in%20their%20neutral%20and%20charged%20%28%2B1%29%20states.%20Yet%2C%20we%20provide%20evidence%20of%20the%20fact%20that%20the%20nature%20of%20the%20metallic%20ion%20does%20not%20have%20a%20significant%20impact%20on%20the%20structure%20of%20the%20ligand.%20Conversely%2C%20the%20removal%20of%20one%20electron%20%28charge%20state%20%2B1%29%20induces%20a%20perturbation%20of%20the%20ligand%20conformation%20and%20this%2C%20in%20turn%2C%20is%20prone%20to%20affect%20the%20catalytic%20properties%20of%20the%20complex%2C%20carrying%20the%20singly%20occupied%20electronic%20level%20%28spin%20distribution%20of%20the%20system%29%20delocalized%20over%20the%20carbene%20ligand.%22%2C%22date%22%3A%222021%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.cplett.2021.138888%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.cplett.2021.138888%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220009-2614%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22CF4ZI7HM%22%2C%22ITCCYZMF%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-10-21T09%3A49%3A44Z%22%7D%7D%2C%7B%22key%22%3A%223AICA6A7%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Mishima%20et%20al.%22%2C%22parsedDate%22%3A%222021%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Mishima%2C%20M.%20Shoji%2C%20Y.%20Umena%2C%20M.%20Boero%2C%20Y.%20Shigeta%2C%20Estimation%20of%20the%20relative%20contributions%20to%20the%20electronic%20energy%20transfer%20rates%20based%20on%20F%26%23xF6%3Brster%20theory%3A%20The%20case%20of%20C-phycocyanin%20chromophores%2C%20Biophysics%20and%20Physicobiology%2018%20%282021%29%20196%26%23x2013%3B214.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.2142%5C%2Fbiophysico.bppb-v18.021%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.2142%5C%2Fbiophysico.bppb-v18.021%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Estimation%20of%20the%20relative%20contributions%20to%20the%20electronic%20energy%20transfer%20rates%20based%20on%20F%5Cu00f6rster%20theory%3A%20The%20case%20of%20C-phycocyanin%20chromophores%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenji%22%2C%22lastName%22%3A%22Mishima%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mitsuo%22%2C%22lastName%22%3A%22Shoji%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yasufumi%22%2C%22lastName%22%3A%22Umena%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yasuteru%22%2C%22lastName%22%3A%22Shigeta%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%222021%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.2142%5C%2Fbiophysico.bppb-v18.021%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.2142%5C%2Fbiophysico.bppb-v18.021%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222022-02-08T08%3A39%3A59Z%22%7D%7D%2C%7B%22key%22%3A%223NX7NYI4%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Pietrucci%20et%20al.%22%2C%22parsedDate%22%3A%222021%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EF.%20Pietrucci%2C%20M.%20Boero%2C%20W.%20Andreoni%2C%20How%20natural%20materials%20remove%20heavy%20metals%20from%20water%3A%20mechanistic%20insights%20from%20molecular%20dynamics%20simulations%2C%20Chemical%20Science%2012%20%282021%29%202979%26%23x2013%3B2985.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd0sc06204a%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd0sc06204a%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22How%20natural%20materials%20remove%20heavy%20metals%20from%20water%3A%20mechanistic%20insights%20from%20molecular%20dynamics%20simulations%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fabio%22%2C%22lastName%22%3A%22Pietrucci%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Wanda%22%2C%22lastName%22%3A%22Andreoni%22%7D%5D%2C%22abstractNote%22%3A%22Water%20pollution%20by%20heavy%20metals%20is%20of%20increasing%20concern%20due%20to%20its%20devastating%20effects%20on%20the%20environment%20and%20on%20human%20health.%20For%20the%20removal%20of%20heavy%20metals%20from%20water%20sources%2C%20natural%20materials%2C%20such%20as%20spent-coffee-grains%20or%20orange%5C%2Fbanana%5C%2Fchestnut%20peels%2C%20appear%20to%20offer%20a%20potential%20cheap%20alternative%20to%20more%20sophisticated%20and%20costly%20technologies%20currently%20in%20use.%20However%2C%20in%20order%20to%20employ%20them%20effectively%2C%20it%20is%20necessary%20to%20gain%20a%20deeper%20understanding%20-%20at%20the%20molecular%20level%20-%20of%20the%20heavy%20metals-bioorganic-water%20system%20and%20exploit%20the%20power%20of%20computer%20simulations.%20As%20a%20step%20in%20this%20direction%2C%20we%20investigate%20via%20atomistic%20simulations%20the%20capture%20of%20lead%20ions%20from%20water%20by%20hemicellulose%20-%20the%20latter%20being%20representative%20of%20the%20polysaccharides%20that%20are%20common%20components%20of%20vegetables%20and%20fruit%20peels%20-%20as%20well%20as%20the%20reverse%20process.%20A%20series%20of%20independent%20molecular%20dynamics%20simulations%2C%20both%20classical%20and%20ab%20initio%2C%20reveals%20a%20coherent%20scenario%20which%20is%20consistent%20with%20what%20one%20would%20expect%20of%20an%20efficient%20capture%2C%20i.e.%20that%20it%20be%20fast%20and%20irreversible%3A%20%28i%29%20binding%20of%20the%20metal%20ions%20via%20adsorption%20is%20found%20to%20happen%20spontaneously%20on%20both%20carboxylate%20and%20hydroxide%20functional%20groups%3B%20%28ii%29%20in%20contrast%2C%20metal%20ion%20desorption%2C%20leading%20to%20solvation%20in%20water%2C%20involves%20sizable%20free-energy%20barriers.%22%2C%22date%22%3A%222021%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fd0sc06204a%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fd0sc06204a%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222041-6520%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222021-05-11T09%3A01%3A38Z%22%7D%7D%5D%7D
[1]
A. Bouzid, T.-L. Pham, Z. Chaker, M. Boero, C. Massobrio, Y.-H. Shin, G. Ori, Quantitative assessment of the structure of Ge20Te73I7 chalcohalide glass by first-principles molecular dynamics, Physical Review B 103 (2021) 094204.
https://doi.org/10.1103/PhysRevB.103.094204.
[1]
T.-Q. Duong, A. Bouzid, C. Massobrio, G. Ori, M. Boero, E. Martin, First-principles thermal transport in amorphous Ge2Sb2Te5 at the nanoscale, RSC Advances 11 (2021) 10747–10752.
https://doi.org/10.1039/d0ra10408f.
[1]
S. Le Roux, G. Ori, S. Bellemin-Laponnaz, M. Boero, Tridentate complexes of group 4 bearing bis-aryloxide N-heterocyclic carbene ligand: Structure, spin density and charge states, Chemical Physics Letters 781 (2021) 138888.
https://doi.org/10.1016/j.cplett.2021.138888.
[1]
K. Mishima, M. Shoji, Y. Umena, M. Boero, Y. Shigeta, Estimation of the relative contributions to the electronic energy transfer rates based on Förster theory: The case of C-phycocyanin chromophores, Biophysics and Physicobiology 18 (2021) 196–214.
https://doi.org/10.2142/biophysico.bppb-v18.021.
[1]
F. Pietrucci, M. Boero, W. Andreoni, How natural materials remove heavy metals from water: mechanistic insights from molecular dynamics simulations, Chemical Science 12 (2021) 2979–2985.
https://doi.org/10.1039/d0sc06204a.
1839302
NZSFH59F
2020
1
surface-science-reports
50
creator
asc
year
7054
https://www.ipcms.unistra.fr/plugins/zotpress/
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3Afalse%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%224HZE8NW8%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Bui%20et%20al.%22%2C%22parsedDate%22%3A%222020%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.M.%20Bui%2C%20M.%20Boero%2C%20K.%20Shiraishi%2C%20A.%20Oshiyama%2C%20A%20two-dimensional%20liquid-like%20phase%20on%20Ga-rich%20GaN%20%280001%29%20surfaces%20evidenced%20by%20first%20principles%20molecular%20dynamics%2C%20in%3A%20Japanese%20Journal%20of%20Applied%20Physics%2C%202020%3A%20p.%20SGGK04.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.7567%5C%2F1347-4065%5C%2Fab650b%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.7567%5C%2F1347-4065%5C%2Fab650b%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22conferencePaper%22%2C%22title%22%3A%22A%20two-dimensional%20liquid-like%20phase%20on%20Ga-rich%20GaN%20%280001%29%20surfaces%20evidenced%20by%20first%20principles%20molecular%20dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kieu%20My%22%2C%22lastName%22%3A%22Bui%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenji%22%2C%22lastName%22%3A%22Shiraishi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Atsushi%22%2C%22lastName%22%3A%22Oshiyama%22%7D%5D%2C%22abstractNote%22%3A%22First%20principles%20molecular%20dynamics%20simulations%20have%20been%20used%20as%20a%20tool%20to%20investigate%20at%20an%20atomistic%20level%20the%20finite%20temperature%20behavior%20of%20a%20typical%20GaN%20growing%20surface.%20At%201300%20K%2C%20Ga%20adatoms%20show%20a%20high%20degree%20of%20destabilization%20and%20a%20departure%20of%20the%20diffusivity%20from%20an%20ordinary%20Arrhenius%20trend.%20Two%20signatures%2C%20a%20drastic%20change%20in%20the%20radial%20distribution%20function%20and%20a%20sudden%20increase%20of%20the%20diffusion%20constant%2C%20allow%20to%20quantify%20such%20a%20diffusion%20process.%20Such%20a%20high%20diffusivity%20results%20in%20the%20arising%20of%20a%20pronounced%20peak%20at%20about%202.6%20angstrom%2C%20typical%20of%20Ga-Ga%20bonds%2C%20indicating%20a%20tendency%20to%20cluster%20together%20of%20the%20Ga%20adatoms.%20Along%20with%20the%20remarkable%20diffusivity%20observed%20at%201300%20K%2C%20this%20result%20indicates%20rather%20clearly%20that%20at%20the%20typical%20experimental%20temperature%2C%20the%20epitaxial%20growth%20occurs%20not%20on%20a%20rigid%20surface%20in%20which%20Ga%20adatoms%20are%20tightly%20bound%20to%20specific%20sites%2C%20but%20rather%20on%20a%20two-dimensional%20liquid%20state.%20%28C%29%202020%20The%20Japan%20Society%20of%20Applied%20Physics%22%2C%22proceedingsTitle%22%3A%22Japanese%20Journal%20of%20Applied%20Physics%22%2C%22conferenceName%22%3A%22International%20Conference%20on%20Solid%20State%20Devices%20and%20Materials%20%28SSDM%29%2C%20Nagoya%20Univ%2C%20Nagoya%2C%20JAPAN%2C%2002-05%20septembre%2C%202019%22%2C%22date%22%3A%222020%22%2C%22eventPlace%22%3A%22%22%2C%22DOI%22%3A%2210.7567%5C%2F1347-4065%5C%2Fab650b%22%2C%22ISBN%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.7567%5C%2F1347-4065%5C%2Fab650b%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222022-01-22T15%3A35%3A11Z%22%7D%7D%2C%7B%22key%22%3A%22E9XIGI5H%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Du%20et%20al.%22%2C%22parsedDate%22%3A%222020%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EJ.%20Du%2C%20J.P.%20Rino%2C%20C.%20Massobrio%2C%20A.N.%20Cormack%2C%20Challenges%20and%20opportunities%20of%20atomistic%20simulations%20for%20glass%20and%20amorphous%20materials%2C%20Journal%20of%20Non-Crystalline%20Solids%20547%20%282020%29%20120270.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2020.120270%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2020.120270%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Challenges%20and%20opportunities%20of%20atomistic%20simulations%20for%20glass%20and%20amorphous%20materials%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jincheng%22%2C%22lastName%22%3A%22Du%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jose%20Pedro%22%2C%22lastName%22%3A%22Rino%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Alastair%20N.%22%2C%22lastName%22%3A%22Cormack%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%222020%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.jnoncrysol.2020.120270%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2020.120270%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220022-3093%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222020-10-14T09%3A29%3A58Z%22%7D%7D%2C%7B%22key%22%3A%22JH8KQNTW%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Duong%20et%20al.%22%2C%22parsedDate%22%3A%222020%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ET.-Q.%20Duong%2C%20C.%20Massobrio%2C%20G.%20Ori%2C%20M.%20Boero%2C%20E.%20Martin%2C%20Thermal%20resistance%20of%20an%20interfacial%20molecular%20layer%20by%20first-principles%20molecular%20dynamics%2C%20Journal%20of%20Chemical%20Physics%20153%20%282020%29%20074704.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F5.0014232%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F5.0014232%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Thermal%20resistance%20of%20an%20interfacial%20molecular%20layer%20by%20first-principles%20molecular%20dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Thuy-Quynh%22%2C%22lastName%22%3A%22Duong%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%5D%2C%22abstractNote%22%3A%22The%20approach-to-equilibrium%20molecular%20dynamics%20%28AEMD%29%20methodology%20is%20applied%20in%20combination%20with%20first-principles%20molecular%20dynamics%20to%20investigate%20the%20thermal%20transfer%20between%20two%20silicon%20blocks%20connected%20by%20a%20molecular%20layer.%20Our%20configuration%20consists%20of%20alkanes%20molecules%20strongly%20coupled%20to%20the%20silicon%20surfaces%20via%20covalent%20bonds.%20In%20phase%201%20of%20AEMD%2C%20the%20two%20Si%20blocks%20are%20thermalized%20at%20high%20and%20low%20temperatures%20to%20form%20the%20hot%20and%20cold%20reservoirs.%20During%20phase%202%20of%20AEMD%2C%20a%20transfer%20between%20reservoirs%20occurs%20until%20thermal%20equilibrium%20is%20reached.%20The%20transfer%20across%20the%20interface%20dominates%20the%20transient%20over%20heat%20conduction%20within%20the%20reservoirs.%20The%20value%20of%20the%20thermal%20interface%20conductance%20is%20in%20agreement%20with%20the%20experimental%20data%20obtained%20for%20analogous%20bonding%20cases%20between%20molecules%20and%20reservoirs.%20The%20dependence%20on%20the%20length%20of%20the%20thermal%20interface%20resistance%20features%20two%20contributions.%20One%20is%20constant%20%28the%20resistance%20at%20the%20silicon%5C%2Fmolecule%20interface%29%2C%20while%20the%20other%20varies%20linearly%20with%20the%20length%20of%20the%20molecular%20chains%20%28diffusive%20transport%29.%20The%20corresponding%20value%20of%20the%20thermal%20conductivity%20agrees%20well%20with%20experiments.%22%2C%22date%22%3A%222020%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1063%5C%2F5.0014232%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1063%5C%2F5.0014232%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220021-9606%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222020-10-09T09%3A02%3A22Z%22%7D%7D%2C%7B%22key%22%3A%22NPZ4EK6G%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Duong%20et%20al.%22%2C%22parsedDate%22%3A%222020%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ET.-Q.%20Duong%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20G.%20Ori%2C%20E.%20Martin%2C%20Heat%20transport%20in%20disordered%20network%20forming%20materials%3A%20Size%20effects%20and%20existence%20of%20propagative%20modes%2C%20Computational%20Materials%20Science%20177%20%282020%29%20109607.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.commatsci.2020.109607%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.commatsci.2020.109607%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Heat%20transport%20in%20disordered%20network%20forming%20materials%3A%20Size%20effects%20and%20existence%20of%20propagative%20modes%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Thuy-Quynh%22%2C%22lastName%22%3A%22Duong%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%5D%2C%22abstractNote%22%3A%22We%20show%20that%20in%20nanoscale%20disordered%20networks%20the%20thermal%20conductivity%20reduces%20with%20respect%20to%20the%20bulk%20value%20in%20a%20way%20remindful%20of%20the%20analogous%20behavior%20in%20nanocrystalline%20systems.%20Our%20rationale%20is%20based%20on%20the%20analogy%20with%20nanostructured%20crystalline%20silicon%20and%20is%20substantiated%20by%20results%20obtained%20%28experimentally%2C%20analytically%20and%20by%20atomic%20scale%20modelling%29%20on%20crystalline%20Si%2C%20amorphous%20Si%2C%20glassy%20SiO2%20and%20glassy%20GeTe4.%20We%20point%20out%20the%20implication%20of%20such%20findings%20on%20the%20potential%20performances%20of%20nanoscale%20heat%20devices.%22%2C%22date%22%3A%222020%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.commatsci.2020.109607%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.commatsci.2020.109607%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220927-0256%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222022-01-22T15%3A38%3A54Z%22%7D%7D%2C%7B%22key%22%3A%22Q8K8FANU%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Essomba%20et%20al.%22%2C%22parsedDate%22%3A%222020%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EI.B.A.%20Essomba%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20G.%20Ori%2C%20Assessing%20the%20Versatility%20of%20Molecular%20Modelling%20as%20a%20Strategy%20for%20Predicting%20Gas%20Adsorption%20Properties%20of%20Chalcogels%2C%20in%3A%20E.%20Levchenko%2C%20Y.%20Dappe%2C%20G.%20Ori%20%28Eds.%29%2C%20THEORY%20AND%20SIMULATION%20IN%20PHYSICS%20FOR%20MATERIALS%20APPLICATIONS%3A%20CUTTING-EDGE%20TECHNIQUES%20IN%20THEORETICAL%20AND%20COMPUTATIONAL%20MATERIALS%20SCIENCE%20%5C%2F%20Edited%20by%20E.V.%20Levchenko...%2C%202020%3A%20pp.%2023%26%23x2013%3B37.%20%3Ca%20class%3D%27zp-ItemURL%27%20href%3D%2710.1007%5C%2F978-3-030-37790-8_2%27%3E10.1007%5C%2F978-3-030-37790-8_2%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22bookSection%22%2C%22title%22%3A%22Assessing%20the%20Versatility%20of%20Molecular%20Modelling%20as%20a%20Strategy%20for%20Predicting%20Gas%20Adsorption%20Properties%20of%20Chalcogels%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Irene%20Berenger%20Amiehe%22%2C%22lastName%22%3A%22Essomba%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22editor%22%2C%22firstName%22%3A%22EV%22%2C%22lastName%22%3A%22Levchenko%22%7D%2C%7B%22creatorType%22%3A%22editor%22%2C%22firstName%22%3A%22YJ%22%2C%22lastName%22%3A%22Dappe%22%7D%2C%7B%22creatorType%22%3A%22editor%22%2C%22firstName%22%3A%22G%22%2C%22lastName%22%3A%22Ori%22%7D%5D%2C%22abstractNote%22%3A%22Modelling%20gas%20adsorption%20of%20porous%20materials%20is%20nowadays%20an%20undeniable%20necessary%20in%20order%20to%20complement%20experiment%20findings%20with%20the%20purpose%20to%20enrich%20our%20fundamental%20understanding%20of%20adsorption%20mechanisms%20as%20well%20as%20develop%20better%20performing%20materials%20for%20gas%20mixture%20separation.%20In%20this%20contribution%2C%20we%20explore%20the%20possibility%20to%20use%20first-principles%20molecular%20dynamics%20%28FPMD%29%20and%20grand%20canonical%20Monte%20Carlo%20%28GCMC%29%20simulations%20to%20target%20the%20gas%20adsorption%20of%20disordered%20nanoporous%20chalcogenides%20%28i.e.%20chalcogels%29.%20This%20computational%20scheme%20allows%20us%20to%20take%20advantage%20of%20the%20ability%20of%20FPMD%20to%20accurately%20describe%20the%20structure%20and%20bonding%20of%20the%20disordered%20nature%20of%20chalcogels%20as%20well%20as%20the%20potential%20of%20GCMC%20to%20model%20the%20adsorption%20mechanisms%20of%20porous%20networks.%20We%20assess%20the%20versatility%20of%20such%20scheme%20by%20evaluating%20the%20role%20of%20pore%20size%2C%20chemical%20stoichiometry%20and%20composition%20for%20multiple%20chalcogenide-based%20systems%20on%20nitrogen%20adsorption%20isotherms.%22%2C%22bookTitle%22%3A%22THEORY%20AND%20SIMULATION%20IN%20PHYSICS%20FOR%20MATERIALS%20APPLICATIONS%3A%20CUTTING-EDGE%20TECHNIQUES%20IN%20THEORETICAL%20AND%20COMPUTATIONAL%20MATERIALS%20SCIENCE%20%5C%2F%20edited%20by%20E.V.%20Levchenko...%22%2C%22date%22%3A%222020%22%2C%22originalDate%22%3A%22%22%2C%22originalPublisher%22%3A%22%22%2C%22originalPlace%22%3A%22%22%2C%22format%22%3A%22%22%2C%22ISBN%22%3A%220933-033X%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%2210.1007%5C%2F978-3-030-37790-8_2%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222022-03-29T13%3A16%3A34Z%22%7D%7D%2C%7B%22key%22%3A%22D6A7PQSS%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Levchenko%20et%20al.%22%2C%22parsedDate%22%3A%222020%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EE.V.%20Levchenko%2C%20Y.J.%20Dappe%2C%20G.%20Ori%2C%20Preface%2C%20in%3A%20E.%20Levchenko%2C%20Y.%20Dappe%2C%20G.%20Ori%20%28Eds.%29%2C%20THEORY%20AND%20SIMULATION%20IN%20PHYSICS%20FOR%20MATERIALS%20APPLICATIONS%3A%20CUTTING-EDGE%20TECHNIQUES%20IN%20THEORETICAL%20AND%20COMPUTATIONAL%20MATERIALS%20SCIENCE%20%5C%2F%20Edited%20by%20Y.J.%20Dappe%20et%20G.%20Ori%2C%202020%3A%20p.%20V%26%23x2013%3BV.%20%3Ca%20class%3D%27zp-ItemURL%27%20href%3D%2710.1007%5C%2F978-3-030-37790-8%27%3E10.1007%5C%2F978-3-030-37790-8%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22bookSection%22%2C%22title%22%3A%22Preface%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Elena%20V.%22%2C%22lastName%22%3A%22Levchenko%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yannick%20J.%22%2C%22lastName%22%3A%22Dappe%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22editor%22%2C%22firstName%22%3A%22EV%22%2C%22lastName%22%3A%22Levchenko%22%7D%2C%7B%22creatorType%22%3A%22editor%22%2C%22firstName%22%3A%22YJ%22%2C%22lastName%22%3A%22Dappe%22%7D%2C%7B%22creatorType%22%3A%22editor%22%2C%22firstName%22%3A%22G%22%2C%22lastName%22%3A%22Ori%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22bookTitle%22%3A%22THEORY%20AND%20SIMULATION%20IN%20PHYSICS%20FOR%20MATERIALS%20APPLICATIONS%3A%20CUTTING-EDGE%20TECHNIQUES%20IN%20THEORETICAL%20AND%20COMPUTATIONAL%20MATERIALS%20SCIENCE%20%5C%2F%20edited%20by%20Y.J.%20Dappe%20et%20G.%20Ori%22%2C%22date%22%3A%222020%22%2C%22originalDate%22%3A%22%22%2C%22originalPublisher%22%3A%22%22%2C%22originalPlace%22%3A%22%22%2C%22format%22%3A%22%22%2C%22ISBN%22%3A%220933-033X%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%2210.1007%5C%2F978-3-030-37790-8%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222022-03-29T13%3A16%3A54Z%22%7D%7D%2C%7B%22key%22%3A%22ATPN95Z8%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Massobrio%20et%20al.%22%2C%22parsedDate%22%3A%222020%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EC.%20Massobrio%2C%20A.%20Bouzid%2C%20M.%20Boero%2C%20G.%20Ori%2C%20E.%20Martin%2C%20S.%20Le%20Roux%2C%20Chalcogenide%20glasses%20for%20innovation%20in%20applied%20science%3A%20fundamental%20issues%20and%20new%20insights%2C%20Journal%20of%20Physics%20D-Applied%20Physics%2053%20%282020%29%20033002.%20%3Ca%20class%3D%27zp-ItemURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1088%5C%2F1361-6463%5C%2Fab48a4%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1088%5C%2F1361-6463%5C%2Fab48a4%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Chalcogenide%20glasses%20for%20innovation%20in%20applied%20science%3A%20fundamental%20issues%20and%20new%20insights%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%5D%2C%22abstractNote%22%3A%22We%20provide%20an%20overview%20of%20what%20has%20been%20accomplished%20by%20our%20team%2C%20since%201998%2C%20in%20the%20area%20of%20first-principles%20molecular%20dynamics%20%28FPMD%29%20modeling%20of%20glassy%20chalcogenides%2C%20a%20prototypical%20family%20of%20network-forming%20disordered%20materials.%20After%20a%20broad%20introduction%20that%20defines%20the%20main%20motivation%20and%20strategy%20of%20our%20approach%2C%20we%20treat%20several%20cases%20by%20focusing%20first%20on%20a%20specific%20issue%20of%20generic%20character%20%28the%20origin%20of%20the%20first%20sharp%20diffraction%20peak%20in%20the%20neutron%20structure%20factor%20as%20a%20signature%20of%20intermediate-range%20order%29%20and%20then%20on%20particular%20systems%20mostly%20within%20%28or%20closely%20related%20to%29%20the%20GexSe1%3Fx%20binary%20family.%20Chalcogenide%20ternary%20glasses%20will%20also%20be%20considered.%20It%20appears%20that%20FPMD%20has%20acquired%20an%20unprecedented%20level%20of%20reliability%20in%20the%20field%20of%20glassy%20chalcogenides%2C%20enriching%20and%20complementing%20successfully%20understanding%20of%20these%20materials.%22%2C%22date%22%3A%222020%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1088%5C%2F1361-6463%5C%2Fab48a4%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1088%5C%2F1361-6463%5C%2Fab48a4%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220022-3727%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-04-30T14%3A42%3A31Z%22%7D%7D%2C%7B%22key%22%3A%22LM5QYM9Z%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Massobrio%20et%20al.%22%2C%22parsedDate%22%3A%222020%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EC.%20Massobrio%2C%20M.%20Boero%2C%20S.%20Le%20Roux%2C%20G.%20Ori%2C%20A.%20Bouzid%2C%20E.%20Martin%2C%20Making%20Computer%20Materials%20Real%3A%20The%20Predictive%20Power%20of%20First-Principles%20Molecular%20Dynamics%2C%20in%3A%20E.%20Levchenko%2C%20Y.%20Dappe%2C%20G.%20Ori%20%28Eds.%29%2C%20THEORY%20AND%20SIMULATION%20IN%20PHYSICS%20FOR%20MATERIALS%20APPLICATIONS%3A%20CUTTING-EDGE%20TECHNIQUES%20IN%20THEORETICAL%20AND%20COMPUTATIONAL%20MATERIALS%20SCIENCE%20%5C%2F%20SCIENCE%20%5C%2F%20Edited%20by%20E.V.%20Levchenko...%2C%202020%3A%20pp.%203%26%23x2013%3B21.%20%3Ca%20class%3D%27zp-ItemURL%27%20href%3D%2710.1007%5C%2F978-3-030-37790-8_1%27%3E10.1007%5C%2F978-3-030-37790-8_1%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22bookSection%22%2C%22title%22%3A%22Making%20Computer%20Materials%20Real%3A%20The%20Predictive%20Power%20of%20First-Principles%20Molecular%20Dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%2C%7B%22creatorType%22%3A%22editor%22%2C%22firstName%22%3A%22EV%22%2C%22lastName%22%3A%22Levchenko%22%7D%2C%7B%22creatorType%22%3A%22editor%22%2C%22firstName%22%3A%22YJ%22%2C%22lastName%22%3A%22Dappe%22%7D%2C%7B%22creatorType%22%3A%22editor%22%2C%22firstName%22%3A%22G%22%2C%22lastName%22%3A%22Ori%22%7D%5D%2C%22abstractNote%22%3A%22First-principles%20molecular%20dynamics%20%28FPMD%29%20is%20awell-established%20method%20to%20study%20materials%20at%20the%20atomic%20scale%20by%20taking%20advantage%20of%20three%20ingredients%3A%20the%20laws%20of%20statistical%20mechanics%2C%20the%20theoretical%20foundations%20of%20density%20functional%20theory%20and%20powerful%20computers.%20FPMDdoes%20its%20bestwhen%20the%20atomic%20structures%20are%20unknown%20or%20poorly%20known%20and%20when%20their%20time%20trajectories%20are%20required%20to%20extract%2C%20via%20statistical%20averages%2C%20a%20thermodynamical%20evolution%20as%20a%20function%20of%20temperature.%20In%20this%20paper%2C%20key%20concepts%20of%20molecular%20dynamics%20are%20recalled%20and%20made%20simple%2C%20by%20insisting%20on%20the%20proper%20use%20of%20some%20definitions%20and%20by%20showing%2C%20via%20a%20prototypical%20example%2C%20the%20sensitivity%20to%20a%20crucial%20part%20%28the%20exchange-correlation%20energy%29%20of%20the%20total%20energy%20functional.%22%2C%22bookTitle%22%3A%22THEORY%20AND%20SIMULATION%20IN%20PHYSICS%20FOR%20MATERIALS%20APPLICATIONS%3A%20CUTTING-EDGE%20TECHNIQUES%20IN%20THEORETICAL%20AND%20COMPUTATIONAL%20MATERIALS%20SCIENCE%20%5C%2F%20SCIENCE%20%5C%2F%20edited%20by%20E.V.%20Levchenko...%22%2C%22date%22%3A%222020%22%2C%22originalDate%22%3A%22%22%2C%22originalPublisher%22%3A%22%22%2C%22originalPlace%22%3A%22%22%2C%22format%22%3A%22%22%2C%22ISBN%22%3A%220933-033X%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%2210.1007%5C%2F978-3-030-37790-8_1%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222022-03-29T13%3A17%3A18Z%22%7D%7D%2C%7B%22key%22%3A%224UAQA6RX%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Mishima%20et%20al.%22%2C%22parsedDate%22%3A%222020%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Mishima%2C%20M.%20Shoji%2C%20Y.%20Umena%2C%20M.%20Boero%2C%20Y.%20Shigeta%2C%20Role%20of%20the%20Propionic%20Acid%20Side-Chain%20of%20C-Phycocyanin%20Chromophores%20in%20the%20Excited%20States%20for%20the%20Photosynthesis%20Process%2C%20Bulletin%20of%20the%20Chemical%20Society%20of%20Japan%2093%20%282020%29%201509%26%23x2013%3B1519.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1246%5C%2Fbcsj.20200187%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1246%5C%2Fbcsj.20200187%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Role%20of%20the%20Propionic%20Acid%20Side-Chain%20of%20C-Phycocyanin%20Chromophores%20in%20the%20Excited%20States%20for%20the%20Photosynthesis%20Process%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenji%22%2C%22lastName%22%3A%22Mishima%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mitsuo%22%2C%22lastName%22%3A%22Shoji%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yasufumi%22%2C%22lastName%22%3A%22Umena%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yasuteru%22%2C%22lastName%22%3A%22Shigeta%22%7D%5D%2C%22abstractNote%22%3A%22This%20paper%20focuses%20on%20a%20theoretical%20investigation%20of%20the%20peculiar%20properties%20of%20the%20chromophore%20in%20the%20C-phycocyanin%20%28C-PC%29%2C%20phycocyanobilin%20%28PCB%29.%20The%20scope%20is%20to%20unravel%20their%20key%20features%20upon%20light%20absorption%20and%20transmission%20occurring%20in%20natural%20photosynthesis.%20To%20this%20aim%2C%20by%20resorting%20to%20the%20time-dependent%20density%20functional%20theory%20%28TDDFT%29%20and%20natural%20bond%20orbital%20%28NBO%29%20methods%2C%20we%20compute%20the%20photoabsorption%20spectra%20and%20electronic%20properties%20of%20PCB%2C%20showing%20that%20three%20different%20orientations%20of%20the%20PCB%20in%20C-PC%20contribute%20to%20the%20nonhomogeneous%20broadening%20of%20the%20entire%20photoabsorption%20spectrum%20of%20C-PC.%20Furthermore%2C%20the%20photoabsorption%20peaks%20of%20PCB%20can%20undergo%20a%20shift%20up%20to%2040%20nm%20because%20of%20solvation%20effects.%20Further%20investigations%20on%20the%20competitive%20influence%20of%20the%20nearby%20aspartate%20residue%20and%20two%20propionic%20acids%20on%20the%20absorption%20spectra%20show%20that%20the%20latter%20play%20a%20significant%20role%20in%20realizing%20the%20different%20photo-response%20among%20the%20three%20isomers%20of%20PCB.%20In%20the%20low-lying%20electronic%20excited%20states%2C%20the%20pi%20conjugated%20C-C%20bonds%20and%20the%20twisting%20angle%20of%20the%20pyrrole%20rings%20turn%20out%20to%20be%20affected.%20The%20NBO%20geometrical%20analyses%20of%20the%20bond%20lengths%2C%20interatomic%20angles%2C%20and%20dihedral%20angles%20evidenced%20that%20the%20intermolecular%20interactions%20of%20the%20propionic%20acid%20side%20chains%20play%20a%20crucial%20role%20in%20the%20determination%20of%20the%20excited%20state%20molecular%20conformations.%20These%20results%20indicate%20that%20the%20absorption%20spectra%20and%20the%20excited%20state%20structures%20of%20PCB%20are%20efficiently%20tuned%20during%20natural%20photosynthesis%20processes.%22%2C%22date%22%3A%222020%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1246%5C%2Fbcsj.20200187%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1246%5C%2Fbcsj.20200187%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220009-2673%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222021-02-17T16%3A15%3A08Z%22%7D%7D%2C%7B%22key%22%3A%22XMFUIBWY%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Oshiyama%20et%20al.%22%2C%22parsedDate%22%3A%222020%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EA.%20Oshiyama%2C%20K.M.%20Bui%2C%20M.%20Boero%2C%20Y.%20Kangawa%2C%20K.%20Shiraishi%2C%20Computics%20Approach%20toward%20Clarification%20of%20Atomic%20Reactions%20during%20Epitaxial%20Growth%20of%20GaN%2C%20in%3A%202020%20INTERNATIONAL%20CONFERENCE%20ON%20SIMULATION%20OF%20SEMICONDUCTOR%20PROCESSES%20AND%20DEVICES%20%28SISPAD%202020%29%2C%20IEEE%2C%202020%3A%20pp.%2011%26%23x2013%3B14.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22conferencePaper%22%2C%22title%22%3A%22Computics%20Approach%20toward%20Clarification%20of%20Atomic%20Reactions%20during%20Epitaxial%20Growth%20of%20GaN%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Atsushi%22%2C%22lastName%22%3A%22Oshiyama%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kieu%20My%22%2C%22lastName%22%3A%22Bui%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yoshihiro%22%2C%22lastName%22%3A%22Kangawa%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenji%22%2C%22lastName%22%3A%22Shiraishi%22%7D%5D%2C%22abstractNote%22%3A%22We%20report%20first-principles%20calculations%20based%20on%20the%20density-functional%20theory%20that%20clarify%20atomic%20reactions%20of%20ammonia%20decomposition%20and%20subsequent%20nitrogen%20incorporation%20during%20GaN%20epitaxial%20growth.%20We%20find%20that%20Ga-Ga%20weak%20bonds%20are%20ubiquitous%20on%20Ga-rich%20growing%20surface%20and%20responsible%20for%20the%20growth%20reactions.%20Furthermore%2C%20Car-Parrinello%20Molecular%20Dynamics%20simulations%20predict%20the%20existence%20of%202-%20dimensional%20Ga%20liquid%20phase%2C%20providing%20new%20insight%20into%20the%20epitaxial%20growth.%20The%20obtained%20results%20are%20expected%20to%20become%20basics%20for%20multi-scale%20growth%20simulations%20in%20future.%22%2C%22proceedingsTitle%22%3A%222020%20INTERNATIONAL%20CONFERENCE%20ON%20SIMULATION%20OF%20SEMICONDUCTOR%20PROCESSES%20AND%20DEVICES%20%28SISPAD%202020%29%22%2C%22conferenceName%22%3A%22International%20Conference%20on%20Simulation%20of%20Semiconductor%20Processes%20and%20Devices%20%28SISPAD%29%2C%20ELECTR%20NETWORK%2C%2023%20septembre-%2006%20octobre%2C%202020%22%2C%22date%22%3A%222020%22%2C%22eventPlace%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22ISBN%22%3A%22978-4-86348-763-5%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222022-01-13T08%3A09%3A24Z%22%7D%7D%2C%7B%22key%22%3A%225ZQJXT6U%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Shoji%20et%20al.%22%2C%22parsedDate%22%3A%222020%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Shoji%2C%20Y.%20Abe%2C%20M.%20Boero%2C%20Y.%20Shigeta%2C%20Y.%20Nishiya%2C%20Reaction%20mechanism%20of%20N-cyclopropylglycine%20oxidation%20by%20monomeric%20sarcosine%20oxidase%2C%20Physical%20Chemistry%20Chemical%20Physics%2022%20%282020%29%2016552%26%23x2013%3B16561.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd0cp01679a%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd0cp01679a%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Reaction%20mechanism%20of%20N-cyclopropylglycine%20oxidation%20by%20monomeric%20sarcosine%20oxidase%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mitsuo%22%2C%22lastName%22%3A%22Shoji%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yukihiro%22%2C%22lastName%22%3A%22Abe%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yasuteru%22%2C%22lastName%22%3A%22Shigeta%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yoshiaki%22%2C%22lastName%22%3A%22Nishiya%22%7D%5D%2C%22abstractNote%22%3A%22Monomeric%20sarcosine%20oxidase%20%28MSOX%29%20is%20a%20fundamental%20-%20yet%20one%20of%20the%20simplest%20-%20member%20of%20a%20family%20of%20flavoenzymes%20able%20to%20catalyze%20the%20oxidation%20of%20sarcosine%20%28N-methylglycine%29%20and%20other%20secondary%20amines.%20MSOX%20is%20one%20of%20the%20best%20characterized%20members%20of%20the%20amine%20oxidoreductases%20%28AOs%29%2C%20however%2C%20its%20reaction%20mechanism%20is%20still%20controversial.%20A%20single%20electron%20transfer%20%28SET%29%20process%20was%20suggested%20on%20the%20basis%20of%20studies%20with%20N-cyclopropylglycine%20%28CPG%29%2C%20although%20a%20hydride%20transfer%20mechanism%20would%20be%20more%20consistent%20in%20general%20for%20AOs.%20To%20shed%20some%20light%20on%20the%20detailed%20reaction%20mechanisms%20of%20CPG%20in%20MSOX%2C%20we%20performed%20hybrid%20quantum%20mechanical%5C%2Fmolecular%20mechanical%20%28QM%5C%2FMM%29%20simulations.%20We%20found%20that%20the%20polar%20mechanism%20is%20energetically%20the%20most%20favorable.%20The%20free%20energy%20profile%20indicates%20that%20the%20first%20rate-limiting%20step%20is%20the%20CPG%20binding%20to%20the%20flavin%20ring%20which%20simultaneously%20proceeds%20with%20the%20ring-opening%20of%20the%20CPG%20cyclopropyl%20group.%20This%20reaction%20step%20of%20the%20CPG%20adduct%20formation%20corresponds%20to%20the%20nucleophilic%20attack%20of%20the%20cyclopropyl%20group%20%28C3%20atom%29%20to%20the%20flavin%20ring%20%28C4a%20atom%29%2C%20whereas%20the%20expected%20radical%20species%20formation%20in%20the%20SET%20mechanism%20was%20not%20observed.%20The%20following%20inactivated%20species%2C%20which%20accumulates%20during%20the%20CPG%20oxidation%20in%20MSOX%2C%20can%20be%20ascribed%20to%20an%20imine%20state%2C%20and%20not%20an%20enamine%20state%2C%20on%20the%20basis%20of%20the%20computed%20UV%5C%2FVis%20spectra.%20The%20conformation%20of%20CPG%20was%20found%20to%20be%20crucial%20for%20reactions%20following%20the%20CPG%20adduct%20formation.%22%2C%22date%22%3A%222020%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fd0cp01679a%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fd0cp01679a%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221463-9076%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222020-09-18T13%3A50%3A20Z%22%7D%7D%2C%7B%22key%22%3A%226K2YRICV%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Shoji%20et%20al.%22%2C%22parsedDate%22%3A%222020%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Shoji%2C%20T.%20Murakawa%2C%20M.%20Boero%2C%20Y.%20Shigeta%2C%20H.%20Hayashi%2C%20T.%20Okajima%2C%20Unique%20protonation%20states%20of%20aspartate%20and%20topaquinone%20in%20the%20active%20site%20of%20copper%20amine%20oxidase%2C%20RSC%20Advances%2010%20%282020%29%2038631%26%23x2013%3B38639.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd0ra06365g%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fd0ra06365g%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Unique%20protonation%20states%20of%20aspartate%20and%20topaquinone%20in%20the%20active%20site%20of%20copper%20amine%20oxidase%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mitsuo%22%2C%22lastName%22%3A%22Shoji%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Takeshi%22%2C%22lastName%22%3A%22Murakawa%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yasuteru%22%2C%22lastName%22%3A%22Shigeta%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hideyuki%22%2C%22lastName%22%3A%22Hayashi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Toshihide%22%2C%22lastName%22%3A%22Okajima%22%7D%5D%2C%22abstractNote%22%3A%22The%20oxidative%20deamination%20of%20biogenic%20amines%2C%20crucial%20in%20the%20metabolism%20of%20a%20wealth%20of%20living%20organisms%2C%20is%20catalyzed%20by%20copper%20amine%20oxidases%20%28CAOs%29.%20In%20this%20work%2C%20on%20the%20ground%20of%20accurate%20molecular%20modeling%2C%20we%20provide%20a%20clear%20insight%20into%20the%20unique%20protonation%20states%20of%20the%20key%20catalytic%20aspartate%20residue%20Asp298%20and%20the%20prosthetic%20group%20of%20topaquinone%20%28TPQ%29%20in%20the%20CAO%20of%20Arthrobacter%20globiformis%20%28AGAO%29.%20This%20provides%20both%20extensions%20and%20complementary%20information%20to%20the%20crystal%20structure%20determined%20by%20our%20recent%20neutron%20diffraction%20%28ND%29%20experiment.%20The%20hybrid%20quantum%20mechanics%5C%2Fmolecular%20mechanics%20%28QM%5C%2FMM%29%20simulations%20suggest%20that%20the%20ND%20structure%20closely%20resembles%20a%20state%20in%20which%20Asp298%20is%20protonated%20and%20the%20TPQ%20takes%20an%20enolate%20form.%20The%20TPQ%20keto%20form%20can%20coexist%20in%20the%20fully%20protonated%20state.%20The%20energetic%20and%20structural%20analyses%20indicate%20that%20the%20active%20site%20structure%20of%20the%20AGAO%20crystal%20is%20not%20a%20single%20state%20but%20rather%20a%20mixture%20of%20the%20different%20protonation%20and%20conformational%20states%20identified%20in%20this%20work.%22%2C%22date%22%3A%222020%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fd0ra06365g%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fd0ra06365g%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222021-02-17T16%3A27%3A02Z%22%7D%7D%2C%7B%22key%22%3A%22YCPXK4HF%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Silvestrelli%20et%20al.%22%2C%22parsedDate%22%3A%222020%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EP.L.%20Silvestrelli%2C%20E.%20Martin%2C%20M.%20Boero%2C%20A.%20Bouzid%2C%20G.%20Ori%2C%20C.%20Massobrio%2C%20Atomic%20Structure%20of%20Glassy%20GeTe4%20as%20a%20Playground%20to%20Assess%20the%20Performances%20of%20Density%20Functional%20Schemes%20Accounting%20for%20Dispersion%20Forces%2C%20Journal%20of%20Physical%20Chemistry%20B%20124%20%282020%29%2011273%26%23x2013%3B11279.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpcb.0c08628%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpcb.0c08628%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Atomic%20Structure%20of%20Glassy%20GeTe4%20as%20a%20Playground%20to%20Assess%20the%20Performances%20of%20Density%20Functional%20Schemes%20Accounting%20for%20Dispersion%20Forces%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Pier%20Luigi%22%2C%22lastName%22%3A%22Silvestrelli%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22The%20atomic%20structure%20of%20glassy%20GeTe4%20is%20obtained%20in%20the%20framework%20of%20first-principles%20molecular%20dynamics%20%28FPMD%29%20by%20considering%20five%20different%20approaches%20for%20the%20description%20of%20the%20electronic%20structure%20within%20density%20functional%20theory%20%28DFT%29.%20Among%20these%20schemes%2C%20one%20is%20not%20corrected%20by%20accounting%20for%20the%20dispersion%20forces%20and%20it%20is%20based%20on%20the%20BLYP%20exchange-correlation%20%28XC%29%20functional%2C%20while%20all%20of%20the%20others%20consider%20the%20dispersion%20forces%20according%20to%20different%20theoretical%20strategies.%20In%20particular%2C%20by%20maintaining%20the%20BLYP%20expression%20for%20the%20XC%20functional%2C%20two%20of%20them%20%28BLYP-D2%20and%20BLYP-D3%29%20exploit%20the%20Grimme%20expressions%20for%20the%20dispersion%20forces%2C%20while%20the%20fourth%20scheme%20is%20based%20on%20the%20maximally%20localized%20Wannier%20functions%20%28MLWFs%29.%20Finally%2C%20we%20also%20considered%20the%20rVVIO%20functional%20constructed%20to%20include%20seamlessly%20the%20dispersion%20part.%20Our%20results%20point%20out%20the%20better%20performances%20of%20BLYP-D3%20and%20MLWF%20in%20terms%20of%20comparison%20with%20experimental%20data%20for%20the%20total%20pair%20correlation%20functions%2C%20with%20BLYP-D2%20and%20rVVIO%20being%20closer%20to%20the%20uncorrected%20BLYP%20data.%20The%20implications%20of%20such%20findings%20are%20discussed%20by%20considering%20the%20overall%20limited%20impact%20of%20dispersion%20forces%20on%20the%20atomic%20structure%20of%20glassy%20GeTe4.%22%2C%22date%22%3A%222020%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Facs.jpcb.0c08628%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Facs.jpcb.0c08628%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221520-6106%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222021-02-17T16%3A27%3A29Z%22%7D%7D%5D%7D
[1]
K.M. Bui, M. Boero, K. Shiraishi, A. Oshiyama, A two-dimensional liquid-like phase on Ga-rich GaN (0001) surfaces evidenced by first principles molecular dynamics, in: Japanese Journal of Applied Physics, 2020: p. SGGK04.
https://doi.org/10.7567/1347-4065/ab650b.
[1]
T.-Q. Duong, C. Massobrio, G. Ori, M. Boero, E. Martin, Thermal resistance of an interfacial molecular layer by first-principles molecular dynamics, Journal of Chemical Physics 153 (2020) 074704.
https://doi.org/10.1063/5.0014232.
[1]
T.-Q. Duong, C. Massobrio, M. Boero, G. Ori, E. Martin, Heat transport in disordered network forming materials: Size effects and existence of propagative modes, Computational Materials Science 177 (2020) 109607.
https://doi.org/10.1016/j.commatsci.2020.109607.
[1]
I.B.A. Essomba, C. Massobrio, M. Boero, G. Ori, Assessing the Versatility of Molecular Modelling as a Strategy for Predicting Gas Adsorption Properties of Chalcogels, in: E. Levchenko, Y. Dappe, G. Ori (Eds.), THEORY AND SIMULATION IN PHYSICS FOR MATERIALS APPLICATIONS: CUTTING-EDGE TECHNIQUES IN THEORETICAL AND COMPUTATIONAL MATERIALS SCIENCE / Edited by E.V. Levchenko..., 2020: pp. 23–37.
10.1007/978-3-030-37790-8_2.
[1]
E.V. Levchenko, Y.J. Dappe, G. Ori, Preface, in: E. Levchenko, Y. Dappe, G. Ori (Eds.), THEORY AND SIMULATION IN PHYSICS FOR MATERIALS APPLICATIONS: CUTTING-EDGE TECHNIQUES IN THEORETICAL AND COMPUTATIONAL MATERIALS SCIENCE / Edited by Y.J. Dappe et G. Ori, 2020: p. V–V.
10.1007/978-3-030-37790-8.
[1]
C. Massobrio, A. Bouzid, M. Boero, G. Ori, E. Martin, S. Le Roux, Chalcogenide glasses for innovation in applied science: fundamental issues and new insights, Journal of Physics D-Applied Physics 53 (2020) 033002.
https://doi.org/10.1088/1361-6463/ab48a4.
[1]
C. Massobrio, M. Boero, S. Le Roux, G. Ori, A. Bouzid, E. Martin, Making Computer Materials Real: The Predictive Power of First-Principles Molecular Dynamics, in: E. Levchenko, Y. Dappe, G. Ori (Eds.), THEORY AND SIMULATION IN PHYSICS FOR MATERIALS APPLICATIONS: CUTTING-EDGE TECHNIQUES IN THEORETICAL AND COMPUTATIONAL MATERIALS SCIENCE / SCIENCE / Edited by E.V. Levchenko..., 2020: pp. 3–21.
10.1007/978-3-030-37790-8_1.
[1]
K. Mishima, M. Shoji, Y. Umena, M. Boero, Y. Shigeta, Role of the Propionic Acid Side-Chain of C-Phycocyanin Chromophores in the Excited States for the Photosynthesis Process, Bulletin of the Chemical Society of Japan 93 (2020) 1509–1519.
https://doi.org/10.1246/bcsj.20200187.
[1]
A. Oshiyama, K.M. Bui, M. Boero, Y. Kangawa, K. Shiraishi, Computics Approach toward Clarification of Atomic Reactions during Epitaxial Growth of GaN, in: 2020 INTERNATIONAL CONFERENCE ON SIMULATION OF SEMICONDUCTOR PROCESSES AND DEVICES (SISPAD 2020), IEEE, 2020: pp. 11–14.
[1]
M. Shoji, Y. Abe, M. Boero, Y. Shigeta, Y. Nishiya, Reaction mechanism of N-cyclopropylglycine oxidation by monomeric sarcosine oxidase, Physical Chemistry Chemical Physics 22 (2020) 16552–16561.
https://doi.org/10.1039/d0cp01679a.
[1]
M. Shoji, T. Murakawa, M. Boero, Y. Shigeta, H. Hayashi, T. Okajima, Unique protonation states of aspartate and topaquinone in the active site of copper amine oxidase, RSC Advances 10 (2020) 38631–38639.
https://doi.org/10.1039/d0ra06365g.
[1]
P.L. Silvestrelli, E. Martin, M. Boero, A. Bouzid, G. Ori, C. Massobrio, Atomic Structure of Glassy GeTe4 as a Playground to Assess the Performances of Density Functional Schemes Accounting for Dispersion Forces, Journal of Physical Chemistry B 124 (2020) 11273–11279.
https://doi.org/10.1021/acs.jpcb.0c08628.
1839302
NZSFH59F
2019
1
surface-science-reports
50
creator
asc
year
7054
https://www.ipcms.unistra.fr/plugins/zotpress/
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3Afalse%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%22ZJMJ7R53%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Barhoumi%20et%20al.%22%2C%22parsedDate%22%3A%222019%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ER.%20Barhoumi%2C%20A.%20Amokrane%2C%20S.%20Klyatskaya%2C%20M.%20Boero%2C%20M.%20Ruben%2C%20J.-P.%20Bucher%2C%20Screening%20the%204f-electron%20spin%20of%20TbPc2%20single-molecule%20magnets%20on%20metal%20substrates%20by%20ligand%20channeling%2C%20Nanoscale%2011%20%282019%29%2021167%26%23x2013%3B21179.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc9nr05873g%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc9nr05873g%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Screening%20the%204f-electron%20spin%20of%20TbPc2%20single-molecule%20magnets%20on%20metal%20substrates%20by%20ligand%20channeling%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Rabei%22%2C%22lastName%22%3A%22Barhoumi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Anis%22%2C%22lastName%22%3A%22Amokrane%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Svetlana%22%2C%22lastName%22%3A%22Klyatskaya%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mario%22%2C%22lastName%22%3A%22Ruben%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jean-Pierre%22%2C%22lastName%22%3A%22Bucher%22%7D%5D%2C%22abstractNote%22%3A%22Bis%28phthalocyaninato%29lanthanide%20%28LnPc%282%29%29%20double-decker-based%20devices%20have%20recently%20attracted%20a%20great%20deal%20of%20interest%20for%20data%20encoding%20purposes.%20Although%20the%204f-electrons%20of%20lanthanide%20ions%20play%20a%20key%20role%20in%20the%20experimental%20methodology%2C%20their%20localized%20character%2C%20deeper%20in%20energy%20compared%20to%20the%203d%20electrons%20of%20transition%20metals%2C%20hampers%20a%20detailed%20investigation.%20Here%2C%20our%20approach%20consists%20of%20the%20follow-up%20of%20the%20entanglement%20process%20with%20other%20molecules%20and%20with%20the%20substrate%20electrons%20by%20means%20of%20space-resolved%20detection%20of%20the%20Kondo%20resonance%20by%20scanning%20tunneling%20spectroscopy%20%28STS%29%2C%20using%20different%20substrates%20%28from%20weak%20to%20strong%20interaction%29.%20It%20is%20found%20that%20TbPc2%20molecules%20firstly%20interact%20with%20their%20environment%20by%20means%20of%20the%20pi-radicals%20of%20the%20ligand.%20The%20radical%20spin%20of%20TbPc2%20can%20be%20identified%20by%20STS%20on%20a%20weakly%20interacting%20substrate%20such%20as%20Au%28111%29.%20In%20the%20case%20of%20a%20Ag%28111%29%20substrate%2C%20we%20are%20able%20to%20analyze%20the%20effect%20of%20an%20electron%20transfer%20on%20the%20molecule%20%28pairing-up%20of%20the%20radical%20spin%29%20and%20the%20subsequent%20quenching%20of%20the%20Kondo%20resonance.%20Finally%2C%20on%20a%20strongly%20interacting%20substrate%20such%20as%20Cu%28111%29%2C%20a%20significant%20rearrangement%20of%20electrons%20takes%20place%20and%20the%20Kondo%20screening%20of%20the%204f%20electrons%20of%20the%20Tb%20ion%20of%20TbPc2%20is%20observed.%20By%20comparative%20STS%20measurements%20on%20YPc2%2C%20that%20has%20empty%204d%20and%204f%20shells%2C%20we%20prove%20that%20the%20Kondo%20resonance%20measured%20in%20the%20center%20of%20the%20TbPc2%20molecule%20indeed%20stems%20from%20the%204f-electrons.%20At%20the%20same%20time%2C%20we%20provide%20evidence%20for%20the%20hybridization%20of%20the%204f%20states%20with%20the%20pi%20electron.%22%2C%22date%22%3A%222019%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fc9nr05873g%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fc9nr05873g%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222040-3364%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22RZX792Q8%22%5D%2C%22dateModified%22%3A%222020-02-03T10%3A32%3A04Z%22%7D%7D%2C%7B%22key%22%3A%22MRCTSDWE%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Duong%20et%20al.%22%2C%22parsedDate%22%3A%222019%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ET.-Q.%20Duong%2C%20C.%20Massobrio%2C%20G.%20Ori%2C%20M.%20Boero%2C%20E.%20Martin%2C%20Thermal%20conductivity%20and%20transport%20modes%20in%20glassy%20GeTe4%20by%20first-principles%20molecular%20dynamics%2C%20Physical%20Review%20Materials%203%20%282019%29%20105401.%20%3Ca%20class%3D%27zp-ItemURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevMaterials.3.105401%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevMaterials.3.105401%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Thermal%20conductivity%20and%20transport%20modes%20in%20glassy%20GeTe4%20by%20first-principles%20molecular%20dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Thuy-Quynh%22%2C%22lastName%22%3A%22Duong%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%5D%2C%22abstractNote%22%3A%22First-principles%20molecular%20dynamics%20is%20employed%20to%20investigate%20thermal%20transport%20in%20glassy%20GeTe4%2C%20a%20subsystem%20of%20several%20ternary%20phase-change%20materials.%20As%20a%20first%20result%2C%20we%20found%20modes%20localized%20on%20a%20few%20atoms%20in%20the%20vibrational%20density%20of%20states.%20The%20thermal%20transport%20is%20further%20rationalized%20by%20calculating%20the%20thermal%20conductivity%20for%20a%20range%20of%20system%20sizes%20and%20shapes%20via%20the%20approach-to-equilibrium%20methodology.%20By%20considering%20the%20length%20dependence%20of%20the%20thermal%20conductivity%2C%20we%20provide%20evidence%20of%20propagative%20modes%20with%20mean%20free%20paths%20as%20long%20as%206%20nm%2C%20i.e.%2C%20well%20beyond%20short-range%20order%20distances.%20Extrapolation%20of%20our%20bulk%20thermal%20conductivity%20to%20macroscopic%20sizes%20is%20in%20full%20agreement%20with%20the%20experimental%20values.%20Finally%2C%20we%20assess%20phenomenological%20models%20developed%20for%20the%20thermal%20conductivity%20of%20disordered%20materials%20by%20enriching%20their%20intrinsic%20significance%20via%20the%20insertion%20in%20their%20analytical%20expression%20of%20values%20obtained%20via%20first-principles%20molecular%20dynamics.%22%2C%22date%22%3A%222019%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevMaterials.3.105401%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevMaterials.3.105401%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222475-9953%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222019-12-18T15%3A47%3A26Z%22%7D%7D%2C%7B%22key%22%3A%22UIHIFDWN%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Hatakeyama%20et%20al.%22%2C%22parsedDate%22%3A%222019%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Hatakeyama%2C%20K.%20Koizurni%2C%20M.%20Boero%2C%20K.%20Nobusada%2C%20H.%20Hori%2C%20T.%20Misonou%2C%20T.%20Kobayashi%2C%20S.%20Nakamura%2C%20Unique%20Structural%20Relaxations%20and%20Molecular%20Conformations%20of%20Porphyra-334%20at%20the%20Excited%20State%2C%20Journal%20of%20Physical%20Chemistry%20B%20123%20%282019%29%207649%26%23x2013%3B7656.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpcb.9b03744%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpcb.9b03744%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Unique%20Structural%20Relaxations%20and%20Molecular%20Conformations%20of%20Porphyra-334%20at%20the%20Excited%20State%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Makoto%22%2C%22lastName%22%3A%22Hatakeyama%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenichi%22%2C%22lastName%22%3A%22Koizurni%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Katsuyuki%22%2C%22lastName%22%3A%22Nobusada%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hirokazu%22%2C%22lastName%22%3A%22Hori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Taku%22%2C%22lastName%22%3A%22Misonou%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Takao%22%2C%22lastName%22%3A%22Kobayashi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Shinichiro%22%2C%22lastName%22%3A%22Nakamura%22%7D%5D%2C%22abstractNote%22%3A%22Quantum%20chemistry%20based%20simulations%20were%20used%20to%20examine%20the%20excited%20state%20of%20porphyra-334%2C%20one%20of%20the%20fundamental%20mycosporine-like%20amino%20acids%20present%20in%20a%20wide%20variety%20of%20aqueous%20organisms.%20Our%20calculations%20reveal%20three%20characteristic%20aspects%20of%20porphyra-334%20related%20to%20either%20its%20ground%20or%20excited%20state.%20Specifically%2C%20%28i%29%20the%20ground%20state%20%28S-0%29%20structure%20consists%20of%20a%20planar%20geometry%20in%20which%20three%20units%20can%20be%20identified%2C%20the%20central%20cyclohexene%20ring%2C%20the%20glycine%20branch%2C%20and%20the%20threonine%20branch%2C%20reflecting%20the%20pi%20conjugation%20of%20the%20system%3B%20%28ii%29%20the%20first%20singlet%20excited%20state%20%28S-1%29%20shows%20a%20large%20oscillator%20strength%20and%20a%20typical%20pi%20pi%2A%20excitation%20character%3B%20and%20%28iii%29%20upon%20relaxation%20at%20S-1%2C%20the%20originally%20ground%20state%20planar%20structure%20undergoes%20a%20relaxation%20to%20a%20nonplanar%20one%2C%20S-1%2C%20especially%20at%20the%20carbon-nitrogen%20%28CN%29%20groups%20linking%20the%20cyclohexene%20ring%20to%20the%20glycine%20or%20threonine%20arm.%20The%20induced%20nonplanarity%20can%20be%20ascribed%20to%20the%20fact%20that%20the%20carbon%20atoms%20of%20the%20CN%20groups%20prefer%20an%20sp%283%29%20hybridization%20in%20the%20S-1%20state.%20At%20the%20singlet%20state%2C%20these%20processes%20are%20unlikely%20to%20be%20trapped%20by%20singlet-triplet%20intersystem%20crossing%20especially%20when%20these%20occur%20in%20the%20hydrophilic%20zwitter-ion%20forms%20of%20porphyra-334.%20These%20results%20provide%20the%20missing%20information%20for%20thorough%20interpretation%20of%20the%20stability%20of%20porphyra-334%20upon%20UV%20irradiation.%22%2C%22date%22%3A%222019%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Facs.jpcb.9b03744%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Facs.jpcb.9b03744%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221520-6106%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222020-02-03T08%3A25%3A37Z%22%7D%7D%2C%7B%22key%22%3A%22JJ7MBAE2%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Koizumi%20et%20al.%22%2C%22parsedDate%22%3A%222019%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Koizumi%2C%20K.%20Nobusada%2C%20M.%20Boero%2C%20Hydrogen%20storage%20mechanism%20and%20diffusion%20in%20metal-organic%20frameworks%2C%20Physical%20Chemistry%20Chemical%20Physics%2021%20%282019%29%207756%26%23x2013%3B7764.%20%3Ca%20class%3D%27zp-ItemURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc8cp07467d%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc8cp07467d%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Hydrogen%20storage%20mechanism%20and%20diffusion%20in%20metal-organic%20frameworks%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenichi%22%2C%22lastName%22%3A%22Koizumi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Katsuyuki%22%2C%22lastName%22%3A%22Nobusada%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%222019%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fc8cp07467d%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc8cp07467d%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221463-9076%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222019-06-11T09%3A10%3A06Z%22%7D%7D%2C%7B%22key%22%3A%224UG6GAKV%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Ori%20et%20al.%22%2C%22parsedDate%22%3A%222019%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EG.%20Ori%2C%20A.%20Bouzid%2C%20E.%20Martin%2C%20C.%20Massobrio%2C%20S.%20Le%20Roux%2C%20M.%20Boero%2C%20Chalcogenide%20glasses%20as%20a%20playground%20for%20the%20application%20of%20first-principles%20molecular%20dynamics%20to%20disordered%20materials%2C%20Solid%20State%20Sciences%2095%20%282019%29%20105925.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.solidstatesciences.2019.06.014%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.solidstatesciences.2019.06.014%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Chalcogenide%20glasses%20as%20a%20playground%20for%20the%20application%20of%20first-principles%20molecular%20dynamics%20to%20disordered%20materials%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22An%20overview%20of%20the%20major%20first-principles%20methods%20used%20to%20simulate%20condensed%20phases%20is%20presented%2C%20with%20special%20emphasis%20on%20chalcogenide%20glasses.%20The%20scope%20of%20this%20review%20article%20is%20to%20offer%20a%20survey%20of%20fundamental%20algorithms%20and%20techniques%2C%20accompanied%20by%20a%20few%20recent%20examples%20particularly%20representative%20of%20computational%20materials%20science%20applied%20to%20disordered%20chalcogenide%20phases.%20Special%20attention%20is%20devoted%20to%20the%20inclusion%20of%20long-range%20van%20der%20Waals%20dispersion%20forces%2C%20treatment%20of%20the%20exact%20exchange%2C%20dynamical%20simulations%20and%20extraction%20of%20optical%20and%20dielectric%20properties.%20Machine%20learning%20techniques%20are%20introduced%20as%20recent%20forefront%20applications%20of%20first-principle%20methods.%20In%20this%20latter%20case%2C%20accurate%20quantum-mechanics%20based%20simulations%20are%20crucial%20to%20generate%20a%20data%20base%20exploited%20by%20neuronal-network%20type%20algorithms%20to%20create%20accurate%20interatomic%20potentials%20%28force%20fields%29%20allowing%20for%20large%20and%20long-lasting%20simulations%20of%20realistic%20disordered%20materials.%20The%20atomic-level%20knowledge%20provided%20by%20the%20combination%20of%20high-performance%20computing%20and%20advanced%20computational%20methods%20pave%20the%20route%20for%20a%20rational%20approach%20to%20the%20design%20of%20novel%20chalcogenides%20possessing%20tuned%20properties%20for%20specific%20applications%20in%20next-generation%20devices.%22%2C%22date%22%3A%222019%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.solidstatesciences.2019.06.014%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.solidstatesciences.2019.06.014%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221293-2558%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-04-30T14%3A42%3A43Z%22%7D%7D%2C%7B%22key%22%3A%222UM3PTG2%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Tuerhong%20et%20al.%22%2C%22parsedDate%22%3A%222019%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ER.%20Tuerhong%2C%20M.%20Boero%2C%20J.-P.%20Bucher%2C%20Molecular%20attachment%20to%20a%20microscope%20tip%3A%20inelastic%20tunneling%2C%20Kondo%20screening%2C%20and%20thermopower%2C%20Beilstein%20Journal%20of%20Nanotechnology%2010%20%282019%29%201243%26%23x2013%3B1250.%20%3Ca%20class%3D%27zp-ItemURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3762%5C%2Fbjnano.10.124%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3762%5C%2Fbjnano.10.124%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Molecular%20attachment%20to%20a%20microscope%20tip%3A%20inelastic%20tunneling%2C%20Kondo%20screening%2C%20and%20thermopower%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Rouzhaji%22%2C%22lastName%22%3A%22Tuerhong%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jean-Pierre%22%2C%22lastName%22%3A%22Bucher%22%7D%5D%2C%22abstractNote%22%3A%22The%20vibrational%20excitation%20related%20transport%20properties%20of%20a%20manganese%20phthalocyanine%20molecule%20suspended%20between%20the%20tip%20of%20a%20scanning%20tunneling%20microsope%20%28STM%29%20and%20a%20surface%20are%20investigated%20by%20combining%20the%20local%20manipulation%20capabilities%20of%20the%20STM%20with%20inelastic%20electron%20tunneling%20spectroscopy.%20By%20attachment%20of%20the%20molecule%20to%20the%20probe%20tip%2C%20the%20intrinsic%20physical%20properties%20similar%20to%20those%20exhibited%20by%20a%20free%20standing%20molecule%20become%20accessible.%20This%20technique%20allows%20one%20to%20study%20locally%20the%20magnetic%20properties%2C%20as%20well%20as%20other%20elementary%20excitations%20and%20their%20mutual%20interaction.%20In%20particular%20a%20clear%20correlation%20is%20observed%20between%20the%20Kondo%20resonance%20and%20the%20vibrations%20with%20a%20strong%20incidence%20of%20the%20Kondo%20correlation%20on%20the%20thermopower%20measured%20across%20the%20single-molecule%20junction.%22%2C%22date%22%3A%222019%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.3762%5C%2Fbjnano.10.124%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3762%5C%2Fbjnano.10.124%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222190-4286%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22RZX792Q8%22%5D%2C%22dateModified%22%3A%222019-07-19T08%3A34%3A39Z%22%7D%7D%2C%7B%22key%22%3A%226M2XK2CB%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Yoshida%20et%20al.%22%2C%22parsedDate%22%3A%222019%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EH.%20Yoshida%2C%20K.%20Koizumi%2C%20M.%20Boero%2C%20M.%20Ehara%2C%20S.%20Misumi%2C%20A.%20Matsumoto%2C%20Y.%20Kuzuhara%2C%20T.%20Sato%2C%20J.%20Ohyama%2C%20M.%20Machida%2C%20High%20Turnover%20Frequency%20CO-NO%20Reactions%20over%20Rh%20Overlayer%20Catalysts%3A%20A%20Comparative%20Study%20Using%20Rh%20Nanoparticles%2C%20Journal%20of%20Physical%20Chemistry%20C%20123%20%282019%29%206080%26%23x2013%3B6089.%20%3Ca%20class%3D%27zp-ItemURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpcc.9b00383%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpcc.9b00383%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22High%20Turnover%20Frequency%20CO-NO%20Reactions%20over%20Rh%20Overlayer%20Catalysts%3A%20A%20Comparative%20Study%20Using%20Rh%20Nanoparticles%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hiroshi%22%2C%22lastName%22%3A%22Yoshida%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenichi%22%2C%22lastName%22%3A%22Koizumi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masahiro%22%2C%22lastName%22%3A%22Ehara%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Satoshi%22%2C%22lastName%22%3A%22Misumi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Akinori%22%2C%22lastName%22%3A%22Matsumoto%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yusuke%22%2C%22lastName%22%3A%22Kuzuhara%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tetsuya%22%2C%22lastName%22%3A%22Sato%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Junya%22%2C%22lastName%22%3A%22Ohyama%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masato%22%2C%22lastName%22%3A%22Machida%22%7D%5D%2C%22abstractNote%22%3A%22A%20comparative%20turnover%20frequency%20%28TOF%29%20study%20for%20structure-sensitive%20CO-NO%20reactions%20between%20overlayer%20%28thin-film%29%20and%20nanoparticle%20Rh%20catalysts%20was%20performed%20using%20a%20combined%20experimental%20and%20theoretical%20approach.%20Two%20types%20of%20honeycomb%20catalysts%20were%20prepared%3A%20one%20by%20the%20arc-plasma%20deposition%20of%20a%203%20nm%20Rh%20overlayer%20having%20a%20%28111%29%20preferential%20orientation%20atop%20a%2020-mu%20m%20Fe-Cr-Al%20metal%20foil%20and%20the%20other%20by%20a%20conventional%20wet%20coating%20of%20Rh%5C%2FZrO2%20powders%20comprised%20of%20Rh%20nanoparticles%20onto%20a%20cordierite%20honeycomb.%20The%20reaction%20rate%20of%20the%20overlayer%20was%20found%20to%20be%20more%20superior%20to%20the%20nanoparticles%2C%20despite%20a%20smaller%20surface%20area%2C%20as%20the%20TOF%20was%2014-fold%20greater%20on%20the%20overlayer%20than%20on%20the%20nanoparticle.%20In%20situ%20infrared%20spectroscopy%20suggested%20that%20a%20hollow-site%20NO%20formed%20onto%20the%20overlayer%20in%20contrast%20to%20the%20bridge-%20and%20on-top-NO%20adsorptions%20on%20the%20nanoparticles.%20The%20energy%20barriers%20for%20surface%20reactions%20of%20adsorbed%20NO%20molecules%20were%20analyzed%20using%20a%20density%20functional%20theory%20based%20molecular%20dynamics%20approach%20for%20a%20Rh%28111%29%20slab%20and%20a%20Rh%20cluster%20to%20model%20the%20overlayer%20and%20the%20nanoparticle.%20The%20nanoparticle%20was%20more%20favorable%20to%20the%20dissociation%20of%20bridge%20NO%20compared%20with%20the%20hollow%20NO%20on%20the%20overlayer%20despite%20needing%20to%20overcome%20much%20greater%20barriers%20for%20N-N%20recombination%2C%20suggesting%20that%20the%20surface%20of%20the%20nanoparticles%20was%20predominately%20covered%20by%20N%20and%20O%20atoms%2C%20where%20N-N%20recombination%20was%20the%20rate-limiting%20step.%20Conversely%2C%20the%20Rh%28111%29%20overlayer%2C%20which%20offered%20not%20only%20moderate%20or%20comparable%20energy%20barriers%20for%20NO%20dissociation%20and%20N-N%20recombination%20but%20also%20a%20lower%20energy%20barrier%20migration%20for%20N%20atoms%20on%20the%20surface%2C%20enabled%20a%20high-TOF%20reaction.%20The%20proposed%20mechanism%20was%20rationalized%20by%20comparing%20the%20results%20with%20the%20empirical%20kinetics%20of%20CO-NO%20reactions.%22%2C%22date%22%3A%222019%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Facs.jpcc.9b00383%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpcc.9b00383%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221932-7447%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222019-07-19T08%3A34%3A52Z%22%7D%7D%5D%7D
[1]
R. Barhoumi, A. Amokrane, S. Klyatskaya, M. Boero, M. Ruben, J.-P. Bucher, Screening the 4f-electron spin of TbPc2 single-molecule magnets on metal substrates by ligand channeling, Nanoscale 11 (2019) 21167–21179.
https://doi.org/10.1039/c9nr05873g.
[1]
T.-Q. Duong, C. Massobrio, G. Ori, M. Boero, E. Martin, Thermal conductivity and transport modes in glassy GeTe4 by first-principles molecular dynamics, Physical Review Materials 3 (2019) 105401.
https://doi.org/10.1103/PhysRevMaterials.3.105401.
[1]
M. Hatakeyama, K. Koizurni, M. Boero, K. Nobusada, H. Hori, T. Misonou, T. Kobayashi, S. Nakamura, Unique Structural Relaxations and Molecular Conformations of Porphyra-334 at the Excited State, Journal of Physical Chemistry B 123 (2019) 7649–7656.
https://doi.org/10.1021/acs.jpcb.9b03744.
[1]
K. Koizumi, K. Nobusada, M. Boero, Hydrogen storage mechanism and diffusion in metal-organic frameworks, Physical Chemistry Chemical Physics 21 (2019) 7756–7764.
https://doi.org/10.1039/c8cp07467d.
[1]
G. Ori, A. Bouzid, E. Martin, C. Massobrio, S. Le Roux, M. Boero, Chalcogenide glasses as a playground for the application of first-principles molecular dynamics to disordered materials, Solid State Sciences 95 (2019) 105925.
https://doi.org/10.1016/j.solidstatesciences.2019.06.014.
[1]
R. Tuerhong, M. Boero, J.-P. Bucher, Molecular attachment to a microscope tip: inelastic tunneling, Kondo screening, and thermopower, Beilstein Journal of Nanotechnology 10 (2019) 1243–1250.
https://doi.org/10.3762/bjnano.10.124.
[1]
H. Yoshida, K. Koizumi, M. Boero, M. Ehara, S. Misumi, A. Matsumoto, Y. Kuzuhara, T. Sato, J. Ohyama, M. Machida, High Turnover Frequency CO-NO Reactions over Rh Overlayer Catalysts: A Comparative Study Using Rh Nanoparticles, Journal of Physical Chemistry C 123 (2019) 6080–6089.
https://doi.org/10.1021/acs.jpcc.9b00383.
1839302
NZSFH59F
2018
1
surface-science-reports
50
creator
asc
year
7054
https://www.ipcms.unistra.fr/plugins/zotpress/
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3Afalse%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%22K9AIZR2D%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Cai%20et%20al.%22%2C%22parsedDate%22%3A%222018%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EW.%20Cai%2C%20L.%20Abella%2C%20J.%20Zhuang%2C%20X.%20Zhang%2C%20L.%20Feng%2C%20Y.%20Wang%2C%20R.%20Morales-Martinez%2C%20R.%20Esper%2C%20M.%20Boero%2C%20A.%20Metta-Magana%2C%20A.%20Rodriguez-Fortea%2C%20J.M.%20Poblet%2C%20L.%20Echegoyen%2C%20N.%20Chen%2C%20Synthesis%20and%20Characterization%20of%20Non-Isolated-Pentagon-Rule%20Actinide%20Endohedral%20Metallofullerenes%20U%40C-1%2817418%29-C-76%2C%20U%40C-1%2828324%29-C-80%2C%20and%20Th%40C-1%2828324%29-C-80%3A%20Low-Symmetry%20Cage%20Selection%20Directed%20by%20a%20Tetravalent%20Ion%2C%20Journal%20of%20the%20American%20Chemical%20Society%20140%20%282018%29%2018039%26%23x2013%3B18050.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjacs.8b10435%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjacs.8b10435%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Synthesis%20and%20Characterization%20of%20Non-Isolated-Pentagon-Rule%20Actinide%20Endohedral%20Metallofullerenes%20U%40C-1%2817418%29-C-76%2C%20U%40C-1%2828324%29-C-80%2C%20and%20Th%40C-1%2828324%29-C-80%3A%20Low-Symmetry%20Cage%20Selection%20Directed%20by%20a%20Tetravalent%20Ion%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Wenting%22%2C%22lastName%22%3A%22Cai%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Laura%22%2C%22lastName%22%3A%22Abella%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jiaxin%22%2C%22lastName%22%3A%22Zhuang%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Xingxing%22%2C%22lastName%22%3A%22Zhang%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Lai%22%2C%22lastName%22%3A%22Feng%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yaofeng%22%2C%22lastName%22%3A%22Wang%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Roser%22%2C%22lastName%22%3A%22Morales-Martinez%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ronda%22%2C%22lastName%22%3A%22Esper%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Alejandro%22%2C%22lastName%22%3A%22Metta-Magana%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Antonio%22%2C%22lastName%22%3A%22Rodriguez-Fortea%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Josep%20M.%22%2C%22lastName%22%3A%22Poblet%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Luis%22%2C%22lastName%22%3A%22Echegoyen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ning%22%2C%22lastName%22%3A%22Chen%22%7D%5D%2C%22abstractNote%22%3A%22For%20the%20first%20time%2C%20actinide%20endohedral%20metallofullerenes%20%28EMFs%29%20with%20non-isolated-pentagon-rule%20%28non-IPR%29%20carbon%20cages%2C%20U%40C-80%2C%20Th%40C-80%2C%20and%20U%40C-76%2C%20have%20been%20successfully%20synthesized%20and%20fully%20characterized%20by%20mass%20spectrometry%2C%20single%20crystal%20X-ray%20diffractometry%2C%20UV-vis-NIR%20and%20Raman%20spectroscopy%2C%20and%20cyclic%20voltammetry.%20Crystallo-graphic%20analysis%20revealed%20that%20the%20U%40C-80%20and%20Th%40C-80%20share%20the%20same%20non-IPR%20cage%20of%20C-1%2828324%29-C-80%2C%20and%20U%40C-76%20was%20assigned%20to%20non-IPR%20U%40C-1%2817418%29-C-76.%20All%20of%20these%20cages%20are%20chiral%20and%20have%20never%20been%20reported%20before.%20Further%20structural%20analyses%20show%20that%20enantiomers%20of%20C-1%2817418%29-C-76%20and%20C-1%2828324%29-C-80%20share%20a%20significant%20continuous%20portion%20of%20the%20cage%20and%20are%20topologically%20connected%20by%20only%20two%20C-2%20insertions.%20DFT%20calculations%20show%20that%20the%20stabilization%20of%20these%20unique%20non-IPR%20fullerenes%20originates%20from%20a%20four-electron%20transfer%2C%20a%20significant%20degree%20of%20covalency%2C%20and%20the%20resulting%20strong%20host-guest%20interactions%20between%20the%20actinide%20ions%20and%20the%20fullerene%20cages.%20Moreover%2C%20because%20the%20actinide%20ion%20displays%20high%20mobility%20within%20the%20fullerene%2C%20both%20the%20symmetry%20of%20the%20carbon%20cage%20and%20the%20possibility%20of%20forming%20chiral%20fullerenes%20play%20important%20roles%20to%20determine%20the%20isomer%20abundances%20at%20temperatures%20of%20fullerene%20formation.%20This%20study%20provides%20what%20is%20probably%20one%20of%20the%20most%20complete%20examples%20in%20which%20carbon%20cage%20selection%20occurs%20through%20thermodynamic%20control%20at%20high%20temperatures%2C%20so%20the%20selected%20cages%20do%20not%20necessarily%20coincide%20with%20the%20most%20stable%20ones%20at%20room%20temperature.%20This%20work%20also%20demonstrated%20that%20the%20metal-cage%20interactions%20in%20actinide%20EMFs%20show%20remarkable%20differences%20from%20those%20previously%20known%20for%20lanthanide%20EMFs.%20These%20unique%20interactions%20not%20only%20could%20stabilize%20new%20carbon%20cage%20structures%2C%20but%20more%20importantly%2C%20they%20lead%20to%20a%20new%20family%20of%20metallofullerenes%20for%20which%20the%20cage%20selection%20pattern%20is%20different%20to%20that%20observed%20so%20far%20for%20nonactinide%20EMFs.%20For%20this%20new%20family%2C%20the%20simple%20ionic%20A%28q%2B%29%40C-2n%28q-%29%20model%20makes%20predictions%20less%20reliable%2C%20and%20in%20general%2C%20unambiguously%20discerning%20the%20isolated%20structures%20requires%20the%20combination%20of%20accurate%20computational%20and%20experimental%20data.%22%2C%22date%22%3A%222018%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Fjacs.8b10435%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Fjacs.8b10435%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220002-7863%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222021-10-20T12%3A48%3A36Z%22%7D%7D%2C%7B%22key%22%3A%22ZT9UPT73%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Chaker%20et%20al.%22%2C%22parsedDate%22%3A%222018%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EZ.%20Chaker%2C%20G.%20Ori%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20E.%20Furet%2C%20A.%20Bouzid%2C%20First-principles%20study%20of%20the%20atomic%20structure%20of%20glassy%20Ga10Ge15Te75%2C%20Journal%20of%20Non-Crystalline%20Solids%20498%20%282018%29%20338%26%23x2013%3B344.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jnonaysol.2018.03.039%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jnonaysol.2018.03.039%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22First-principles%20study%20of%20the%20atomic%20structure%20of%20glassy%20Ga10Ge15Te75%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ziyad%22%2C%22lastName%22%3A%22Chaker%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Gudo%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22E.%22%2C%22lastName%22%3A%22Furet%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A.%22%2C%22lastName%22%3A%22Bouzid%22%7D%5D%2C%22abstractNote%22%3A%22First-principles%20molecular%20dynamics%20have%20been%20employed%20to%20highlight%20the%20structural%20properties%20of%20glassy%20Ga10Ge15Te75%20%28GGT%29%2C%20a%20promising%20disordered%20system%20for%20infrared%20applications.%20Our%20approach%20relies%20entirely%20on%20the%20predictive%20power%20of%20density%20functional%20theory%2C%20in%20combination%20with%20a%20careful%20choice%20of%20the%20exchange-correlation%20functional%20and%20the%20account%20of%20van%20der%20Waals%20interactions.%20Glassy%20GGT%20can%20be%20viewed%20as%20a%20predominantly%20tetrahedral%20network%2C%20both%20with%20respect%20to%20Ge%20and%20Ga%20atoms.%20Te%20atoms%20are%20mostly%20twofold%20coordinated%20to%20all%20combinations%20of%20pairs%20of%20Ge%2C%20Ga%20or%20Te%20atoms.%20We%20find%20little%20evidence%20of%20intermediate%20range%20order%20in%20the%20partial%20structure%20factors.%22%2C%22date%22%3A%222018%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.jnonaysol.2018.03.039%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.jnonaysol.2018.03.039%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220022-3093%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222018-11-07T13%3A12%3A14Z%22%7D%7D%2C%7B%22key%22%3A%226TNGM2AD%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Chaker%20et%20al.%22%2C%22parsedDate%22%3A%222018%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EZ.%20Chaker%2C%20G.%20Ori%2C%20C.%20Tug%26%23xE8%3Bne%2C%20S.%20Le%20Roux%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20E.%20Martin%2C%20A.%20Bouzid%2C%20The%20role%20of%20dispersion%20forces%20on%20the%20atomic%20structure%20of%20glassy%20chalcogenides%3A%20The%20case%20of%20GeSe4%20and%20GeS4%2C%20Journal%20of%20Non-Crystalline%20Solids%20499%20%282018%29%20167%26%23x2013%3B172.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2018.07.012%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2018.07.012%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22The%20role%20of%20dispersion%20forces%20on%20the%20atomic%20structure%20of%20glassy%20chalcogenides%3A%20The%20case%20of%20GeSe4%20and%20GeS4%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ziyad%22%2C%22lastName%22%3A%22Chaker%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Christine%22%2C%22lastName%22%3A%22Tug%5Cu00e8ne%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%5D%2C%22abstractNote%22%3A%22Recent%20first-principles%20molecular%20dynamics%20simulations%20%28FPMD%29%20results%20on%20two%20chalcogenide%20glasses%20%28glassy%20GeS4%20and%20GeSe4%29%20are%20revisited%20by%20accounting%20explicitly%20for%20van%20der%20Waals%20%28vdW%29%20dispersion%20forces.%20This%20effort%20is%20motivated%20by%20the%20observation%20that%20such%20contributions%20were%20found%20to%20be%20important%20in%20the%20case%20of%20glassy%20GeTe4%20while%20they%20were%20negligible%20for%20another%20disordered%20chalcogenide%20system%2C%20liquid%20GeSe2.%20With%20the%20present%20results%2C%20we%20provide%20additional%20evidence%20intended%20to%20establish%20under%20which%20conditions%20and%20for%20which%20systems%20the%20consideration%20of%20dispersion%20forces%20plays%20a%20role%20in%20determining%20the%20atomic%20structure.%20For%20these%20purposes%20we%20employ%20two%20different%20dispersion%20schemes%20used%20in%20conjunction%20with%20the%20BLYP%20%28Becke%2C%20Lee%2C%20Yang%20and%20Parr%29%20exchange%20correlation%20functional.%20The%20first%20is%20the%20DFT-D2%20vdW%20correction%20introduced%20by%20Grimme%20et%20al.%20%28Grimme%20%282006%29%20%5B1%5D%29%20while%20the%20second%20is%20the%20vdW%28w%29%20approach%20based%20on%20the%20Wannier%20functions%20analysis%20%28Silvestrelli%2C%202008%20%5B2%5D%29.%20The%20atomic%20structures%20obtained%20agree%20well%20with%20the%20ones%20obtained%20by%20Bouzid%20et%20al.%20%5B3%5D%20without%20considering%20dispersion%20forces.%20Due%20to%20the%20vdW%20interactions%2C%20glassy%20GeSe4%20features%20a%20higher%20number%20of%20Ge%20fourfold%20coordinated.%20The%20two%20vdW%20approaches%20also%20agree%20to%20a%20large%20extent%2C%20exceptions%20occurring%20for%20some%20moderate%20differences%20in%20the%20intensity%20of%20the%20peaks%20in%20the%20Ge-Ge%20pair%20correlation%20function.%22%2C%22date%22%3A%222018%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.jnoncrysol.2018.07.012%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2018.07.012%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220022-3093%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-04-30T14%3A44%3A04Z%22%7D%7D%2C%7B%22key%22%3A%22ERXUG5JH%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Chaker%20et%20al.%22%2C%22parsedDate%22%3A%222018%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EZ.%20Chaker%2C%20A.%20Bouzid%2C%20B.%20Coasne%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20G.%20Ori%2C%20The%20structure%20and%20dipolar%20properties%20of%20CO2%20adsorbed%20in%20a%20porous%20glassy%20chalcogel%3A%20Insights%20from%20first-principles%20molecular%20dynamics%2C%20Journal%20of%20Non-Crystalline%20Solids%20498%20%282018%29%20288%26%23x2013%3B293.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2018.06.031%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2018.06.031%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22The%20structure%20and%20dipolar%20properties%20of%20CO2%20adsorbed%20in%20a%20porous%20glassy%20chalcogel%3A%20Insights%20from%20first-principles%20molecular%20dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ziyad%22%2C%22lastName%22%3A%22Chaker%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Benoit%22%2C%22lastName%22%3A%22Coasne%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%5D%2C%22abstractNote%22%3A%22A%20thorough%20understanding%20of%20the%20interactions%20of%20CO2%20with%20the%20hosting%20porous%20network%20is%20crucial%20for%20the%20design%20of%20new%20adsorbent%20materials%20with%20enhanced%20gas%20adsorption%20capacity%20and%20selectivity.%20In%20this%20paper%2C%20first-principles%20molecular%20dynamics%20simulations%20are%20performed%20to%20assess%20the%20interactions%20of%20CO2%20adsorbed%20in%20a%20nanoporous%20glassy%20chalcogenide%20%28i.e.%20chalcogel%29%20under%20relevant%20laboratory%20conditions.%20The%20structure%20and%20local%20organization%20of%20the%20confined%20CO2%20molecules%20are%20analyzed%20in%20terms%20of%20atomic%20density%2C%20orientational%20order%20parameter%20and%20pair%20correlation%20functions.%20Maximally%20localized%20Wannier%20functions%20are%20used%20to%20unravel%20the%20electronic%20structure%2C%20the%20local%20molecular%20dipole%20and%20the%20nature%20of%20the%20chemical%20bonding%20at%20the%20interface%20between%20CO2%20and%20the%20glassy%20surface.%20Our%20results%20provide%20a%20useful%20insight%20on%20the%20complex%20interplay%20between%20the%20chemical%20interactions%20competing%20at%20the%20buried%20interface%20made%20by%20the%20CO2%20molecules%20adsorbed%20in%20g-GeS2%20chalcogel.%22%2C%22date%22%3A%222018%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.jnoncrysol.2018.06.031%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2018.06.031%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220022-3093%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222021-10-20T12%3A49%3A51Z%22%7D%7D%2C%7B%22key%22%3A%22V55SQNS2%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Cotin%20et%20al.%22%2C%22parsedDate%22%3A%222018%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EG.%20Cotin%2C%20C.%20Kiefer%2C%20F.%20Perton%2C%20M.%20Boero%2C%20B.%20%26%23xD6%3Bzdamar%2C%20A.%20Bouzid%2C%20G.%20Ori%2C%20C.%20Massobrio%2C%20D.%20Begin%2C%20B.%20Pichon%2C%20D.%20Mertz%2C%20S.%20B%26%23xE9%3Bgin-Colin%2C%20Evaluating%20the%20Critical%20Roles%20of%20Precursor%20Nature%20and%20Water%20Content%20When%20Tailoring%20Magnetic%20Nanoparticles%20for%20Specific%20Applications%2C%20ACS%20Appl.%20Nano%20Mater.%201%20%282018%29%204306%26%23x2013%3B4316.%20%3Ca%20class%3D%27zp-ItemURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facsanm.8b01123%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facsanm.8b01123%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Evaluating%20the%20Critical%20Roles%20of%20Precursor%20Nature%20and%20Water%20Content%20When%20Tailoring%20Magnetic%20Nanoparticles%20for%20Specific%20Applications%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Geoffrey%22%2C%22lastName%22%3A%22Cotin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22C%5Cu00e9line%22%2C%22lastName%22%3A%22Kiefer%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Francis%22%2C%22lastName%22%3A%22Perton%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Burak%22%2C%22lastName%22%3A%22%5Cu00d6zdamar%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Dominique%22%2C%22lastName%22%3A%22Begin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Benoit%22%2C%22lastName%22%3A%22Pichon%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Damien%22%2C%22lastName%22%3A%22Mertz%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sylvie%22%2C%22lastName%22%3A%22B%5Cu00e9gin-Colin%22%7D%5D%2C%22abstractNote%22%3A%22Because%20of%20the%20broad%20range%20of%20application%20of%20iron%20oxide%20nanoparticles%20%28NPs%29%2C%20the%20control%20of%20their%20size%20and%20shape%20on%20demand%20remains%20a%20great%20challenge%2C%20as%20these%20parameters%20are%20of%20upmost%20importance%20to%20provide%20NPs%20with%20magnetic%20properties%20tailored%20to%20the%20targeted%20application.%20One%20promising%20synthesis%20process%20to%20tune%20their%20size%20and%20shape%20is%20the%20thermal%20decomposition%20one%2C%20for%20which%20a%20lot%20of%20parameters%20were%20investigated.%20But%20two%20crucial%20issues%20were%20scarcely%20addressed%3A%20the%20precursor%5Cu2019s%20nature%20and%20water%20content.%20Two%20in%20house%20iron%20stearates%20with%20two%20or%20three%20stearate%20chains%20were%20synthesized%2C%20dehydrated%2C%20and%20then%20tested%20in%20standard%20synthesis%20conditions%20of%20spherical%20and%20cubic%20NPs.%20Investigations%20combined%20with%20modeling%20showed%20that%20the%20precursor%5Cu2019s%20nature%20and%20hydration%20rate%20strongly%20affect%20the%20thermal%20decomposition%20kinetics%20and%20yields%2C%20which%2C%20in%20turn%2C%20influence%20the%20NP%20size.%20The%20cubic%20shape%20depends%20on%20the%20decomposition%20kinetics%20but%20also%20crucially%20on%20the%20water%20content.%20A%20microscopic%20insight%20was%20provided%20by%20first-principles%20simulation%20showing%20an%20iron%20reduction%20along%20the%20reaction%20pathway%20and%20a%20participation%20of%20water%20molecules%20to%20the%20building%20unit%20formation.%22%2C%22date%22%3A%222018%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Facsanm.8b01123%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facsanm.8b01123%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%226IWM732K%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222022-02-08T08%3A18%3A06Z%22%7D%7D%2C%7B%22key%22%3A%229M53Z7UI%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Iannuccelli%20et%20al.%22%2C%22parsedDate%22%3A%222018%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EV.%20Iannuccelli%2C%20E.%20Maretti%2C%20A.%20Bellini%2C%20D.%20Malferrari%2C%20G.%20Ori%2C%20M.%20Montorsi%2C%20M.%20Bondi%2C%20E.%20Truzzi%2C%20E.%20Leo%2C%20Organo-modified%20bentonite%20for%20gentamicin%20topical%20application%3A%20Interlayer%20structure%20and%20in%20vivo%20skin%20permeation%2C%20Applied%20Clay%20Science%20158%20%282018%29%20158%26%23x2013%3B168.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.clay.2018.03.029%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.clay.2018.03.029%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Organo-modified%20bentonite%20for%20gentamicin%20topical%20application%3A%20Interlayer%20structure%20and%20in%20vivo%20skin%20permeation%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Valentina%22%2C%22lastName%22%3A%22Iannuccelli%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Eleonora%22%2C%22lastName%22%3A%22Maretti%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Alessia%22%2C%22lastName%22%3A%22Bellini%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Daniele%22%2C%22lastName%22%3A%22Malferrari%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Monia%22%2C%22lastName%22%3A%22Montorsi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Moreno%22%2C%22lastName%22%3A%22Bondi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Eleonora%22%2C%22lastName%22%3A%22Truzzi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Eliana%22%2C%22lastName%22%3A%22Leo%22%7D%5D%2C%22abstractNote%22%3A%22Recent%20biomedical%20applications%20of%20clay%20materials%20have%20included%20organically%20modified%20clays%20or%20clay%20minerals%20with%20the%20purpose%20of%20modifying%20and%20improving%20drug%20biological%20activity.%20The%20present%20research%20aims%20to%20explore%20the%20potential%20benefits%20provided%20by%20a%20raw%20bentonite%20%28Bt%29%20modified%20by%20gentamicin%20%28GM%29%20adsorbed%20within%20montmorillonite%20interlayers%20in%20the%20management%20of%20cutaneous%20infectious%20diseases.%20Information%20arisen%20from%20controlled%20X-ray%20powder%20diffraction%2C%20thermogravimetry%20coupled%20with%20evolved%20gas%20mass%20spectrometry%2C%20and%20molecular%20dynamics%20simulations%20pointed%20out%20GM%20monolayer%20arrangement%20within%20montmorillonite%20framework%20without%20producing%20substantial%20effects%20on%20the%20layer%20periodicity.%20Concerning%20skin%20biomedical%20application%2C%20unlike%20the%20pure%20antibiotic%20permeating%20along%20the%20trans-follicular%20pathway%20across%20stratum%20corneum%2C%20the%20organo-modified%20Bt%5C%2FGM%20would%20favor%20the%20trans-epidermal%20route%20along%20inter-cluster%20corneocyte%20region%2C%20as%20in%20vivo%20skin%20penetration%20studies%20by%20means%20of%20tape%20stripping%20test%20indicated.%20Based%20on%20the%20results%20obtained%2C%20GM%20intercalation%20could%20represent%20a%20potential%20advantageous%20approach%20allowing%20a%20long-term%20Bt%5C%2FGM%20reservoir%20for%20sustained%20antibacterial%20activity.%22%2C%22date%22%3A%222018%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.clay.2018.03.029%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.clay.2018.03.029%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220169-1317%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222018-11-07T14%3A18%3A17Z%22%7D%7D%2C%7B%22key%22%3A%22N8R9HW3W%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Koizumi%20et%20al.%22%2C%22parsedDate%22%3A%222018%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Koizumi%2C%20H.%20Yoshida%2C%20M.%20Boero%2C%20K.%20Tamai%2C%20S.%20Hosokawa%2C%20T.%20Tanaka%2C%20K.%20Nobusada%2C%20M.%20Machida%2C%20A%20detailed%20insight%20into%20the%20catalytic%20reduction%20of%20NO%20operated%20by%20Cr-Cu%20nanostructures%20embedded%20in%20a%20CeO2%20surface%2C%20Physical%20Chemistry%20Chemical%20Physics%2020%20%282018%29%2025592--25601.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc8cp04314k%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc8cp04314k%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22A%20detailed%20insight%20into%20the%20catalytic%20reduction%20of%20NO%20operated%20by%20Cr-Cu%20nanostructures%20embedded%20in%20a%20CeO2%20surface%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenichi%22%2C%22lastName%22%3A%22Koizumi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hiroshi%22%2C%22lastName%22%3A%22Yoshida%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kazuki%22%2C%22lastName%22%3A%22Tamai%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Saburo%22%2C%22lastName%22%3A%22Hosokawa%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tsunehiro%22%2C%22lastName%22%3A%22Tanaka%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Katsuyuki%22%2C%22lastName%22%3A%22Nobusada%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masato%22%2C%22lastName%22%3A%22Machida%22%7D%5D%2C%22abstractNote%22%3A%22Replacing%20rare%20and%20expensive%20elements%2C%20such%20as%20Pt%2C%20Pd%2C%20and%20Rh%2C%20commonly%20used%20in%20catalytic%20devices%20with%20more%20abundant%20and%20less%20expensive%20ones%20is%20mandatory%20to%20realize%20efficient%2C%20sustainable%20and%20economically%20appealing%20three-way%20catalysts.%20In%20this%20context%2C%20the%20surface%20of%20a%20Cr-Cu%5C%2FCeO2%20system%20represents%20a%20versatile%20catalyst%20for%20the%20conversion%20of%20toxic%20NO%20into%20harmless%20N-2.%20Yet%2C%20a%20clear%20picture%20of%20the%20underlying%20mechanism%20is%20still%20missing.%20We%20provide%20here%20a%20detailed%20insight%20into%20such%20a%20reaction%20mechanism%20by%20means%20of%20a%20combined%20experimental%20and%20theoretical%20study.%20Fourier-transform%20infrared%20spectroscopy%20is%20used%20to%20detect%20all%20the%20products%20resulting%20from%20catalytic%20reactions%20of%20NO%20and%20CO%20on%20the%20surface%20of%20a%20Cr-Cu%5C%2FCeO2%20nanocatalyst.%20CO%20pulsing%20experiments%20unveil%20that%20reactions%20of%20CO%20with%20O%20atoms%20at%20the%20Cr-Cu%5C%2FCeO2%20surface%20are%20the%20major%20factors%20responsible%20for%20the%20formation%20of%20surface%20vacancies.%20On%20these%20grounds%2C%20a%20comprehensive%20picture%20of%20the%20NO%20reduction%20and%20the%20role%20of%20both%20Cu%20and%20Cr%20dopants%20and%20vacancies%20is%20rationalized%20by%20first-principles%20modeling.%20Our%20findings%20provide%20a%20general%20route%20for%20the%20realization%20of%20ceria-based%20cost-effective%20catalysts.%22%2C%22date%22%3A%222018%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fc8cp04314k%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fc8cp04314k%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221463-9076%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222019-01-15T13%3A14%3A12Z%22%7D%7D%2C%7B%22key%22%3A%22TNME8T76%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Kromik%20et%20al.%22%2C%22parsedDate%22%3A%222018%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EA.%20Kromik%2C%20E.V.%20Levchenko%2C%20C.%20Massobrio%2C%20A.V.%20Evteev%2C%20Diffusion%20in%20Ni%26%23x2013%3BZr%20Melts%3A%20Insights%20from%20Statistical%20Mechanics%20and%20Atomistic%20Modeling%2C%20Advanced%20Theory%20and%20Simulations%201%20%282018%29%201800109.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Fadts.201800109%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Fadts.201800109%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Diffusion%20in%20Ni%5Cu2013Zr%20Melts%3A%20Insights%20from%20Statistical%20Mechanics%20and%20Atomistic%20Modeling%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Andreas%22%2C%22lastName%22%3A%22Kromik%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Elena%20V.%22%2C%22lastName%22%3A%22Levchenko%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Alexander%20V.%22%2C%22lastName%22%3A%22Evteev%22%7D%5D%2C%22abstractNote%22%3A%22Abstract%20An%20accurate%20database%20of%20diffusion%20properties%20of%20Ni%5Cu2013Zr%20melts%20is%20generated%20within%20the%20framework%20of%20the%20molecular-dynamics%20method%20in%20conjunction%20with%20a%20semi-empirical%20many-body%20interatomic%20potential.%20The%20reliability%20of%20the%20model%20description%20of%20Ni%5Cu2013Zr%20melts%20is%20confirmed%20via%20comparison%20of%20the%20simulation%20results%20with%20the%20existing%20experimental%20data%20on%20diffusion%20properties%20of%20Ni%5Cu2013Zr%20melts.%20A%20statistical%20mechanical%20formalism%20is%20employed%20to%20understand%20the%20behavior%20of%20the%20cross-correlation%20between%20the%20interdiffusion%20flux%20and%20the%20force%20caused%20by%20the%20difference%20in%20the%20average%20random%20accelerations%20of%20atoms%20of%20different%20species%20in%20the%20short%20time%20limit%20t%5Cu21920.%20This%20theoretical%20description%20is%20exploited%20to%20analyze%20the%20simulation%20data%20on%20the%20diffusion%20properties%20of%20Ni%5Cu2013Zr%20melts.%20On%20this%20basis%2C%20it%20is%20found%20that%20in%20the%20composition%20range%200.25%3C%5Cu223cxc%20Ni%20%3C%5Cu223cx0.5%20both%20single-particle%20and%20collective%20diffusion%20dynamics%20slow%20down%20homogeneously%20upon%20undercooling%20of%20Ni%5Cu2013Zr%20melts.%20Furthermore%2C%20it%20is%20inferred%20that%20such%20homogeneous%20dynamical%20slowdown%20is%20related%20to%20the%20enhanced%20stability%20of%20undercooled%20melt%20against%20crystallization.%20As%20a%20consequence%2C%20Ni%5Cu2013Zr%20alloys%20within%20this%20composition%20range%20are%20identified%20as%20viable%20glass%20formers.%22%2C%22date%22%3A%222018%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1002%5C%2Fadts.201800109%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1002%5C%2Fadts.201800109%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222019-01-15T16%3A38%3A09Z%22%7D%7D%2C%7B%22key%22%3A%22TAZXWEF8%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Martin%20et%20al.%22%2C%22parsedDate%22%3A%222018%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EE.%20Martin%2C%20P.L.%20Palla%2C%20F.%20Cleri%2C%20A.%20Bouzid%2C%20G.%20Ori%2C%20S.%20Le%20Roux%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20On%20the%20occurrence%20of%20size%20effects%20in%20the%20calculation%20of%20thermal%20conductivity%20by%20first-principles%20molecular%20dynamics%3A%20The%20case%20of%20glassy%20GeTe4%2C%20Journal%20of%20Non-Crystalline%20Solids%20498%20%282018%29%20190%26%23x2013%3B193.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2018.05.014%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2018.05.014%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22On%20the%20occurrence%20of%20size%20effects%20in%20the%20calculation%20of%20thermal%20conductivity%20by%20first-principles%20molecular%20dynamics%3A%20The%20case%20of%20glassy%20GeTe4%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Pier%20Luca%22%2C%22lastName%22%3A%22Palla%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fabrizio%22%2C%22lastName%22%3A%22Cleri%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22The%20thermal%20conductivity%20of%20a%20glass%20can%20be%20obtained%20by%20first-principles%20molecular%20dynamics%20provided%20we%20exploit%20a%20methodology%20that%20has%20been%20termed%20the%20%5Cu201capproach-to-equilibrium%5Cu201d%20molecular%20dynamics%20%28AEMD%29%20%5B1%2C2%2C%20Chap.%208%5D.%20In%20the%20present%20work%2C%20we%20investigate%20the%20occurrence%20of%20size%20effects%20by%20comparing%20the%20thermal%20conductivity%20of%20two%20g-GeTe4%20atomic%20models%20of%20different%20sizes.%20This%20issue%20is%20far%20from%20being%20trivial%20since%2C%20in%20principle%2C%20size%20effects%20are%20not%20expected%20to%20occur%20in%20disordered%20systems%20beyond%20a%20few%20interatomic%20distances.%20For%20this%20reason%2C%20it%20is%20important%20to%20search%20unambiguous%20pieces%20of%20evidence%20substantiating%20this%20point.%20The%20first%20system%20of%20length%20L%20%3D%2018%20angstrom%20contains%20185%20atoms.%20By%20duplicating%20it%20along%20one%20direction%20we%20form%20the%20second%20system%2C%20that%20contains%20370%20atoms%20and%20features%20a%20double%20length%20L%20%3D%2036%20angstrom%20and%20an%20identical%20cross%20section.%20The%20thermal%20conductivity%20increases%20by%20a%20factor%203%20from%20L%20%3D%2018%20to%2036%20angstrom%2C%20thereby%20approaching%20the%20experimental%20value.%20Our%20investigation%20exemplifies%20the%20crucial%20role%20of%20the%20system%20size%20to%20take%20full%20advantage%20of%20the%20AEMD%20methodology%20and%20bring%20the%20calculated%20values%20in%20better%20agreement%20with%20experiments.%22%2C%22date%22%3A%222018%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.jnoncrysol.2018.05.014%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.jnoncrysol.2018.05.014%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220022-3093%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222022-02-10T07%3A54%3A32Z%22%7D%7D%2C%7B%22key%22%3A%22NZ24E9PX%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Massobrio%20et%20al.%22%2C%22parsedDate%22%3A%222018%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EC.%20Massobrio%2C%20E.%20Martin%2C%20Z.%20Chaker%2C%20M.%20Boero%2C%20A.%20Bouzid%2C%20S.%20Le%20Roux%2C%20G.%20Ori%2C%20Sensitivity%20to%20Dispersion%20Forces%20in%20First-Principles%20Modeling%20of%20Disordered%20Chalcogenides%2C%20Frontiers%20in%20Materials%205%20%282018%29.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3389%5C%2Ffmats.2018.00078%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3389%5C%2Ffmats.2018.00078%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Sensitivity%20to%20Dispersion%20Forces%20in%20First-Principles%20Modeling%20of%20Disordered%20Chalcogenides%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Martin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ziyad%22%2C%22lastName%22%3A%22Chaker%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%5D%2C%22abstractNote%22%3A%22The%20inclusion%20of%20dispersion%20%28van%20der%20Waals%2C%20vdW%29%20forces%20in%20first-principles%20modeling%20of%20disordered%20chalcogenides%20is%20analyzed%20and%20critically%20discussed%20in%20view%20of%20their%20impact%20on%20the%20atomic%20structure.%20To%20this%20purpose%20we%20considered%20the%20case%20of%20glassy%20GeTe4.%20We%20selected%20a%20vdW%20correction%20%28termed%20vdW%28G%29%20hereafter%29%20introduced%20by%20Grimme%20%282006%29%20and%2C%20as%20an%20alternative%2C%20the%20approach%20%28termed%20vdW%28W%29%20hereafter%29%20based%20on%20the%20Wannier%20functions%20formalism%20%28Silvestrelli%2C%202008%29.%20It%20appears%20that%20a%20strategy%20based%20on%20the%20update%20of%20the%20vdW%20interactions%20due%20to%20changes%20in%20the%20electronic%20structure%20during%20the%20dynamical%20evolution%20%28i.e.%2C%20the%20vdW%28W%29%20one%29%20provides%20results%20clearly%20different%20from%20those%20obtained%20in%20the%20vdWG%20framework.%20By%20keeping%20in%20mind%20that%20the%20nature%20of%20the%20present%20results%20is%20preliminary%20and%20reflect%20a%20trend%20to%20be%20confirmed%2C%20we%20draw%20attention%20to%20the%20different%20levels%20of%20agreement%20with%20experimental%20data%20obtained%20with%20the%20two%20vdW%20schemes%20employed.%22%2C%22date%22%3A%222018%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.3389%5C%2Ffmats.2018.00078%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.3389%5C%2Ffmats.2018.00078%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222296-8016%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-04-30T14%3A42%3A55Z%22%7D%7D%2C%7B%22key%22%3A%22NGCUR3T2%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Ozdamar%20et%20al.%22%2C%22parsedDate%22%3A%222018%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EB.%20Ozdamar%2C%20A.%20Bouzid%2C%20G.%20Ori%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20First-Principles%20Study%20of%20Dissociation%20Processes%20for%20the%20Synthesis%20of%20Fe%20and%20Co%20Oxide%20Nanoparticles%2C%20Journal%20of%20Chemical%20Theory%20and%20Computation%2014%20%282018%29%20225%26%23x2013%3B235.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jctc.7b00869%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jctc.7b00869%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22First-Principles%20Study%20of%20Dissociation%20Processes%20for%20the%20Synthesis%20of%20Fe%20and%20Co%20Oxide%20Nanoparticles%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Burak%22%2C%22lastName%22%3A%22Ozdamar%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22Thermal%20decomposition%20is%20a%20practical%20and%20reliable%20tool%20to%20synthesize%20nanoparticles%20with%20monodisperse%20size%20distribution%20and%20reproducible%20accuracy.%20The%20nature%20of%20the%20precursor%20molecules%20and%20their%20interaction%20with%20the%20environment%20during%20the%20synthesis%20process%20have%20a%20direct%20impact%20on%20the%20resulting%20nanoparticles.%20Our%20study%20focuses%20on%20widely%20used%20transition-metal%20%28Co%2C%20Fe%29%20stearates%20precursors%20and%20their%20thermal%20decomposition%20reaction%20pathway.%20We%20show%20how%20the%20nature%20of%20the%20metal%20and%20the%20presence%20or%20absence%20of%20water%20molecules%2C%20directly%20related%20to%20the%20humidity%20conditions%20during%20the%20synthesis%20process%2C%20affect%20the%20decomposition%20mechanism%20and%20the%20resulting%20transition-metal%20oxide%20building%20blocks.%20This%2C%20in%20turn%2C%20has%20a%20direct%20effect%20on%20the%20physical%20and%20chemical%20properties%20of%20the%20produced%20nanoparticles%20and%20deeply%20influences%20their%20composition%20and%20morphology.%22%2C%22date%22%3A%222018%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Facs.jctc.7b00869%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Facs.jctc.7b00869%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221549-9618%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222018-05-31T15%3A00%3A04Z%22%7D%7D%5D%7D
[1]
W. Cai, L. Abella, J. Zhuang, X. Zhang, L. Feng, Y. Wang, R. Morales-Martinez, R. Esper, M. Boero, A. Metta-Magana, A. Rodriguez-Fortea, J.M. Poblet, L. Echegoyen, N. Chen, Synthesis and Characterization of Non-Isolated-Pentagon-Rule Actinide Endohedral Metallofullerenes U@C-1(17418)-C-76, U@C-1(28324)-C-80, and Th@C-1(28324)-C-80: Low-Symmetry Cage Selection Directed by a Tetravalent Ion, Journal of the American Chemical Society 140 (2018) 18039–18050.
https://doi.org/10.1021/jacs.8b10435.
[1]
Z. Chaker, G. Ori, M. Boero, C. Massobrio, E. Furet, A. Bouzid, First-principles study of the atomic structure of glassy Ga10Ge15Te75, Journal of Non-Crystalline Solids 498 (2018) 338–344.
https://doi.org/10.1016/j.jnonaysol.2018.03.039.
[1]
Z. Chaker, G. Ori, C. Tugène, S. Le Roux, M. Boero, C. Massobrio, E. Martin, A. Bouzid, The role of dispersion forces on the atomic structure of glassy chalcogenides: The case of GeSe4 and GeS4, Journal of Non-Crystalline Solids 499 (2018) 167–172.
https://doi.org/10.1016/j.jnoncrysol.2018.07.012.
[1]
Z. Chaker, A. Bouzid, B. Coasne, C. Massobrio, M. Boero, G. Ori, The structure and dipolar properties of CO2 adsorbed in a porous glassy chalcogel: Insights from first-principles molecular dynamics, Journal of Non-Crystalline Solids 498 (2018) 288–293.
https://doi.org/10.1016/j.jnoncrysol.2018.06.031.
[1]
G. Cotin, C. Kiefer, F. Perton, M. Boero, B. Özdamar, A. Bouzid, G. Ori, C. Massobrio, D. Begin, B. Pichon, D. Mertz, S. Bégin-Colin, Evaluating the Critical Roles of Precursor Nature and Water Content When Tailoring Magnetic Nanoparticles for Specific Applications, ACS Appl. Nano Mater. 1 (2018) 4306–4316.
https://doi.org/10.1021/acsanm.8b01123.
[1]
V. Iannuccelli, E. Maretti, A. Bellini, D. Malferrari, G. Ori, M. Montorsi, M. Bondi, E. Truzzi, E. Leo, Organo-modified bentonite for gentamicin topical application: Interlayer structure and in vivo skin permeation, Applied Clay Science 158 (2018) 158–168.
https://doi.org/10.1016/j.clay.2018.03.029.
[1]
K. Koizumi, H. Yoshida, M. Boero, K. Tamai, S. Hosokawa, T. Tanaka, K. Nobusada, M. Machida, A detailed insight into the catalytic reduction of NO operated by Cr-Cu nanostructures embedded in a CeO2 surface, Physical Chemistry Chemical Physics 20 (2018) 25592--25601.
https://doi.org/10.1039/c8cp04314k.
[1]
A. Kromik, E.V. Levchenko, C. Massobrio, A.V. Evteev, Diffusion in Ni–Zr Melts: Insights from Statistical Mechanics and Atomistic Modeling, Advanced Theory and Simulations 1 (2018) 1800109.
https://doi.org/10.1002/adts.201800109.
[1]
E. Martin, P.L. Palla, F. Cleri, A. Bouzid, G. Ori, S. Le Roux, M. Boero, C. Massobrio, On the occurrence of size effects in the calculation of thermal conductivity by first-principles molecular dynamics: The case of glassy GeTe4, Journal of Non-Crystalline Solids 498 (2018) 190–193.
https://doi.org/10.1016/j.jnoncrysol.2018.05.014.
[1]
C. Massobrio, E. Martin, Z. Chaker, M. Boero, A. Bouzid, S. Le Roux, G. Ori, Sensitivity to Dispersion Forces in First-Principles Modeling of Disordered Chalcogenides, Frontiers in Materials 5 (2018).
https://doi.org/10.3389/fmats.2018.00078.
[1]
B. Ozdamar, A. Bouzid, G. Ori, C. Massobrio, M. Boero, First-Principles Study of Dissociation Processes for the Synthesis of Fe and Co Oxide Nanoparticles, Journal of Chemical Theory and Computation 14 (2018) 225–235.
https://doi.org/10.1021/acs.jctc.7b00869.
1839302
NZSFH59F
2017
1
surface-science-reports
50
creator
asc
year
7054
https://www.ipcms.unistra.fr/plugins/zotpress/
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3Afalse%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%22P9Q8XMNM%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Amokrane%20et%20al.%22%2C%22parsedDate%22%3A%222017%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EA.%20Amokrane%2C%20S.%20Klyatskaya%2C%20M.%20Boero%2C%20M.%20Ruben%2C%20J.-P.%20Bucher%2C%20Role%20of%20pi-Radicals%20in%20the%20Spin%20Connectivity%20of%20Clusters%20and%20Networks%20of%20Tb%20Double-Decker%20Single%20Molecule%20Magnets%2C%20ACS%20Nano%2011%20%282017%29%2010750%26%23x2013%3B10760.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facsnano.7b05804%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facsnano.7b05804%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Role%20of%20pi-Radicals%20in%20the%20Spin%20Connectivity%20of%20Clusters%20and%20Networks%20of%20Tb%20Double-Decker%20Single%20Molecule%20Magnets%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Anis%22%2C%22lastName%22%3A%22Amokrane%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Svetlana%22%2C%22lastName%22%3A%22Klyatskaya%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mario%22%2C%22lastName%22%3A%22Ruben%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jean-Pierre%22%2C%22lastName%22%3A%22Bucher%22%7D%5D%2C%22abstractNote%22%3A%22When%20single%20molecule%20magnets%20%28SMMs%29%20self%20assemble%20into%202D%20networks%20on%20a%20surface%2C%20they%20interact%20via%20the%20pi-electrons%20of%20their%20ligands.%20This%20interaction%20is%20relevant%20to%20the%20quantum%20entanglement%20between%20molecular%20qubits%2C%20a%20key%20issue%20in%20quantum%20computing.%20Here%2C%20we%20examine%20the%20role%20played%20by%20the%20unpaired%20radical%20electron%20in%20the%20top%20ligand%20of%20Tb%20double-decker%20SMMs%20by%20comparing%20the%20spectroscopic%20features%20of%20isolated%20and%202D%20assembled%20entities%20on%20surfaces.%20High-resolution%20scanning%20tunneling%20microscopy%20%28STM%29%20is%20used%20to%20evidence%20experimentally%20the%20Kondo%20resonance%20of%20the%20unpaired%20radical%20spins%20in%20clusters%20and%20islands%20and%20its%20quenching%20due%20to%20up-pairing%20at%20orbital%20overlaps.%20The%20presence%20or%20the%20absence%20of%20the%20Kondo%20feature%20in%20the%20dI%5C%2FdV%20maps%20turns%20out%20to%20be%20a%20good%20measure%20of%20the%20lateral%20interaction%20between%20molecules%20in%202D%20networks.%20In%20a%202D%20cluster%20of%20molecules%2C%20the%20pr-orbital%20lobes%20that%20are%20linked%20through%20the%20orbital%20overlap%20show%20paired-up%20electron%20wave%20function%20%28one%20singly%20occupied%20molecular%20orbital%20%28SOMO%29%20with%20spin-up%20and%20the%20other%20with%20spin%20down%29%20and%20therefore%20do%20not%20experience%20the%20Kondo%20resonance%20in%20the%20experiment.%20As%20a%20result%2C%20small%20clusters%20built%20by%20STM-assisted%20manipulation%20of%20molecules%20show%20alternating%20Kondo%20features%20of%20quantum%20mechanical%20origin%2C%20from%20the%20monomer%20to%20the%20dimer%20and%20the%20trimer.%20On%20the%20other%20hand%2C%20when%20the%20TbPc2%20molecular%20clusters%20grow%20larger%20and%20form%20extended%20domains%2C%20a%20geometric%20rearrangement%20occurs%2C%20leading%20to%20the%20quenching%20of%20the%20Kondo%20signal%20on%20one%20lobe%20out%20of%20two.%20The%20even%20distribution%20of%20overlapping%20%28SOMO%29%20lobes%20on%20the%20perimeter%20of%20the%20molecule%20is%20induced%20by%20the%20square%20symmetry%20of%20the%20semi%20infinite%20lattice%20and%20clearly%20distinguishes%20the%20lattice%20from%20the%20clusters.%22%2C%22date%22%3A%222017%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Facsnano.7b05804%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Facsnano.7b05804%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221936-0851%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222018-05-30T15%3A26%3A38Z%22%7D%7D%2C%7B%22key%22%3A%227CPVZIJJ%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Bouzid%20et%20al.%22%2C%22parsedDate%22%3A%222017%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EA.%20Bouzid%2C%20H.%20Zaoui%2C%20P.L.%20Palla%2C%20G.%20Ori%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20F.%20Clerib%2C%20E.%20Lampin%2C%20Thermal%20conductivity%20of%20glassy%20GeTe4%20by%20first-principles%20molecular%20dynamics%2C%20Physical%20Chemistry%20Chemical%20Physics%2019%20%282017%29%209729%26%23x2013%3B9732.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc7cp01063j%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc7cp01063j%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Thermal%20conductivity%20of%20glassy%20GeTe4%20by%20first-principles%20molecular%20dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hayat%22%2C%22lastName%22%3A%22Zaoui%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Pier%20Luca%22%2C%22lastName%22%3A%22Palla%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fabrizio%22%2C%22lastName%22%3A%22Clerib%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Lampin%22%7D%5D%2C%22abstractNote%22%3A%22A%20transient%20thermal%20regime%20is%20achieved%20in%20glassy%20GeTe4%20by%20first-principles%20molecular%20dynamics%20following%20the%20recently%20proposed%20%60%27approach-to-equilibrium%5Cu201d%20methodology.%20The%20temporal%20and%20spatial%20evolution%20of%20the%20temperature%20do%20comply%20with%20the%20time-dependent%20solution%20of%20the%20heat%20equation.%20We%20demonstrate%20that%20the%20time%20scales%20required%20to%20create%20the%20hot%20and%20the%20cold%20parts%20of%20the%20system%20and%20observe%20the%20resulting%20approach%20to%20equilibrium%20are%20accessible%20to%20first-principles%20molecular%20dynamics.%20Such%20a%20strategy%20provides%20the%20thermal%20conductivity%20from%20the%20characteristic%20decay%20time.%20We%20rationalize%20in%20detail%20the%20impact%20on%20the%20thermal%20conductivity%20of%20the%20initial%20temperature%20difference%2C%20the%20equilibration%20duration%2C%20and%20the%20main%20simulation%20features.%22%2C%22date%22%3A%222017%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fc7cp01063j%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fc7cp01063j%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221463-9076%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222017-08-02T12%3A45%3A14Z%22%7D%7D%2C%7B%22key%22%3A%22TXDPQ97W%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Bouzid%20et%20al.%22%2C%22parsedDate%22%3A%222017%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EA.%20Bouzid%2C%20G.%20Ori%2C%20M.%20Boero%2C%20E.%20Lampin%2C%20C.%20Massobrio%2C%20Atomic-scale%20structure%20of%20the%20glassy%20Ge2Sb2Te5%20phase%20change%20material%3A%20A%20quantitative%20assessment%20via%20first-principles%20molecular%20dynamics%2C%20Physical%20Review%20B%2096%20%282017%29%20224204.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.96.224204%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.96.224204%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Atomic-scale%20structure%20of%20the%20glassy%20Ge2Sb2Te5%20phase%20change%20material%3A%20A%20quantitative%20assessment%20via%20first-principles%20molecular%20dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Lampin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22The%20amorphous%20structure%20of%20the%20phase%20change%20material%20Ge2Sb2Te5%20%28GST%29%20has%20been%20the%20object%20of%20controversial%20structural%20models.%20By%20employing%20first-principles%20molecular%20dynamics%20within%20density%20functional%20theory%2C%20we%20are%20able%20to%20obtain%20quantitative%20agreement%20with%20experimental%20structural%20findings%20for%20the%20topology%20of%20glassy%20GST.%20To%20this%20end%2C%20we%20take%20full%20advantage%20of%20a%20thoughtful%2C%20well%20established%20choice%20of%20the%20exchange-correlation%20%28XC%29%20functional%20%28Becke-Lee-Yang-Parr%2C%20BLYP%29%2C%20combined%20with%20appropriate%20options%20for%20the%20nonlocal%20part%20in%20the%20pseudopotential%20construction%20for%20Ge.%20Results%20obtained%20by%20using%20the%20Perdew-Burke-Ernzerhof%20%28PBE%29%20XC%20functional%20and%20a%20similar%20strategy%20for%20the%20Ge%20pseudopotential%20constructions%20are%20also%20presented%2C%20since%20they%20are%20very%20valuable%20and%20worthy%20of%20consideration.%20The%20atomic%20structure%20of%20glassy%20GST%20is%20characterized%20by%20Ge%20atoms%20lying%20in%20a%20predominant%20tetrahedral%20network%2C%20albeit%20a%20non-negligible%20fraction%20of%20Ge%20atoms%20are%20also%20found%20in%20defective%20octahedra.%22%2C%22date%22%3A%222017%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevB.96.224204%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevB.96.224204%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222469-9950%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222022-01-24T13%3A20%3A48Z%22%7D%7D%2C%7B%22key%22%3A%22H85S79H4%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Chai%20et%20al.%22%2C%22parsedDate%22%3A%222017%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EG.-L.%20Chai%2C%20M.%20Boero%2C%20Z.%20Hou%2C%20K.%20Terakura%2C%20W.%20Cheng%2C%20Indirect%20Four-Electron%20Oxygen%20Reduction%20Reaction%20on%20Carbon%20Materials%20Catalysts%20in%20Acidic%20Solutions%2C%20ACS%20Catalysis%207%20%282017%29%207908%26%23x2013%3B7916.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facscatal.7b02548%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facscatal.7b02548%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Indirect%20Four-Electron%20Oxygen%20Reduction%20Reaction%20on%20Carbon%20Materials%20Catalysts%20in%20Acidic%20Solutions%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guo-Liang%22%2C%22lastName%22%3A%22Chai%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Zhufeng%22%2C%22lastName%22%3A%22Hou%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kiyoyuki%22%2C%22lastName%22%3A%22Terakura%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Wendan%22%2C%22lastName%22%3A%22Cheng%22%7D%5D%2C%22abstractNote%22%3A%22Developing%20cost-effective%20and%20high-performance%20oxygen%20reduction%20reaction%20%28ORR%29%20catalysts%20is%20a%20fundamental%20issue%20in%20fuel%20cells%20and%20metal-air%20batteries.%20To%20this%20aim%2C%20carbon%20materials%20catalysts%20%28CMCs%29%20are%20extensively%20investigated%2C%20because%20of%20their%20performance%20comparable%20to%20noble-metal-based%20catalysts%20in%20alkaline%20solution.%20Yet%2C%20acidic%20solutions%20are%20desirable%20for%20an%20efficient%20proton%20exchange%20across%20Nafion%20membranes%20to%20yield%20high%20power%20density%20for%20commercial%20applications.%20However%2C%20the%20ORR%20performance%20of%20CMCs%20in%20acidic%20solutions%20is%20rather%20low%2C%20because%20of%20undesirable%20two-electron%20processes%20and%20OH%20radical%20formation.%20By%20using%20first-principles%20simulations%2C%20we%20elucidate%20the%20mechanisms%20and%20identify%20the%20active%20sites%20of%202e%28-%29%20ORR%20processes%20for%20indirect%204e%28-%29%20ORR%20We%20provide%20evidence%20for%20the%20fact%20that%20nitrogen-doped%20Stone%20Wales%20defects%20in%20graphene%20favor%20an%20indirect%20four-electron%20ORR%20upon%20H2O2%20formation%20and%20reduction.%20The%20low%20ORR%20potential%20for%20metal-free%20CMCs%20is%20ascribed%20to%20H2O2%20formation%20via%20hydrogen%20abstraction%20and%20the%20critical%20point%20for%20OH%20radical%20generation%20on%20transition-metal-based%20CMCs%20is%200.82%20V.%20Moreover%2C%20we%20provide%20an%20insight%20into%20the%20indirect%204e%28-%29%20ORR%2C%20which%20serves%20as%20a%20guide%20for%20suppressing%20undesired%202e%28-%29%20ORR%2C%20avoiding%20OH%20production%2C%20and%20promoting%20direct%20four%20electron%20ORR%20on%20CMCs.%20These%20results%20disclose%20a%20new%20strategy%20for%20developing%20high-efficiency%20ORR%20on%20CMCs%20in%20acidic%20solutions.%22%2C%22date%22%3A%222017%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Facscatal.7b02548%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Facscatal.7b02548%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222155-5435%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222018-05-30T12%3A35%3A43Z%22%7D%7D%2C%7B%22key%22%3A%223P3W8QUC%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Chaker%20et%20al.%22%2C%22parsedDate%22%3A%222017%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EZ.%20Chaker%2C%20G.%20Ori%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20The%20role%20of%202D%5C%2F3D%20spin-polarization%20interactions%20in%20hybrid%20copper%20hydroxide%20acetate%3A%20new%20insights%20from%20first-principles%20molecular%20dynamics%2C%20Beilstein%20Journal%20of%20Nanotechnology%208%20%282017%29%20857%26%23x2013%3B860.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3762%5C%2Fbjnano.8.86%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3762%5C%2Fbjnano.8.86%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22The%20role%20of%202D%5C%2F3D%20spin-polarization%20interactions%20in%20hybrid%20copper%20hydroxide%20acetate%3A%20new%20insights%20from%20first-principles%20molecular%20dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ziyad%22%2C%22lastName%22%3A%22Chaker%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22The%20magnetic%20properties%20response%20of%20the%20layered%20hybrid%20material%20copper%20hydroxide%20acetate%20Cu-2%28OH%29%283%29CH3COO%20center%20dot%20H2O%20is%20studied%20as%20a%20function%20of%20the%20applied%20pressure%20within%20first-principles%20molecular%20dynamics.%20We%20are%20able%20to%20elucidate%20the%20interplay%20between%20the%20structural%20properties%20of%20this%20material%20and%20its%20magnetic%20character%2C%20both%20at%20the%20local%20%28atomic%29%20level%20and%20at%20the%20bulk%20level.%20We%20performed%20a%20detailed%20analysis%20of%20the%20intralayer%20spin%20configurations%20occurring%20for%20each%20value%20of%20the%20imposed%20projection%20along%20the%20z-axis%20for%20the%20total%20spin%20and%20of%20the%20applied%20pressure.%20The%20transition%20from%20an%20antiferromagnetic%20to%20a%20ferromagnetic%20state%20at%20high%20pressure%20%28above%203%20GPa%29%20goes%20along%20with%20a%20vanishing%20difference%20between%20the%20spin%20polarizations%20pertaining%20to%20each%20layer.%20Therefore%2C%20at%20high%20pressure%2C%20copper%20hydroxide%20acetate%20is%20a%20ferromagnet%20with%20no%20changes%20of%20spin%20polarization%20in%20the%20direction%20perpendicular%20to%20the%20inorganic%20layers.%22%2C%22date%22%3A%222017%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.3762%5C%2Fbjnano.8.86%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.3762%5C%2Fbjnano.8.86%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222190-4286%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222017-09-21T09%3A19%3A18Z%22%7D%7D%2C%7B%22key%22%3A%227TAPDQ9X%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Evrard%20et%20al.%22%2C%22parsedDate%22%3A%222017%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EQ.%20Evrard%2C%20Z.%20Chaker%2C%20M.%20Roger%2C%20C.M.%20Sevrain%2C%20E.%20Delahaye%2C%20M.%20Gallart%2C%20P.%20Gilliot%2C%20C.%20Leuvrey%2C%20J.-M.%20Rueff%2C%20P.%20Rabu%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20A.%20Pautrat%2C%20P.-A.%20Jaffr%26%23xE8%3Bs%2C%20G.%20Ori%2C%20G.%20Rogez%2C%20Layered%20Simple%20Hydroxides%20Functionalized%20by%20Fluorene-Phosphonic%20Acids%3A%20Synthesis%2C%20Interface%20Theoretical%20Insights%2C%20and%20Magnetoelectric%20Effect%2C%20Advanced%20Functional%20Materials%2027%20%282017%29%201703576.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Fadfm.201703576%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Fadfm.201703576%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Layered%20Simple%20Hydroxides%20Functionalized%20by%20Fluorene-Phosphonic%20Acids%3A%20Synthesis%2C%20Interface%20Theoretical%20Insights%2C%20and%20Magnetoelectric%20Effect%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Quentin%22%2C%22lastName%22%3A%22Evrard%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ziyad%22%2C%22lastName%22%3A%22Chaker%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M%5Cu00e9lissa%22%2C%22lastName%22%3A%22Roger%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Charlotte%20M.%22%2C%22lastName%22%3A%22Sevrain%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Emilie%22%2C%22lastName%22%3A%22Delahaye%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mathieu%22%2C%22lastName%22%3A%22Gallart%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Pierre%22%2C%22lastName%22%3A%22Gilliot%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22C%5Cu00e9dric%22%2C%22lastName%22%3A%22Leuvrey%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jean-Michel%22%2C%22lastName%22%3A%22Rueff%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Pierre%22%2C%22lastName%22%3A%22Rabu%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Alain%22%2C%22lastName%22%3A%22Pautrat%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Paul-Alain%22%2C%22lastName%22%3A%22Jaffr%5Cu00e8s%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guillaume%22%2C%22lastName%22%3A%22Rogez%22%7D%5D%2C%22abstractNote%22%3A%22Copper-%20and%20cobalt-based%20layered%20simple%20hydroxides%20%28LSH%29%20are%20successfully%20functionalized%20by%20a%20series%20of%20fluorene%20mono-%20and%20diphosphonic%20acids%2C%20using%20anionic%20exchange%20reactions%20and%20a%20preintercalation%20strategy.%20The%20lateral%20functionalization%20of%20the%20fluorene%20moieties%20has%20only%20little%20impact%20on%20the%20overall%20structure%20of%20the%20obtained%20layered%20hybrid%20materials%20but%20it%20influences%20the%20organization%20of%20the%20molecules%20within%20the%20interlamellar%20spacing.%20For%20bulky%20fluorene%20%289%2C9-dioctyl%20derivative%29%2C%20luminescence%20is%20preserved%20when%20inserted%20into%20copper%20and%20cobalt%20hydroxydes%2C%20whereas%20it%20is%20completely%20quenched%20for%20the%20other%20fluorenes.%20Detailed%20characterization%20of%20the%20internal%20structure%20and%20chemical%20bonding%20properties%20for%20copper-%20and%20cobalt-based%20hybrids%20is%20performed%20via%20ancillary%20experimental%20techniques.%20For%20the%20copper-based%20LSH%20class%2C%20for%20which%20more%20elusive%20findings%20are%20found%2C%20first-principles%20molecular%20dynamics%20simulations%20unravel%20the%20fundamental%20stabilizing%20role%20of%20the%20H-bonding%20network%20promoted%20within%20the%20local%20environments%20of%20the%20fluorene%20mono-%20and%20diphosphonic%20acids.%20The%20cobalt%20series%20of%20compounds%20constitute%20a%20new%20class%20of%20hybrid%20magnets%2C%20with%20ordering%20temperatures%20ranging%20from%2011.8%20to%2017.8%20K%20and%20show%20a%20clear%20magnetoelectric%20effect.%20This%20effect%20appears%20above%20a%20threshold%20magnetic%20field%2C%20which%20is%20null%20below%20the%20magnetic%20ordering%20temperature%2C%20and%20it%20persists%20in%20the%20paramagnetic%20regime%20till%20about%20110%20K.%22%2C%22date%22%3A%222017%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1002%5C%2Fadfm.201703576%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1002%5C%2Fadfm.201703576%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221616-3028%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%226IWM732K%22%2C%22CF4ZI7HM%22%2C%22CHW2VGSR%22%2C%22HVHG5Z72%22%2C%22M244N6AF%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222018-05-30T12%3A43%3A53Z%22%7D%7D%2C%7B%22key%22%3A%22I5E44CDP%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Gentili%20et%20al.%22%2C%22parsedDate%22%3A%222017%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ED.%20Gentili%2C%20G.%20Ori%2C%20L.%20Ortolani%2C%20V.%20Morandi%2C%20M.%20Cavallini%2C%20Cooperative%20and%20Reversible%20Anisotropic%20Assembly%20of%20Gold%20Nanoparticles%20by%20Modulation%20of%20Noncovalent%20Interparticle%20Interactions%2C%20ChemNanoMat%203%20%282017%29%20874%26%23x2013%3B878.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Fcnma.201700212%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Fcnma.201700212%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Cooperative%20and%20Reversible%20Anisotropic%20Assembly%20of%20Gold%20Nanoparticles%20by%20Modulation%20of%20Noncovalent%20Interparticle%20Interactions%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Denis%22%2C%22lastName%22%3A%22Gentili%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Luca%22%2C%22lastName%22%3A%22Ortolani%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Vittorio%22%2C%22lastName%22%3A%22Morandi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Massimiliano%22%2C%22lastName%22%3A%22Cavallini%22%7D%5D%2C%22abstractNote%22%3A%22Controlled%20and%20reversible%20anisotropic%20self-assembly%20of%20gold%20nanoparticles%20%28GNPs%29%20is%20achieved%20by%20ligand%20exchange%20with%20concentrations%20of%20ligand%20below%20that%20needed%20for%20their%20complete%20capping%2C%20that%20is%2C%20using%20substoichiometric%20ligand%20coverages.%20A%20thoughtful%20rationale%20of%20the%20ligand%20chemical%20structure%20and%20control%20over%20exchange%20conditions%20have%20been%20pursued%20to%20foster%20a%20reversible%20process%20grounded%20on%20the%20cooperative%20balance%20between%20attractive%2C%20and%20repulsive%20interactions.%20The%20gradual%20replacement%20of%20the%20electrostatic%20and%20steric%20repulsions%20between%20the%20GNPs%20with%20attractive%20dipolar%20interactions%20and%20enhanced%20steric%20repulsions%20is%20accomplished%20by%20ligand%20exchange%20with%20an%20oligo%20%28ethylene%20glycol%29%20alkanethiol.%20The%20modulation%20of%20interparticle%20interactions%20is%20driven%20by%20the%20extent%20of%20ligand%20substitution%20and%20allows%20the%20reversible%20assembly%20of%20nanoparticles%20and%20tuning%20of%20their%20optical%20properties.%22%2C%22date%22%3A%222017%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1002%5C%2Fcnma.201700212%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1002%5C%2Fcnma.201700212%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222199-692X%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222022-01-24T13%3A22%3A28Z%22%7D%7D%2C%7B%22key%22%3A%224369AQ9B%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Imoto%20et%20al.%22%2C%22parsedDate%22%3A%222017%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EF.%20Imoto%2C%20J.-I.%20Iwata%2C%20M.%20Boero%2C%20A.%20Oshiyama%2C%20Microscopic%20Mechanisms%20of%20Initial%20Formation%20Process%20of%20Graphene%20on%20SiC%280001%29%20Surfaces%3A%20Selective%20Si%20Desorption%20from%20Step%20Edges%2C%20Journal%20of%20Physical%20Chemistry%20C%20121%20%282017%29%205041%26%23x2013%3B5049.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpcc.6b11985%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpcc.6b11985%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Microscopic%20Mechanisms%20of%20Initial%20Formation%20Process%20of%20Graphene%20on%20SiC%280001%29%20Surfaces%3A%20Selective%20Si%20Desorption%20from%20Step%20Edges%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fumihiro%22%2C%22lastName%22%3A%22Imoto%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jun-Ichi%22%2C%22lastName%22%3A%22Iwata%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Atsushi%22%2C%22lastName%22%3A%22Oshiyama%22%7D%5D%2C%22abstractNote%22%3A%22Simulations%20within%20the%20density%20functional%20theory%20framework%20clarify%20the%20microscopic%20mechanism%20responsible%20for%20the%20initial%20stage%20of%20graphene%20formation%20on%20the%20SiC%280001%29%20surface.%20Favorable%20reaction%20pathways%20for%20the%20desorption%20of%20either%20Si%20or%20C%20atoms%20from%20the%20stepped%20surface%20have%20been%20found%20by%20determining%20the%20desorption%20and%20the%20subsequent%20migration%20pathways%2C%20quantified%20in%20terms%20of%20the%20corresponding%20energy%20barriers%20for%20the%20first%20time.%20We%20find%20that%20the%20energy%20barrier%20for%20the%20desorption%20of%20an%20Si%20atom%20at%20the%20step%20edge%20and%20the%20subsequent%20migration%20toward%20stable%20terrace%20sites%20is%20lower%20than%20that%20of%20a%20C%20atom%20by%200.75%20eV%2C%20indicative%20of%20the%20selective%20desorption%20of%20Si%20from%20the%20SiC%20surface.%20We%20also%20find%20that%20the%20subsequent%20Si%20desorption%20is%20an%20exothermic%20reaction.%20This%20exotherrnicity%20comes%20from%20the%20energy%20gain%20due%20to%20the%20bond%20formation%20of%20C%20atoms%20being%20left%20near%20the%20step%20edges.%20This%20is%20likely%20to%20be%20a%20seed%20of%20graphene%20flakes.%22%2C%22date%22%3A%222017%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Facs.jpcc.6b11985%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Facs.jpcc.6b11985%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221932-7447%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222017-05-09T13%3A32%3A04Z%22%7D%7D%2C%7B%22key%22%3A%22968P4ZVQ%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Koizumi%20et%20al.%22%2C%22parsedDate%22%3A%222017%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Koizumi%2C%20K.%20Nobusada%2C%20M.%20Boero%2C%20Simple%20but%20Efficient%20Method%20for%20Inhibiting%20Sintering%20and%20Aggregation%20of%20Catalytic%20Pt%20Nanoclusters%20on%20Metal-Oxide%20Supports%2C%20Chemistry-a%20European%20Journal%2023%20%282017%29%201531%26%23x2013%3B1538.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Fchem.201604188%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Fchem.201604188%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Simple%20but%20Efficient%20Method%20for%20Inhibiting%20Sintering%20and%20Aggregation%20of%20Catalytic%20Pt%20Nanoclusters%20on%20Metal-Oxide%20Supports%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenichi%22%2C%22lastName%22%3A%22Koizumi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Katsuyuki%22%2C%22lastName%22%3A%22Nobusada%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22A%20simple%20and%20efficient%20method%20to%20inhibit%20aggregation%20of%20Pt%20clusters%20supported%20on%20metal%20oxide%20was%20developed%2C%20preserving%20the%20accessible%20clusters%20surface%20where%20catalytically%20active%20sites%20are%20located%20even%20at%20relatively%20high%20temperatures%20up%20to%20700%20K.%20The%20key%20idea%20was%20the%20inclusion%20of%20transition%20metal%20atoms%20such%20as%20Ni%20into%20the%20Pt%20clusters%2C%20thus%20anchoring%20the%20clusters%20through%20formation%20of%20strong%20chemical%20bonds%20with%20oxygen%20atoms%20of%20the%20metal-oxide%20support.%20To%20elucidate%20the%20efficiency%20of%20the%20method%2C%20first-principles%20molecular%20dynamics%20enhanced%20with%20free-energy%20sampling%20methods%20were%20used.%20These%20virtual%20experiments%20showed%20how%20doped%20Ni%20atoms%2C%20having%20a%20stronger%20affinity%20to%20O%20than%20Pt%2C%20anchor%20the%20Pt%20clusters%20tightly%20to%20the%20metal-oxide%20supports%20and%20inhibit%20their%20tendency%20to%20aggregate%20on%20the%20support.%22%2C%22date%22%3A%222017%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1002%5C%2Fchem.201604188%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1002%5C%2Fchem.201604188%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220947-6539%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222017-04-03T15%3A06%3A06Z%22%7D%7D%2C%7B%22key%22%3A%22SSUPDRVD%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Koizumi%20et%20al.%22%2C%22parsedDate%22%3A%222017%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Koizumi%2C%20M.%20Hatakeyama%2C%20M.%20Boero%2C%20K.%20Nobusada%2C%20H.%20Hori%2C%20T.%20Misonou%2C%20S.%20Nakamura%2C%20How%20seaweeds%20release%20the%20excess%20energy%20from%20sunlight%20to%20surrounding%20sea%20water%2C%20Physical%20Chemistry%20Chemical%20Physics%2019%20%282017%29%2015745%26%23x2013%3B15753.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc7cp02699d%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc7cp02699d%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22How%20seaweeds%20release%20the%20excess%20energy%20from%20sunlight%20to%20surrounding%20sea%20water%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenichi%22%2C%22lastName%22%3A%22Koizumi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Makoto%22%2C%22lastName%22%3A%22Hatakeyama%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Katsuyuki%22%2C%22lastName%22%3A%22Nobusada%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hirokazu%22%2C%22lastName%22%3A%22Hori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Taku%22%2C%22lastName%22%3A%22Misonou%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Shinichiro%22%2C%22lastName%22%3A%22Nakamura%22%7D%5D%2C%22abstractNote%22%3A%22We%20report%20an%20atomistic%20insight%20into%20the%20mechanism%20regulating%20the%20energy%20released%20by%20a%20porphyra-334%20molecule%2C%20the%20ubiquitous%20photosensitive%20component%20of%20marine%20algae%2C%20in%20a%20liquid%20water%20environment%20upon%20an%20electron%20excitation.%20To%20quantify%20this%20rapidly%20occurring%20process%2C%20we%20resort%20to%20the%20Fourier%20analysis%20of%20the%20mass-weighted%20auto-correlation%20function%2C%20providing%20evidence%20for%20a%20remarkable%20dynamic%20change%20in%20the%20number%20of%20hydrogen%20bonds%20among%20water%20molecules%20and%20between%20the%20porphyra-334%20and%20its%20surrounding%20hydrating%20water.%20Hydrogen%20bonds%20between%20the%20porphyra-334%20and%20close%20by%20water%20molecules%20can%20act%20directly%20and%20rather%20easily%20to%20promote%20an%20efficient%20transfer%20of%20the%20excess%20kinetic%20energies%20of%20the%20porphyra-334%20to%20the%20surrounding%20solvating%20water%20molecules%20via%20an%20activation%20of%20the%20collective%20modes%20identified%20as%20hydrogen-bond%20stretching%20modes%20in%20liquid%20water%20which%20eventually%20results%20in%20a%20disruption%20of%20the%20hydrogen%20bond%20network.%20Since%20porphyra-334%20is%20present%20in%20seaweeds%2C%20aquatic%20cyanobacteria%20%28blue-green%20algae%29%20and%20red%20algae%2C%20our%20findings%20allow%20addressing%20the%20question%20how%20algae%20in%20oceans%20or%20lakes%2C%20upon%20sunlight%20absorption%2C%20can%20release%20large%20amounts%20of%20energy%20into%20surrounding%20water%20without%20destabilizing%20neither%20their%20own%20nor%20the%20H2O%20molecular%20structure.%22%2C%22date%22%3A%222017%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fc7cp02699d%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fc7cp02699d%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221463-9076%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222017-08-03T09%3A24%3A21Z%22%7D%7D%2C%7B%22key%22%3A%22S8RSGX4P%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Koizumi%20et%20al.%22%2C%22parsedDate%22%3A%222017%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Koizumi%2C%20K.%20Nobusada%2C%20M.%20Boero%2C%20An%20atomic-level%20insight%20into%20the%20basic%20mechanism%20responsible%20for%20the%20enhancement%20of%20the%20catalytic%20oxidation%20of%20carbon%20monoxide%20on%20a%20Cu%5C%2FCeO2%20surface%2C%20Physical%20Chemistry%20Chemical%20Physics%2019%20%282017%29%203498%26%23x2013%3B3505.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc6cp05957k%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc6cp05957k%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22An%20atomic-level%20insight%20into%20the%20basic%20mechanism%20responsible%20for%20the%20enhancement%20of%20the%20catalytic%20oxidation%20of%20carbon%20monoxide%20on%20a%20Cu%5C%2FCeO2%20surface%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenichi%22%2C%22lastName%22%3A%22Koizumi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Katsuyuki%22%2C%22lastName%22%3A%22Nobusada%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22The%20reaction%20mechanisms%20of%20CO%20molecules%20interacting%20with%20a%20Cu%5C%2FCeO2%20surface%20and%20related%20morphological%20modifications%20occurring%20upon%20removal%20of%20O%20atoms%20to%20generate%20CO2%20are%20investigated%20by%20first-principles%20dynamical%20simulations%20complemented%20by%20a%20free-energy%20sampling%20technique.%20We%20show%20that%20the%20reactivity%20of%20oxygen%20atoms%20located%20in%20the%20first%20layer%20in%20the%20vicinity%20of%20the%20Cu%20site%20is%20remarkably%20high%20because%20of%20a%20reduction%20of%20the%20O%20coordination%20number.%20Moreover%2C%20we%20evidence%20that%20the%20doped%20Cu%20atoms%20are%20responsible%20for%20an%20enhancement%20of%20the%20exposure%20of%20other%20surrounding%20O%20atoms%2C%20even%20below%20the%20first%20surface%20layer%2C%20which%20can%20then%20easily%20react%20with%20CO%20and%20are%20gradually%20removed%20from%20the%20system%20in%20the%20oxidation%20process.%20The%20underlying%20mechanism%20responsible%20for%20such%20a%20high%20catalytic%20reactivity%20of%20the%20Cu%5C%2FCeO2%20surface%20toward%20CO%20oxidation%20is%20rationalized%20in%20terms%20of%20the%20characteristics%20of%20the%20doped%20Cu.%20In%20fact%2C%20this%20copper%20site%20is%20responsible%20for%20providing%20an%20increasing%20number%20of%20O%20atoms%20participating%20in%20the%20catalysis%20by%20exposing%20subsequently%20all%20O%20atoms%20in%20the%20vicinity%20which%20are%20likely%20to%20react%20with%20an%20approaching%20CO.%20This%20peculiarity%20of%20the%20Cu%20atoms%20extends%20to%20O%20atoms%20which%20initially%20can%20be%20deeply%20buried%20up%20to%20the%20fourth%20layer%20underneath%20the%20surface.%20The%20mechanism%20unveiled%20here%20provides%20useful%20insights%20into%20the%20fundamental%20mechanism%20and%20suggests%20strategies%20for%20the%20engineering%20and%20design%20of%20more%20effective%20ceria-based%20catalysts%20via%20metal%20doping.%22%2C%22date%22%3A%222017%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fc6cp05957k%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fc6cp05957k%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221463-9076%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222022-01-24T13%3A24%3A35Z%22%7D%7D%2C%7B%22key%22%3A%226PUVDCXX%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Lampin%20et%20al.%22%2C%22parsedDate%22%3A%222017%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EE.%20Lampin%2C%20A.%20Bouzid%2C%20G.%20Ori%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20Impact%20of%20dispersion%20forces%20on%20the%20atomic%20structure%20of%20a%20prototypical%20network-forming%20disordered%20system%3A%20The%20case%20of%20liquid%20GeSe2%2C%20Journal%20of%20Chemical%20Physics%20147%20%282017%29%20044504.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4986166%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4986166%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Impact%20of%20dispersion%20forces%20on%20the%20atomic%20structure%20of%20a%20prototypical%20network-forming%20disordered%20system%3A%20The%20case%20of%20liquid%20GeSe2%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evelyne%22%2C%22lastName%22%3A%22Lampin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22Aset%20of%20structural%20properties%20of%20liquid%20GeSe2%20are%20calculated%20by%20using%20first-principles%20molecular%20dynamics%20and%20including%2C%20for%20the%20first%20time%2C%20van%20derWaals%20dispersion%20forces.%20None%20of%20the%20numerous%20atomic-scale%20simulations%20performed%20in%20the%20past%20on%20this%20prototypical%20disordered%20network-forming%20material%20had%20ever%20accounted%20for%20dispersion%20forces%20in%20the%20expression%20of%20the%20total%20energy.%20For%20this%20purpose%2C%20we%20employed%20either%20the%20Grimme-D2%20or%20the%20maximally%20localized%20Wannier%20function%20scheme.%20We%20assessed%20the%20impact%20of%20dispersion%20forces%20on%20properties%20such%20as%20partial%20structure%20factors%2C%20pair%20correlation%20functions%2C%20bond%20angle%20distribution%2C%20and%20number%20of%20corner%20vs%20edge%20sharing%20connections.%20The%20maximally%20localized%20Wannier%20function%20scheme%20is%20more%20reliable%20than%20the%20Grimme-D2%20scheme%20in%20reproducing%20existing%20first-principles%20results.%20In%20particular%2C%20the%20Grimme-D2%20scheme%20worsens%20the%20agreement%20with%20experiments%20in%20the%20case%20of%20the%20Ge-Ge%20pair%20correlation%20function.%20Our%20study%20shows%20that%20the%20impact%20of%20dispersion%20forces%20on%20disordered%20chalcogenides%20has%20to%20be%20considered%20with%20great%20care%20since%20it%20cannot%20be%20necessarily%20the%20same%20when%20adopting%20different%20recipes.%20Published%20by%20AIP%20Publishing.%22%2C%22date%22%3A%222017%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1063%5C%2F1.4986166%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1063%5C%2F1.4986166%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220021-9606%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222018-05-30T12%3A30%3A33Z%22%7D%7D%2C%7B%22key%22%3A%226UCVA2ZS%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Li%20et%20al.%22%2C%22parsedDate%22%3A%222017%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EH.%20Li%2C%20Y.-I.%20Matsushita%2C%20M.%20Boero%2C%20A.%20Oshiyama%2C%20First-Principles%20Calculations%20That%20Clarify%20Energetics%20and%20Reactions%20of%20Oxygen%20Adsorption%20and%20Carbon%20Desorption%20on%204H-SiC%20%2811%282%29over-bar0%29%20Surface%2C%20Journal%20of%20Physical%20Chemistry%20C%20121%20%282017%29%203920%26%23x2013%3B3928.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpcc.6b11942%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpcc.6b11942%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22First-Principles%20Calculations%20That%20Clarify%20Energetics%20and%20Reactions%20of%20Oxygen%20Adsorption%20and%20Carbon%20Desorption%20on%204H-SiC%20%2811%282%29over-bar0%29%20Surface%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Han%22%2C%22lastName%22%3A%22Li%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yu-Ichiro%22%2C%22lastName%22%3A%22Matsushita%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Atsushi%22%2C%22lastName%22%3A%22Oshiyama%22%7D%5D%2C%22abstractNote%22%3A%22We%20report%20static%20and%20dynamic%20first-principles%20calculations%20that%20provide%20atomistic%20pictures%20of%20the%20initial%20stage%20of%20the%20oxidation%20processes%20occurring%20at%20the%20%2811%20%282%29%20over%20bar0%29%20surface%20of%204H-SiC.%20Our%20results%20reveal%20reaction%20pathways%20and%20their%20associated%20free-energy%20barriers%20for%20the%20adsorption%20of%20oxygen%20and%20the%20desorption%20of%20carbon%20atoms.%20We%20find%20that%20oxygen%20adsorption%20shows%20structural%20multistability%20and%20that%20the%20surface-bridge%20sites%20are%20the%20most%20stable%20and%20crucial%20sites%20for%20subsequent%20oxidation.%20We%20find%20that%20an%20approaching%2002%20molecule%20is%20adsorbed%2C%20then%20is%20dissociated%2C%20and%20finally%20migrates%20toward%20these%20surface%20bridge%20sites%20with%20a%20free-energy%20barrier%20of%200.7%20eV%20at%20the%20%2811%20%282%29%20over%20bar0%29%20surface.%20We%20also%20find%20that%20a%20CO%20molecule%20is%20desorbed%20from%20the%20metastable%20oxidized%20structure%20upon%20overcoming%20a%20free-energy%20barrier%20of%202.4-2.6%20eV%2C%20thus%20constituting%20one%20of%20the%20annihilation%20processes%20of%20C%20during%20the%20oxidation.%20The%20results%20of%20the%20CO%20molecule%20desorption%20on%20the%20%2811%20%282%29%20over%20bar0%29%20surface%20are%20compared%20with%20that%20of%20the%20%28000%20%281%29%20over%20bar%29%20surface.%20A%20catalytic%20effect%20of%20dangling%20bonds%20at%20the%20surface%2C%20causing%20a%20drastic%20reduction%20of%20the%20CO%20desorption%20energy%2C%20is%20found%20on%20the%20%28000%20%281%29%20over%20bar%29%20surface%2C%20and%20the%20microscopic%20picture%20of%20the%20effect%20is%20ascribed%20to%20an%20electron%20transfer%20from%20the%20Si%20to%20C%20dangling%20bonds.%20The%20intrinsic%20%2811%20%282%29%20over%20bar0%29%20surface%20does%20not%20show%20this%20catalytic%20effect%3B%20this%20is%20because%20the%20surface%20consists%20of%20an%20equal%20amount%20of%20Si%20and%20C%20dangling%20bonds%20and%20the%20electron%20transfer%20occurs%20before%20the%20desorption.%22%2C%22date%22%3A%222017%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Facs.jpcc.6b11942%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Facs.jpcc.6b11942%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221932-7447%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222017-04-03T15%3A06%3A16Z%22%7D%7D%5D%7D
[1]
A. Amokrane, S. Klyatskaya, M. Boero, M. Ruben, J.-P. Bucher, Role of pi-Radicals in the Spin Connectivity of Clusters and Networks of Tb Double-Decker Single Molecule Magnets, ACS Nano 11 (2017) 10750–10760.
https://doi.org/10.1021/acsnano.7b05804.
[1]
A. Bouzid, H. Zaoui, P.L. Palla, G. Ori, M. Boero, C. Massobrio, F. Clerib, E. Lampin, Thermal conductivity of glassy GeTe4 by first-principles molecular dynamics, Physical Chemistry Chemical Physics 19 (2017) 9729–9732.
https://doi.org/10.1039/c7cp01063j.
[1]
A. Bouzid, G. Ori, M. Boero, E. Lampin, C. Massobrio, Atomic-scale structure of the glassy Ge2Sb2Te5 phase change material: A quantitative assessment via first-principles molecular dynamics, Physical Review B 96 (2017) 224204.
https://doi.org/10.1103/PhysRevB.96.224204.
[1]
G.-L. Chai, M. Boero, Z. Hou, K. Terakura, W. Cheng, Indirect Four-Electron Oxygen Reduction Reaction on Carbon Materials Catalysts in Acidic Solutions, ACS Catalysis 7 (2017) 7908–7916.
https://doi.org/10.1021/acscatal.7b02548.
[1]
Z. Chaker, G. Ori, M. Boero, C. Massobrio, The role of 2D/3D spin-polarization interactions in hybrid copper hydroxide acetate: new insights from first-principles molecular dynamics, Beilstein Journal of Nanotechnology 8 (2017) 857–860.
https://doi.org/10.3762/bjnano.8.86.
[1]
Q. Evrard, Z. Chaker, M. Roger, C.M. Sevrain, E. Delahaye, M. Gallart, P. Gilliot, C. Leuvrey, J.-M. Rueff, P. Rabu, C. Massobrio, M. Boero, A. Pautrat, P.-A. Jaffrès, G. Ori, G. Rogez, Layered Simple Hydroxides Functionalized by Fluorene-Phosphonic Acids: Synthesis, Interface Theoretical Insights, and Magnetoelectric Effect, Advanced Functional Materials 27 (2017) 1703576.
https://doi.org/10.1002/adfm.201703576.
[1]
D. Gentili, G. Ori, L. Ortolani, V. Morandi, M. Cavallini, Cooperative and Reversible Anisotropic Assembly of Gold Nanoparticles by Modulation of Noncovalent Interparticle Interactions, ChemNanoMat 3 (2017) 874–878.
https://doi.org/10.1002/cnma.201700212.
[1]
F. Imoto, J.-I. Iwata, M. Boero, A. Oshiyama, Microscopic Mechanisms of Initial Formation Process of Graphene on SiC(0001) Surfaces: Selective Si Desorption from Step Edges, Journal of Physical Chemistry C 121 (2017) 5041–5049.
https://doi.org/10.1021/acs.jpcc.6b11985.
[1]
K. Koizumi, K. Nobusada, M. Boero, Simple but Efficient Method for Inhibiting Sintering and Aggregation of Catalytic Pt Nanoclusters on Metal-Oxide Supports, Chemistry-a European Journal 23 (2017) 1531–1538.
https://doi.org/10.1002/chem.201604188.
[1]
K. Koizumi, M. Hatakeyama, M. Boero, K. Nobusada, H. Hori, T. Misonou, S. Nakamura, How seaweeds release the excess energy from sunlight to surrounding sea water, Physical Chemistry Chemical Physics 19 (2017) 15745–15753.
https://doi.org/10.1039/c7cp02699d.
[1]
K. Koizumi, K. Nobusada, M. Boero, An atomic-level insight into the basic mechanism responsible for the enhancement of the catalytic oxidation of carbon monoxide on a Cu/CeO2 surface, Physical Chemistry Chemical Physics 19 (2017) 3498–3505.
https://doi.org/10.1039/c6cp05957k.
[1]
E. Lampin, A. Bouzid, G. Ori, M. Boero, C. Massobrio, Impact of dispersion forces on the atomic structure of a prototypical network-forming disordered system: The case of liquid GeSe2, Journal of Chemical Physics 147 (2017) 044504.
https://doi.org/10.1063/1.4986166.
[1]
H. Li, Y.-I. Matsushita, M. Boero, A. Oshiyama, First-Principles Calculations That Clarify Energetics and Reactions of Oxygen Adsorption and Carbon Desorption on 4H-SiC (11(2)over-bar0) Surface, Journal of Physical Chemistry C 121 (2017) 3920–3928.
https://doi.org/10.1021/acs.jpcc.6b11942.
1839302
NZSFH59F
2016
1
surface-science-reports
50
creator
asc
year
7054
https://www.ipcms.unistra.fr/plugins/zotpress/
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3Afalse%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%22QS7N9MZB%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Bouzid%20et%20al.%22%2C%22parsedDate%22%3A%222016%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EA.%20Bouzid%2C%20K.J.%20Pizzey%2C%20A.%20Zeidler%2C%20G.%20Ori%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20S.%20Klotz%2C%20H.E.%20Fischer%2C%20C.L.%20Bull%2C%20P.S.%20Salmon%2C%20Pressure-induced%20structural%20changes%20in%20the%20network-forming%20isostatic%20glass%20GeSe4%3A%20An%20investigation%20by%20neutron%20diffraction%20and%20first-principles%20molecular%20dynamics%2C%20Physical%20Review%20B%2093%20%282016%29%20014202.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.93.014202%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.93.014202%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Pressure-induced%20structural%20changes%20in%20the%20network-forming%20isostatic%20glass%20GeSe4%3A%20An%20investigation%20by%20neutron%20diffraction%20and%20first-principles%20molecular%20dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Keiron%20J.%22%2C%22lastName%22%3A%22Pizzey%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Anita%22%2C%22lastName%22%3A%22Zeidler%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Stefan%22%2C%22lastName%22%3A%22Klotz%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Henry%20E.%22%2C%22lastName%22%3A%22Fischer%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Craig%20L.%22%2C%22lastName%22%3A%22Bull%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Philip%20S.%22%2C%22lastName%22%3A%22Salmon%22%7D%5D%2C%22abstractNote%22%3A%22The%20changes%20to%20the%20topological%20and%20chemical%20ordering%20in%20the%20network-forming%20isostatic%20glass%20GeSe4%20are%20investigated%20at%20pressures%20up%20to%20similar%20to%2014.4%20GPa%20by%20using%20a%20combination%20of%20neutron%20diffraction%20and%20first-principles%20molecular%20dynamics.%20The%20results%20show%20a%20network%20built%20from%20corner-and%20edge-sharing%20Ge%28Se-1%5C%2F2%29%284%29%20tetrahedra%2C%20where%20linkages%20by%20Se-2%20dimers%20or%20longer%20Se-n%20chains%20are%20prevalent.%20These%20linkages%20confer%20the%20network%20with%20a%20local%20flexibility%20that%20helps%20to%20retain%20the%20network%20connectivity%20at%20pressures%20up%20to%20similar%20to%208%20GPa%2C%20corresponding%20to%20a%20density%20increase%20of%20similar%20to%2037%25.%20The%20network%20reorganization%20at%20constant%20topology%20maintains%20a%20mean%20coordination%20number%20%28n%29%20over%20bar%20similar%20or%20equal%20to%202.4%2C%20the%20value%20expected%20from%20mean-field%20constraint-counting%20theory%20for%20a%20rigid%20stress-free%20network.%20Isostatic%20networks%20may%20therefore%20remain%20optimally%20constrained%20to%20avoid%20stress%20and%20retain%20their%20favorable%20glass-forming%20ability%20over%20a%20large%20density%20range.%20As%20the%20pressure%20is%20increased%20to%20around%2013%20GPa%2C%20corresponding%20to%20a%20density%20increase%20of%20similar%20to%2049%25%2C%20Ge%28Se-1%5C%2F2%29%284%29%20tetrahedra%20remain%20as%20the%20predominant%20structural%20motifs%2C%20but%20there%20is%20an%20appearance%20of%205-fold%20coordinated%20Ge%20atoms%20and%20homopolar%20Ge-Ge%20bonds%20that%20accompany%20an%20increase%20in%20the%20fraction%20of%203-fold%20coordinated%20Se%20atoms.%20The%20band%20gap%20energy%20decreases%20with%20increasing%20pressure%2C%20and%20midgap%20states%20appear%20at%20pressures%20beyond%20similar%20to%206.7%20GPa.%20The%20latter%20originate%20from%20undercoordinated%20Se%20atoms%20that%20terminate%20broken%20Se-n%20chains.%22%2C%22date%22%3A%222016%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevB.93.014202%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevB.93.014202%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221098-0121%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222016-02-10T14%3A14%3A47Z%22%7D%7D%2C%7B%22key%22%3A%22383N2HGW%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Koizumi%20et%20al.%22%2C%22parsedDate%22%3A%222016%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Koizumi%2C%20K.%20Nobusada%2C%20M.%20Boero%2C%20The%20absence%20of%20a%20gap%20state%20and%20enhancement%20of%20the%20Mars-van%20Krevelen%20reaction%20on%20oxygen%20defective%20Cu%5C%2FCeO2%20surfaces%2C%20Physical%20Chemistry%20Chemical%20Physics%2018%20%282016%29%2020708%26%23x2013%3B20712.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc6cp03880h%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc6cp03880h%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22The%20absence%20of%20a%20gap%20state%20and%20enhancement%20of%20the%20Mars-van%20Krevelen%20reaction%20on%20oxygen%20defective%20Cu%5C%2FCeO2%20surfaces%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenichi%22%2C%22lastName%22%3A%22Koizumi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Katsuyuki%22%2C%22lastName%22%3A%22Nobusada%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22We%20report%20a%20detailed%20first-principles%20analysis%20of%20the%20electronic%20structures%20of%20oxygen%20defective%20CeO2%20and%20Cu%5C%2FCeO2%20surfaces%20aimed%20at%20elucidating%20the%20disappearance%20of%20the%20gap%20state%20of%20defective%20CeO2%20when%20a%20Cu%20atom%20is%20added%20at%20the%20surface.%20The%20top%20of%20the%20valence%20band%20of%20Cu%5C%2FCeO2%20originates%20from%20the%20O%202p%20states%20around%20this%20Cu%20atom.%20We%20show%20that%20this%20redistribution%20of%20electronic%20states%20at%20the%20defective%20Cu%5C%2FCeO2%20surface%20enhances%20the%20reactivity%20of%20the%20surface%20O%20atoms.%20Indeed%2C%20dynamical%20simulations%20show%20an%20acceleration%20of%20catalytic%20NO%20oxidation%20occurring%20via%20the%20Mars-van%20Krevelen%20mechanism%20mediated%20by%20these%20highly%20reactive%20oxygens.%22%2C%22date%22%3A%222016%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fc6cp03880h%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fc6cp03880h%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221463-9076%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222016-11-17T09%3A58%3A35Z%22%7D%7D%2C%7B%22key%22%3A%22DFDR69FB%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Koizumi%20et%20al.%22%2C%22parsedDate%22%3A%222016%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Koizumi%2C%20K.%20Nobusada%2C%20M.%20Boero%2C%20Reducing%20the%20Cost%20and%20Preserving%20the%20Reactivity%20in%20Noble-Metal-Based%20Catalysts%3A%20Oxidation%20of%20CO%20by%20Pt%20and%20Al%26%23x2013%3BPt%20Alloy%20Clusters%20Supported%20on%20Graphene%2C%20Chemistry-a%20European%20Journal%2022%20%282016%29%205181%26%23x2013%3B5188.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Fchem.201504379%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Fchem.201504379%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Reducing%20the%20Cost%20and%20Preserving%20the%20Reactivity%20in%20Noble-Metal-Based%20Catalysts%3A%20Oxidation%20of%20CO%20by%20Pt%20and%20Al%5Cu2013Pt%20Alloy%20Clusters%20Supported%20on%20Graphene%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenichi%22%2C%22lastName%22%3A%22Koizumi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Katsuyuki%22%2C%22lastName%22%3A%22Nobusada%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22The%20oxidation%20mechanisms%20of%20CO%20to%20CO2%20on%20graphene-supported%20Pt%20and%20Pt-Al%20alloy%20clusters%20are%20elucidated%20by%20reactive%20dynamical%20simulations.%20The%20general%20mechanism%20evidenced%20is%20a%20Langmuir%5Cu2013Hinshelwood%20%28LH%29%20pathway%20in%20which%20O2%20is%20adsorbed%20on%20the%20cluster%20prior%20to%20the%20CO%20oxidation.%20The%20adsorbed%20O2%20dissociates%20into%20two%20atomic%20oxygen%20atoms%20thus%20promoting%20the%20CO%20oxidation.%20Auxiliary%20simulations%20on%20alloy%20clusters%20in%20which%20other%20metals%20%28Al%2C%20Co%2C%20Cr%2C%20Cu%2C%20Fe%2C%20Ni%29%20replace%20a%20Pt%20atom%20have%20pointed%20to%20the%20aluminum%20doped%20cluster%20as%20a%20special%20case.%20In%20the%20nanoalloy%2C%20the%20reaction%20mechanism%20for%20CO%20oxidation%20is%20still%20a%20LH%20pathway%20with%20an%20activation%20barrier%20sufficiently%20low%20to%20be%20overcome%20at%20room%20temperature%2C%20thus%20preserving%20the%20catalyst%20efficiency.%20This%20provides%20a%20generalizable%20strategy%20for%20the%20design%20of%20efficient%2C%20yet%20sustainable%2C%20Pt-based%20catalysts%20at%20reduced%20cost.%22%2C%22date%22%3A%222016%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1002%5C%2Fchem.201504379%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1002%5C%2Fchem.201504379%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221521-3765%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222022-06-07T12%3A26%3A00Z%22%7D%7D%2C%7B%22key%22%3A%22TW7R8JAC%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Le%20Roux%20et%20al.%22%2C%22parsedDate%22%3A%222016%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ES.%20Le%20Roux%2C%20A.%20Bouzid%2C%20K.Y.%20Kim%2C%20S.%20Han%2C%20A.%20Zeidler%2C%20P.S.%20Salmon%2C%20C.%20Massobrio%2C%20Structure%20of%20amorphous%20GeSe9%20by%20neutron%20diffraction%20and%20first-principles%20molecular%20dynamics%3A%20Impact%20of%20trajectory%20sampling%20and%20size%20effects%2C%20Journal%20of%20Chemical%20Physics%20145%20%282016%29%20084502.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4961265%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4961265%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Structure%20of%20amorphous%20GeSe9%20by%20neutron%20diffraction%20and%20first-principles%20molecular%20dynamics%3A%20Impact%20of%20trajectory%20sampling%20and%20size%20effects%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kye%20Yeop%22%2C%22lastName%22%3A%22Kim%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Seungwu%22%2C%22lastName%22%3A%22Han%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Anita%22%2C%22lastName%22%3A%22Zeidler%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Philip%20S.%22%2C%22lastName%22%3A%22Salmon%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22The%20structure%20of%20glassy%20GeSe9%20was%20investigated%20by%20combining%20neutron%20diffraction%20with%20density-functional-theory-based%20first-principles%20molecular%20dynamics.%20In%20the%20simulations%2C%20three%20different%20models%20of%20N%20%3D%20260%20atoms%20were%20prepared%20by%20sampling%20three%20independent%20temporal%20trajectories%2C%20and%20the%20glass%20structures%20were%20found%20to%20be%20substantially%20different%20from%20those%20obtained%20for%20models%20in%20which%20smaller%20numbers%20of%20atoms%20or%20more%20rapid%20quench%20rates%20were%20employed.%20In%20particular%2C%20the%20overall%20network%20structure%20is%20based%20on%20Se-n%20chains%20that%20are%20cross-linked%20by%20Ge%28Se-4%29%281%5C%2F2%29%20tetrahedra%2C%20where%20the%20latter%20are%20predominantly%20corner%20as%20opposed%20to%20edge%20sharing.%20The%20occurrence%20of%20a%20substantial%20proportion%20of%20Ge-Se-Se%20connections%20does%20not%20support%20a%20model%20in%20which%20the%20material%20is%20phase%20separated%20into%20Se-rich%20and%20GeSe2-rich%20domains.%20The%20appearance%20of%20a%20first-sharp%20diffraction%20peak%20in%20the%20Bhatia-Thornton%20concentration-concentration%20partial%20structure%20factor%20does%2C%20however%2C%20indicate%20a%20non-uniform%20distribution%20of%20the%20Ge-centered%20structural%20motifs%20on%20an%20intermediate%20length%20scale.%20Published%20by%20AIP%20Publishing.%22%2C%22date%22%3A%222016%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1063%5C%2F1.4961265%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1063%5C%2F1.4961265%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220021-9606%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-04-30T14%3A44%3A10Z%22%7D%7D%2C%7B%22key%22%3A%22FUEWWP8D%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Ori%20et%20al.%22%2C%22parsedDate%22%3A%222016%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EG.%20Ori%2C%20C.%20Massobrio%2C%20A.%20Pradel%2C%20M.%20Ribes%2C%20B.%20Coasne%2C%20Nanoporous%20chalcogenides%20for%20adsorption%20and%20gas%20separation%2C%20Physical%20Chemistry%20Chemistry%20Physics%2018%20%282016%29%2013449%26%23x2013%3B13458.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc6cp00467a%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc6cp00467a%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Nanoporous%20chalcogenides%20for%20adsorption%20and%20gas%20separation%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Annie%22%2C%22lastName%22%3A%22Pradel%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Michel%22%2C%22lastName%22%3A%22Ribes%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Benoit%22%2C%22lastName%22%3A%22Coasne%22%7D%5D%2C%22abstractNote%22%3A%22The%20adsorption%20and%20gas%20separation%20properties%20of%20amorphous%20porous%20chalcogenides%20such%20as%20GeS2%20are%20investigated%20using%20statistical%20mechanics%20molecular%20simulation.%20Using%20a%20realistic%20molecular%20model%20of%20such%20amorphous%20adsorbents%2C%20we%20show%20that%20they%20can%20be%20used%20efficiently%20to%20separate%20different%20gases%20relevant%20to%20environmental%20and%20energy%20applications%20%28H-2%2C%20CO2%2C%20CH4%2C%20N-2%29.%20In%20addition%20to%20shedding%20light%20on%20the%20microscopic%20adsorption%20mechanisms%2C%20we%20show%20that%20coadsorption%20in%20this%20novel%20class%20of%20porous%20materials%20can%20be%20described%20using%20the%20ideal%20adsorbed%20solution%20theory%20%28IAST%29.%20Such%20a%20simple%20thermodynamic%20model%2C%20which%20allows%20avoiding%20complex%20coadsorption%20measurements%2C%20describes%20the%20adsorption%20of%20mixture%20from%20pure%20component%20adsorption%20isotherms.%20Our%20results%2C%20which%20are%20found%20to%20be%20in%20good%20agreement%20with%20available%20experimental%20data%2C%20paves%20the%20way%20for%20the%20design%20of%20gas%20separation%20membranes%20using%20the%20large%20family%20of%20porous%20chalcogenides.%22%2C%22date%22%3A%222016%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fc6cp00467a%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fc6cp00467a%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221463-9076%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222016-06-15T09%3A57%3A12Z%22%7D%7D%2C%7B%22key%22%3A%22GNHTCTN5%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22%5Cu00d6zdamar%20et%20al.%22%2C%22parsedDate%22%3A%222016%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EB.%20%26%23xD6%3Bzdamar%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20Stability%20and%20Destabilization%20Processes%20in%20the%20Formation%20of%20Ferrocene-Based%20Metal-Organic%20Molecule-Metal%20Nano-Junctions%2C%20Journal%20of%20Physical%20Chemistry%20C%20120%20%282016%29%2013825%26%23x2013%3B13830.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpcc.6b04149%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpcc.6b04149%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Stability%20and%20Destabilization%20Processes%20in%20the%20Formation%20of%20Ferrocene-Based%20Metal-Organic%20Molecule-Metal%20Nano-Junctions%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Burak%22%2C%22lastName%22%3A%22%5Cu00d6zdamar%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%222016%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Facs.jpcc.6b04149%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Facs.jpcc.6b04149%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221932-7447%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222016-08-18T14%3A06%3A36Z%22%7D%7D%2C%7B%22key%22%3A%22TTW4V92C%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Voltz%20et%20al.%22%2C%22parsedDate%22%3A%222016%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Voltz%2C%20J.%20L%26%23xE9%3Bonard%2C%20P.T.%20Touceda%2C%20J.%20Conyard%2C%20Z.%20Chaker%2C%20A.%20Dejaegere%2C%20J.%20Godet%2C%20Y.%20M%26%23xE9%3Bly%2C%20S.%20Haacke%2C%20R.H.%20Stote%2C%20Quantitative%20sampling%20of%20conformational%20heterogeneity%20of%20a%20DNA%20hairpin%20using%20molecular%20dynamics%20simulations%20and%20ultrafast%20fluorescence%20spectroscopy%2C%20Nucleic%20Acids%20Research%2044%20%282016%29%203408%26%23x2013%3B3419.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1093%5C%2Fnar%5C%2Fgkw077%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1093%5C%2Fnar%5C%2Fgkw077%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Quantitative%20sampling%20of%20conformational%20heterogeneity%20of%20a%20DNA%20hairpin%20using%20molecular%20dynamics%20simulations%20and%20ultrafast%20fluorescence%20spectroscopy%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Karine%22%2C%22lastName%22%3A%22Voltz%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22J%5Cu00e9r%5Cu00e9mie%22%2C%22lastName%22%3A%22L%5Cu00e9onard%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Patricia%20Tour%5Cu00f3n%22%2C%22lastName%22%3A%22Touceda%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jamie%22%2C%22lastName%22%3A%22Conyard%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ziyad%22%2C%22lastName%22%3A%22Chaker%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Annick%22%2C%22lastName%22%3A%22Dejaegere%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Julien%22%2C%22lastName%22%3A%22Godet%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yves%22%2C%22lastName%22%3A%22M%5Cu00e9ly%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Stefan%22%2C%22lastName%22%3A%22Haacke%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Roland%20H%22%2C%22lastName%22%3A%22Stote%22%7D%5D%2C%22abstractNote%22%3A%22Molecular%20dynamics%20%28MD%29%20simulations%20and%20time%20resolved%20fluorescence%20%28TRF%29%20spectroscopy%20were%20combined%20to%20quantitatively%20describe%20the%20conformational%20landscape%20of%20the%20DNA%20primary%20binding%20sequence%20%28PBS%29%20of%20the%20HIV-1%20genome%2C%20a%20short%20hairpin%20targeted%20by%20retroviral%20nucleocapsid%20proteins%20implicated%20in%20the%20viral%20reverse%20transcription.%20Three%202-aminopurine%20%282AP%29%20labeled%20PBS%20constructs%20were%20studied.%20For%20each%20variant%2C%20the%20complete%20distribution%20of%20fluorescence%20lifetimes%20covering%205%20orders%20of%20magnitude%20in%20timescale%20was%20measured%20and%20the%20populations%20of%20conformers%20experimentally%20observed%20to%20undergo%20static%20quenching%20were%20quantified.%20A%20binary%20quantification%20permitted%20the%20comparison%20of%20populations%20from%20experimental%20lifetime%20amplitudes%20to%20populations%20of%20aromatically%20stacked%202AP%20conformers%20obtained%20from%20simulation.%20Both%20populations%20agreed%20well%2C%20supporting%20the%20general%20assumption%20that%20quenching%20of%202AP%20fluorescence%20results%20from%20pi-stacking%20interactions%20with%20neighboring%20nucleobases%20and%20demonstrating%20the%20success%20of%20the%20proposed%20methodology%20for%20the%20combined%20analysis%20of%20TRF%20and%20MD%20data.%20Cluster%20analysis%20of%20the%20latter%20further%20identified%20predominant%20conformations%20that%20were%20consistent%20with%20the%20fluorescence%20decay%20times%20and%20amplitudes%2C%20providing%20a%20structure-based%20rationalization%20for%20the%20wide%20range%20of%20fluorescence%20lifetimes.%20Finally%2C%20the%20simulations%20provided%20evidence%20of%20local%20structural%20perturbations%20induced%20by%202AP.%20The%20approach%20presented%20is%20a%20general%20tool%20to%20investigate%20fine%20structural%20heterogeneity%20in%20nucleic%20acid%20and%20nucleoprotein%20assemblies.%22%2C%22date%22%3A%222016%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1093%5C%2Fnar%5C%2Fgkw077%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fwww.ncbi.nlm.nih.gov%5C%2Fpmc%5C%2Farticles%5C%2FPMC4838372%5C%2F%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220305-1048%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%2295EJ8IDX%22%2C%22CF4ZI7HM%22%2C%22CHW2VGSR%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222024-06-03T09%3A40%3A54Z%22%7D%7D%5D%7D
[1]
A. Bouzid, K.J. Pizzey, A. Zeidler, G. Ori, M. Boero, C. Massobrio, S. Klotz, H.E. Fischer, C.L. Bull, P.S. Salmon, Pressure-induced structural changes in the network-forming isostatic glass GeSe4: An investigation by neutron diffraction and first-principles molecular dynamics, Physical Review B 93 (2016) 014202.
https://doi.org/10.1103/PhysRevB.93.014202.
[1]
K. Koizumi, K. Nobusada, M. Boero, The absence of a gap state and enhancement of the Mars-van Krevelen reaction on oxygen defective Cu/CeO2 surfaces, Physical Chemistry Chemical Physics 18 (2016) 20708–20712.
https://doi.org/10.1039/c6cp03880h.
[1]
K. Koizumi, K. Nobusada, M. Boero, Reducing the Cost and Preserving the Reactivity in Noble-Metal-Based Catalysts: Oxidation of CO by Pt and Al–Pt Alloy Clusters Supported on Graphene, Chemistry-a European Journal 22 (2016) 5181–5188.
https://doi.org/10.1002/chem.201504379.
[1]
S. Le Roux, A. Bouzid, K.Y. Kim, S. Han, A. Zeidler, P.S. Salmon, C. Massobrio, Structure of amorphous GeSe9 by neutron diffraction and first-principles molecular dynamics: Impact of trajectory sampling and size effects, Journal of Chemical Physics 145 (2016) 084502.
https://doi.org/10.1063/1.4961265.
[1]
G. Ori, C. Massobrio, A. Pradel, M. Ribes, B. Coasne, Nanoporous chalcogenides for adsorption and gas separation, Physical Chemistry Chemistry Physics 18 (2016) 13449–13458.
https://doi.org/10.1039/c6cp00467a.
[1]
B. Özdamar, C. Massobrio, M. Boero, Stability and Destabilization Processes in the Formation of Ferrocene-Based Metal-Organic Molecule-Metal Nano-Junctions, Journal of Physical Chemistry C 120 (2016) 13825–13830.
https://doi.org/10.1021/acs.jpcc.6b04149.
[1]
K. Voltz, J. Léonard, P.T. Touceda, J. Conyard, Z. Chaker, A. Dejaegere, J. Godet, Y. Mély, S. Haacke, R.H. Stote, Quantitative sampling of conformational heterogeneity of a DNA hairpin using molecular dynamics simulations and ultrafast fluorescence spectroscopy, Nucleic Acids Research 44 (2016) 3408–3419.
https://doi.org/10.1093/nar/gkw077.
1839302
NZSFH59F
2015
1
surface-science-reports
50
creator
asc
year
7054
https://www.ipcms.unistra.fr/plugins/zotpress/
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3Afalse%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%22QAIA5QGB%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Baba%20et%20al.%22%2C%22parsedDate%22%3A%222015%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ET.%20Baba%2C%20M.%20Boero%2C%20K.%20Kamiya%2C%20H.%20Ando%2C%20S.%20Negoro%2C%20M.%20Nakano%2C%20Y.%20Shigeta%2C%20Unraveling%20the%20degradation%20of%20artificial%20amide%20bonds%20in%20nylon%20oligomer%20hydrolase%3A%20from%20induced-fit%20to%20acylation%20processes%2C%20Physical%20Chemistry%20Chemical%20Physics%2017%20%282015%29%204492%26%23x2013%3B4504.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc4cp04419c%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc4cp04419c%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Unraveling%20the%20degradation%20of%20artificial%20amide%20bonds%20in%20nylon%20oligomer%20hydrolase%3A%20from%20induced-fit%20to%20acylation%20processes%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Takeshi%22%2C%22lastName%22%3A%22Baba%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Katsumasa%22%2C%22lastName%22%3A%22Kamiya%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hiroyuki%22%2C%22lastName%22%3A%22Ando%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Seiji%22%2C%22lastName%22%3A%22Negoro%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masayoshi%22%2C%22lastName%22%3A%22Nakano%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yasuteru%22%2C%22lastName%22%3A%22Shigeta%22%7D%5D%2C%22abstractNote%22%3A%22To%20elucidate%20how%20the%20nylon%20oligomer%20hydrolase%20%28NylB%29%20acquires%20its%20peculiar%20degradation%20activity%20towards%20non-biological%20amide%20bonds%2C%20we%20inspected%20the%20underlying%20enzymatic%20processes%20going%20from%20the%20induced-fit%20upon%20substrate%20binding%20to%20acylation.%20Specifically%20we%20investigated%20the%20mutational%20effects%20of%20two%20mutants%2C%20Y170F%20and%20D181G%2C%20indicated%20in%20former%20experiments%20as%20crucial%20systems%20because%20of%20their%20specific%20amino%20acid%20residues.%20Therefore%2C%20by%20adopting%20first-principles%20molecular%20dynamics%20complemented%20with%20metadynamics%20we%20provide%20a%20detailed%20insight%20into%20the%20underlying%20acylation%20mechanism.%20Our%20results%20show%20that%20while%20in%20the%20wild%20type%20%28WT%29%20the%20Tyr170%20residue%20points%20the%20NH%20group%20towards%20the%20proton-acceptor%20site%20of%20an%20artificial%20amide%20bond%2C%20hence%20ready%20to%20react%2C%20in%20the%20Y170F%20this%20does%20not%20occur.%20The%20reason%20is%20ascribed%20to%20the%20absence%20of%20Tyr170%20in%20the%20mutant%2C%20which%20is%20replaced%20by%20phenylalanine%2C%20which%20is%20unable%20to%20form%20hydrogen%20bond%20with%20the%20amide%20bond%3B%20thus%2C%20resulting%20in%20an%20increase%20in%20the%20activation%20barrier%20of%20more%20than%2010%20kcal%20mol%28-1%29.%20Nonetheless%2C%20despite%20the%20lack%20of%20hydrogen%20bonding%20between%20the%20Y170F%20and%20the%20substrate%2C%20the%20highest%20free%20energy%20barrier%20for%20the%20induced-fit%20is%20similar%20to%20that%20of%20WT.%20This%20seems%20to%20suggest%20that%20in%20the%20induced-fit%20process%2C%20kinetics%20is%20little%20affected%20by%20the%20mutation.%20On%20the%20basis%20of%20additional%20structural%20homology%20analyses%20on%20the%20enzymes%20of%20the%20same%20family%2C%20we%20suggest%20that%20natural%20selection%20is%20responsible%20for%20the%20development%20of%20the%20peculiar%20hydrolytic%20activity%20of%20Arthrobacter%20sp.%20KI72.%22%2C%22date%22%3A%222015%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fc4cp04419c%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fc4cp04419c%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221463-9076%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-06-24T12%3A58%3A58Z%22%7D%7D%2C%7B%22key%22%3A%22EMYWW6YI%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Boero%20et%20al.%22%2C%22parsedDate%22%3A%222015%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Boero%2C%20A.%20Bouzid%2C%20S.%20Le%20Roux%2C%20B.%20Ozdamar%2C%20C.%20Massobrio%2C%20Firts-principles%20molecular%20dynamics%20methods%3A%20an%20overview%2C%20in%3A%20Molecular%20Dynamics%20Simulations%20of%20Disordered%20Materials%26%23x202F%3B%3A%20From%20Network%20Glasses%20to%20Phase-Change%20Memory%20Alloys%2C%202015%3A%20pp.%2033%26%23x2013%3B55.%20%3Ca%20class%3D%27zp-ItemURL%27%20href%3D%27https%3A%5C%2F%5C%2Flink.springer.com%5C%2Fchapter%5C%2F10.1007%5C%2F978-3-319-15675-0_2%27%3Ehttps%3A%5C%2F%5C%2Flink.springer.com%5C%2Fchapter%5C%2F10.1007%5C%2F978-3-319-15675-0_2%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22bookSection%22%2C%22title%22%3A%22Firts-principles%20molecular%20dynamics%20methods%3A%20an%20overview%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22S%5Cu00e9bastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Burak%22%2C%22lastName%22%3A%22Ozdamar%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22bookTitle%22%3A%22Molecular%20dynamics%20simulations%20of%20disordered%20materials%20%3A%20from%20network%20glasses%20to%20phase-change%20memory%20alloys%22%2C%22date%22%3A%222015%22%2C%22originalDate%22%3A%22%22%2C%22originalPublisher%22%3A%22%22%2C%22originalPlace%22%3A%22%22%2C%22format%22%3A%22%22%2C%22ISBN%22%3A%22978-3-319-15674-3%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Flink.springer.com%5C%2Fchapter%5C%2F10.1007%5C%2F978-3-319-15675-0_2%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222023-02-24T14%3A31%3A18Z%22%7D%7D%2C%7B%22key%22%3A%22RWGRBTGS%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Bouzid%20et%20al.%22%2C%22parsedDate%22%3A%222015%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EA.%20Bouzid%2C%20S.%20Gabardi%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20M.%20Bernasconi%2C%20First-principles%20study%20of%20amorphous%20Ga4Sb6Te3%20phase-change%20alloys%2C%20Physical%20Review%20B%2091%20%282015%29%20184201%20%5C%2Fp.1%26%23x2013%3B9.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.91.184201%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.91.184201%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22First-principles%20study%20of%20amorphous%20Ga4Sb6Te3%20phase-change%20alloys%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Silvia%22%2C%22lastName%22%3A%22Gabardi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Marco%22%2C%22lastName%22%3A%22Bernasconi%22%7D%5D%2C%22abstractNote%22%3A%22First-principles%20molecular%20dynamics%20simulations%20within%20the%20density%20functional%20theory%20framework%20were%20performed%20to%20generate%20amorphous%20models%20of%20the%20Ga4Sb6Te3%20phase%20change%20alloy%20by%20quenching%20from%20the%20melt.%20We%20find%20that%20Ga-Sb%20and%20Ga-Te%20are%20the%20most%20abundant%20bonds%20with%20only%20a%20minor%20amount%20of%20Sb-Te%20bonds%20participating%20to%20the%20alloy%20network.%20Ga%20and%20four-coordinated%20Sb%20atoms%20present%20a%20tetrahedral-like%20geometry%2C%20whereas%20three-coordinated%20Sb%20atoms%20are%20in%20a%20pyramidal%20configuration.%20The%20tetrahedral-like%20geometries%20are%20similar%20to%20those%20of%20the%20crystalline%20phase%20of%20the%20two%20binary%20compounds%20GaTe%20and%20GaSb.%20A%20sizable%20fraction%20of%20Sb-Sb%20bonds%20is%20also%20present%2C%20indicating%20a%20partial%20nanoscale%20segregation%20of%20Sb.%20Despite%20the%20fact%20that%20the%20composition%20Ga4Sb6Te3%20lies%20on%20the%20pseudobinary%20GaSb-Sb2Te3%20tie%20line%2C%20the%20amorphous%20network%20can%20be%20seen%20as%20a%20mixture%20of%20the%20two%20binary%20compounds%20GaTe%20and%20GaSb%20with%20intertwined%20elemental%20Sb.%22%2C%22date%22%3A%222015%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevB.91.184201%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevB.91.184201%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221098-0121%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-06-24T13%3A00%3A09Z%22%7D%7D%2C%7B%22key%22%3A%226MSTSKZG%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Bouzid%20et%20al.%22%2C%22parsedDate%22%3A%222015%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EA.%20Bouzid%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20G.%20Ori%2C%20K.%20Sykina%2C%20E.%20Furet%2C%20Role%20of%20the%20van%20der%20Waals%20interactions%20and%20impact%20of%20the%20exchange-correlation%20functional%20in%20determining%20the%20structure%20of%20glassy%20GeTe4%2C%20Physical%20Review%20B%2092%20%282015%29%20134208.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.92.134208%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.92.134208%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Role%20of%20the%20van%20der%20Waals%20interactions%20and%20impact%20of%20the%20exchange-correlation%20functional%20in%20determining%20the%20structure%20of%20glassy%20GeTe4%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kateryna%22%2C%22lastName%22%3A%22Sykina%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Eric%22%2C%22lastName%22%3A%22Furet%22%7D%5D%2C%22abstractNote%22%3A%22The%20structural%20properties%20of%20amorphous%20GeTe4%20are%20studied%20within%20the%20framework%20of%20first-principles%20molecular%20dynamics%20combined%20with%20density%20functional%20theory.%20Four%20different%20theoretical%20schemes%20are%20considered%2C%20each%20one%20intended%20to%20correspond%20to%20a%20distinct%20structural%20model.%20These%20are%20obtained%20by%20selecting%20either%20the%20PBE%20%28Perdew%2C%20Burke%2C%20and%20Ernzerhof%29%20or%20the%20BLYP%20%28Becke%2C%20Lee%2C%20Yang%2C%20and%20Parr%29%20exchange-correlation%20functionals%20and%2C%20for%20each%20one%20of%20the%20two%2C%20by%20disregarding%20or%20including%20van%20der%20Waals%20dispersion%20forces.%20Based%20on%20the%20comparison%20with%20experimental%20total%20structure%20factor%20S%28k%29%20and%20total%20pair%20correlation%20function%20g%28r%29%2C%20one%20can%20infer%20the%20quantitative%20character%20of%20such%20models%2C%20thereby%20extracting%20information%20on%20the%20underlying%20structures.%20We%20found%20that%20the%20inclusion%20of%20the%20vdW%20forces%20improves%20the%20predictive%20power%20of%20our%20approach%20for%20the%20PBE%20and%20%28to%20a%20smaller%20extent%29%20the%20BLYP%20exchange-correlation%20functionals.%20Overall%2C%20BLYP%20performs%20better%20than%20PBE%20in%20reproducing%20the%20available%20experimental%20quantities%2C%20providing%20a%20tetrahedral%20atomic-scale%20network%20profoundly%20different%20from%20the%20one%20predicted%20by%20PBE%2C%20in%20which%20tetrahedral%20and%20octahedral%20motifs%20do%20coexist.%20Our%20work%20demonstrates%20that%20a%20careful%20choice%20of%20the%20exchange-correlation%20functional%2C%20combined%20with%20the%20account%20of%20van%20der%20Waals%20forces%2C%20is%20crucial%20to%20achieve%20realistic%20structural%20predictions%20for%20glassy%20GeTe4.%22%2C%22date%22%3A%222015%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevB.92.134208%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevB.92.134208%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221098-0121%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222016-03-02T12%3A04%3A02Z%22%7D%7D%2C%7B%22key%22%3A%225ZGJ8W95%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Bouzid%20et%20al.%22%2C%22parsedDate%22%3A%222015%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EA.%20Bouzid%2C%20S.%20Le%20Roux%2C%20G.%20Ori%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20Origin%20of%20structural%20analogies%20and%20differences%20between%20the%20atomic%20structures%20of%20GeSe4%20and%20GeS4%20glasses%3A%20A%20first%20principles%20study%2C%20Journal%20of%20Chemical%20Physics%20143%20%282015%29%20034504%20%5C%2Fp.%201%26%23x2013%3B12.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4926830%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4926830%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Origin%20of%20structural%20analogies%20and%20differences%20between%20the%20atomic%20structures%20of%20GeSe4%20and%20GeS4%20glasses%3A%20A%20first%20principles%20study%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22First-principles%20molecular%20dynamics%20simulations%20based%20on%20density%20functional%20theory%20are%20employed%20for%20a%20comparative%20study%20of%20structural%20and%20bonding%20properties%20of%20two%20stoichiometrically%20identical%20chalcogenide%20glasses%2C%20GeSe4%20and%20GeS4.%20Two%20periodic%20cells%20of%20120%20and%20480%20atoms%20are%20adopted.%20Both%20glasses%20feature%20a%20coexistence%20of%20Ge-centered%20tetrahedra%20and%20Se%28S%29%20homopolar%20connections.%20Results%20obtained%20for%20N%20%3D%20480%20indicate%20substantial%20differences%20at%20the%20level%20of%20the%20Se%28S%29%20environment%2C%20since%20Ge-Se-Se%20connections%20are%20more%20frequent%20than%20the%20corresponding%20Ge-S-S%20ones.%20The%20presence%20of%20a%20more%20prominent%20first%20sharp%20diffraction%20peak%20in%20the%20total%20neutron%20structure%20factor%20of%20glassy%20GeS4%20is%20rationalized%20in%20terms%20of%20a%20higher%20number%20of%20large%20size%20rings%2C%20accounting%20for%20extended%20Ge-Se%20correlations.%20Both%20the%20electronic%20density%20of%20states%20and%20appropriate%20electronic%20localization%20tools%20provide%20evidence%20of%20a%20higher%20ionic%20character%20of%20Ge-S%20bonds%20when%20compared%20to%20Ge-Se%20bonds.%20An%20interesting%20byproduct%20of%20these%20investigations%20is%20the%20occurrence%20of%20discernible%20size%20effects%20that%20affect%20structural%20motifs%20involving%20next%20nearest%20neighbor%20distances%2C%20when%20120%20or%20480%20atoms%20are%20used.%20%28C%29%202015%20AIP%20Publishing%20LLC.%22%2C%22date%22%3A%222015%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1063%5C%2F1.4926830%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1063%5C%2F1.4926830%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220021-9606%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-04-30T14%3A44%3A25Z%22%7D%7D%2C%7B%22key%22%3A%2247VXLS6G%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Bouzid%20et%20al.%22%2C%22parsedDate%22%3A%222015%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EA.%20Bouzid%2C%20S.%20Le%20Roux%2C%20G.%20Ori%2C%20C.%20Tug%26%23xE8%3Bne%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20First-Principles%20Modeling%20of%20Binary%20Chalcogenides%3A%20Recent%20Accomplishments%20and%20New%20Achievements%2C%20in%3A%20Molecular%20Dynamics%20Simulations%20of%20Disordered%20Materials%26%23x202F%3B%3A%20From%20Network%20Glasses%20to%20Phase-Change%20Memory%20Alloys%2C%202015.%20%3Ca%20class%3D%27zp-ItemURL%27%20href%3D%27https%3A%5C%2F%5C%2Flink.springer.com%5C%2Fchapter%5C%2F10.1007%5C%2F978-3-319-15675-0_12%27%3Ehttps%3A%5C%2F%5C%2Flink.springer.com%5C%2Fchapter%5C%2F10.1007%5C%2F978-3-319-15675-0_12%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22bookSection%22%2C%22title%22%3A%22First-Principles%20Modeling%20of%20Binary%20Chalcogenides%3A%20Recent%20Accomplishments%20and%20New%20Achievements%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22S%5Cu00e9bastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Christine%22%2C%22lastName%22%3A%22Tug%5Cu00e8ne%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22bookTitle%22%3A%22Molecular%20dynamics%20simulations%20of%20disordered%20materials%20%3A%20from%20network%20glasses%20to%20phase-change%20memory%20alloys%22%2C%22date%22%3A%222015%22%2C%22originalDate%22%3A%22%22%2C%22originalPublisher%22%3A%22%22%2C%22originalPlace%22%3A%22%22%2C%22format%22%3A%22%22%2C%22ISBN%22%3A%22978-3-319-15674-3%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Flink.springer.com%5C%2Fchapter%5C%2F10.1007%5C%2F978-3-319-15675-0_12%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222023-02-24T14%3A31%3A25Z%22%7D%7D%2C%7B%22key%22%3A%228VHVAX8D%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Ikeda%20and%20Boero%22%2C%22parsedDate%22%3A%222015%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ET.%20Ikeda%2C%20M.%20Boero%2C%20Role%20of%20van%20der%20Waals%20corrections%20in%20first%20principles%20simulations%20of%20alkali%20metal%20ions%20in%20aqueous%20solutions%2C%20Journal%20of%20Chemical%20Physics%20143%20%282015%29%20194510.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4935932%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4935932%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Role%20of%20van%20der%20Waals%20corrections%20in%20first%20principles%20simulations%20of%20alkali%20metal%20ions%20in%20aqueous%20solutions%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Takashi%22%2C%22lastName%22%3A%22Ikeda%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22By%20resorting%20to%20a%20novel%20implementation%20of%20the%20first-principles-based%20van%20derWaals%20correction%20%28vdWC%29%20based%20on%20maximally%20localized%20Wannier%20functions%20%28MLWFs%29%2C%20we%20inspect%20its%20performance%20and%20assess%20its%20reliability%20for%20aqueous%20solutions%20of%20alkali%20metal%20ions.%20In%20our%20implementation%20of%20vdWCs%2C%20an%20efficient%20extrapolation%20scheme%20is%20introduced%20to%20allow%20for%20affordable%20first%20principles%20molecular%20dynamics%20avoiding%20the%20explicit%20recalculation%20of%20MLWFs%20at%20each%20step.%20We%20find%20that%20vdWCs%2C%20when%20added%20to%20the%20widely%20used%20revPBE%20gradient%20corrected%20functional%2C%20influence%20substantially%20both%20structural%20and%20dynamical%20properties%20of%20water%20molecules%2C%20with%20particular%20emphasis%20on%20the%20hydration%20shell%20of%20the%20alkali%20cations.%20These%20effects%20are%20more%20evident%20for%20strong%20structure-making%20and%20-breaking%20cationic%20species.%20Moreover%2C%20self-diffusion%20coefficients%20and%20reorientation%20correlation%20times%20of%20solvating%20water%20molecules%20change%20systematically%2C%20showing%20a%20trend%20in%20better%20agreement%20with%20experiments%20with%20respect%20to%20simulations%20neglecting%20the%20long-range%20dispersion%20contributions.%20%28C%29%202015%20AIP%20Publishing%20LLC.%22%2C%22date%22%3A%222015%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1063%5C%2F1.4935932%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1063%5C%2F1.4935932%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220021-9606%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222016-01-21T07%3A54%3A56Z%22%7D%7D%2C%7B%22key%22%3A%22QGVSW78X%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Koizumi%20et%20al.%22%2C%22parsedDate%22%3A%222015%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Koizumi%2C%20K.%20Nobusada%2C%20M.%20Boero%2C%20Reaction%20Pathway%20and%20Free%20Energy%20Landscape%20of%20Catalytic%20Oxidation%20of%20Carbon%20Monoxide%20Operated%20by%20a%20Novel%20Supported%20Gold-Copper%20Alloy%20Cluster%2C%20Journal%20of%20Physical%20Chemistry%20C%20119%20%282015%29%2015421%26%23x2013%3B15427.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpcc.5b04223%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.jpcc.5b04223%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Reaction%20Pathway%20and%20Free%20Energy%20Landscape%20of%20Catalytic%20Oxidation%20of%20Carbon%20Monoxide%20Operated%20by%20a%20Novel%20Supported%20Gold-Copper%20Alloy%20Cluster%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenichi%22%2C%22lastName%22%3A%22Koizumi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Katsuyuki%22%2C%22lastName%22%3A%22Nobusada%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22An%20insight%20into%20the%20catalytic%20activities%20of%20pure%20gold%20%28Au-8%29%20and%20gold-alloy%20%28CuAu7%29%20clusters%20supported%20on%20either%20MgO%28100%29%20or%20graphene%20has%20been%20undertaken%20in%20the%20search%20for%20an%20efficient%2C%20yet%20commercially%20appealing%20production%20of%20Au-based%20nanocatalysts.%20The%20present%20set%20of%20first-principles%20dynamical%20simulations%20shows%20that%20the%20gold%20and%20gold%20copper%20alloy%20clusters%20destabilize%20to%20various%20extents%20on%20MgO%20but%20preserve%20their%20structures%20on%20the%20graphene%20support%20at%20room%20temperature.%20Consequently%2C%20the%20Cu%20atom%20remains%20embedded%20inside%20the%20Au%20cluster%20on%20MgO%2C%20whereas%20it%20can%20be%20easily%20exposed%20on%20the%20cluster%20surface%20on%20the%20graphene%20substrate.%20This%20feature%20appears%20to%20be%20a%20general%20key%20issue%20to%20trigger%20the%20catalytic%20reaction%20and%20discloses%20new%20perspectives%20for%20a%20rational%20synthesis%20of%20supported%20Au-based%20catalysts%20relying%20on%20the%20intrinsic%20chemical%20character%20of%20Cu%20which%20possesses%20a%20stronger%20affinity%20to%20oxygen%20than%20Au.%20Indeed%2C%20the%20Cu%20atom%20acts%20as%20an%20active%20site%20for%20the%20approach%20of%20O-2%20and%20keeps%20the%20molecule%20bound%20to%20the%20cluster.%20We%20clarified%20that%20the%20catalytic%20oxidation%20of%20CO%20occurs%20on%20the%20graphene-supported%20CuAu7%20in%20a%20highly%20selective%20Langmuir-Hinshelwood%20type%20reaction%2C%20addressing%20the%20long-standing%20controversy%20about%20the%20actual%20reaction%20mechanism%20for%20this%20type%20of%20catalysis.%20Our%20findings%20contribute%20to%20the%20development%20of%20efficient%20and%20commercially%20appealing%20supported%20alloy%20clusters%20driven%20by%20a%20proper%20choice%20of%20dopants%20and%20supports%2C%20thus%20reducing%20the%20use%20of%20expensive%20gold.%22%2C%22date%22%3A%222015%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Facs.jpcc.5b04223%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Facs.jpcc.5b04223%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221932-7447%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-08-10T09%3A07%3A57Z%22%7D%7D%2C%7B%22key%22%3A%224HW2PBFC%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Koizumi%20et%20al.%22%2C%22parsedDate%22%3A%222015%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Koizumi%2C%20K.%20Nobusada%2C%20M.%20Boero%2C%20Theoretical%20Design%20of%20a%20Novel%20Copper%20Doped%20Gold%20Cluster%20supported%20on%20Graphene%20Utilizing%20ab%20initio%20Molecular%20Dynamics%20Simulations%2C%20in%3A%20Simos%2C%20TE%20and%20Kalogiratou%2C%20Z%20and%20Monovasilis%2C%20T%20%28Ed.%29%2C%20AIP%20Conference%20Proceedings%2C%20AMER%20INST%20PHYSICS%2C%202%20HUNTINGTON%20QUADRANGLE%2C%20STE%201NO1%2C%20MELVILLE%2C%20NY%2011747-4501%20USA%2C%202015%3A%20p.%20090055.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4938863%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4938863%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22conferencePaper%22%2C%22title%22%3A%22Theoretical%20Design%20of%20a%20Novel%20Copper%20Doped%20Gold%20Cluster%20supported%20on%20Graphene%20Utilizing%20ab%20initio%20Molecular%20Dynamics%20Simulations%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenichi%22%2C%22lastName%22%3A%22Koizumi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Katsuyuki%22%2C%22lastName%22%3A%22Nobusada%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22editor%22%2C%22name%22%3A%22Simos%2C%20TE%20and%20Kalogiratou%2C%20Z%20and%20Monovasilis%2C%20T%22%7D%5D%2C%22abstractNote%22%3A%22Ab%20initio%20molecular%20dynamics%20simulations%20have%20been%20used%20to%20inspect%20the%20adsorption%20of%20O-2%20to%20a%20small%20gold-copper%20alloy%20cluster%20supported%20on%20graphene.%20The%20exposed%20Cu%20atom%20in%20this%20cluster%20acts%20as%20a%20crucial%20attractive%20site%20for%20the%20approaching%20of%20O-2%20and%20consequently%20widens%20the%20reaction%20channel%20for%20the%20adsorption%20process.%20Conversely%2C%20a%20pure%20Au%20cluster%20on%20the%20same%20graphene%20support%20is%20inactive%20for%20the%20O-2%20adsorption%20because%20the%20corresponding%20reaction%20channel%20for%20the%20adsorption%20is%20very%20narrow.%20These%20results%20clearly%20indicate%20that%20doping%20a%20different%20metal%20to%20the%20Au%20cluster%20is%20a%20way%20to%20enhance%20the%20oxygen%20adsorption%20and%20to%20promote%20catalytic%20reactions.%22%2C%22proceedingsTitle%22%3A%22AIP%20Conference%20Proceedings%22%2C%22conferenceName%22%3A%22International%20Conference%20of%20Computational%20Methods%20in%20Sciences%20and%20Engineering%20%28ICCMSE%29%2C%20Ath%5Cu00e8nes%2C%20Gr%5Cu00e8ce%2C%2020-23%20mars%202015%22%2C%22date%22%3A%222015%22%2C%22eventPlace%22%3A%22%22%2C%22DOI%22%3A%2210.1063%5C%2F1.4938863%22%2C%22ISBN%22%3A%22978-0-7354-1349-8%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1063%5C%2F1.4938863%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222016-04-27T08%3A36%3A35Z%22%7D%7D%2C%7B%22key%22%3A%22UAHR9ZSF%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Massobrio%20et%20al.%22%2C%22parsedDate%22%3A%222015%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EC.%20Massobrio%2C%20J.%20Du%2C%20M.%20Bernasconi%2C%20P.S.%20Salmon%2C%20Molecular%20dynamics%20simulations%20of%20disordered%20materials%26%23x202F%3B%3A%20from%20network%20glasses%20to%20phase-change%20memory%20alloys%2C%202015.%20%3Ca%20class%3D%27zp-ItemURL%27%20href%3D%27https%3A%5C%2F%5C%2Flink.springer.com%5C%2Fbook%5C%2F10.1007%5C%2F978-3-319-15675-0%3Fpage%3D1%23toc%27%3Ehttps%3A%5C%2F%5C%2Flink.springer.com%5C%2Fbook%5C%2F10.1007%5C%2F978-3-319-15675-0%3Fpage%3D1%23toc%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22book%22%2C%22title%22%3A%22Molecular%20dynamics%20simulations%20of%20disordered%20materials%20%3A%20from%20network%20glasses%20to%20phase-change%20memory%20alloys%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jincheng%22%2C%22lastName%22%3A%22Du%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Marco%22%2C%22lastName%22%3A%22Bernasconi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Philip%20S.%22%2C%22lastName%22%3A%22Salmon%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%222015%22%2C%22originalDate%22%3A%22%22%2C%22originalPublisher%22%3A%22%22%2C%22originalPlace%22%3A%22%22%2C%22format%22%3A%22%22%2C%22ISBN%22%3A%22978-3-319-15674-3%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Flink.springer.com%5C%2Fbook%5C%2F10.1007%5C%2F978-3-319-15675-0%3Fpage%3D1%23toc%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222023-02-24T14%3A30%3A42Z%22%7D%7D%2C%7B%22key%22%3A%22AU4FJDCK%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Ozdamar%20et%20al.%22%2C%22parsedDate%22%3A%222015%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EB.%20Ozdamar%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20D.%20Felder-Flesch%2C%20S.%20Le%20Roux%2C%20Exohedral%20M-C-60%20and%20M-2-C-60%20%28M%20%3D%20Pt%2C%20Pd%29%20systems%20as%20tunable-gap%20building%20blocks%20for%20nanoarchitecture%20and%20nanocatalysis%2C%20Journal%20of%20Chemical%20Physics%20143%20%282015%29%20114308.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4930264%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4930264%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Exohedral%20M-C-60%20and%20M-2-C-60%20%28M%20%3D%20Pt%2C%20Pd%29%20systems%20as%20tunable-gap%20building%20blocks%20for%20nanoarchitecture%20and%20nanocatalysis%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Burak%22%2C%22lastName%22%3A%22Ozdamar%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Delphine%22%2C%22lastName%22%3A%22Felder-Flesch%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%5D%2C%22abstractNote%22%3A%22Transition%20metal-fullerenes%20complexes%20with%20metal%20atoms%20bound%20on%20the%20external%20surface%20of%20C-60%20are%20promising%20building%20blocks%20for%20next-generation%20fuel%20cells%20and%20catalysts.%20Yet%2C%20at%20variance%20with%20endohedral%20M%40C-60%2C%20they%20have%20received%20a%20limited%20attention.%20By%20resorting%20to%20first%20principles%20simulations%2C%20we%20elucidate%20structural%20and%20electronic%20properties%20for%20the%20Pd-C-60%2C%20Pt-C-60%2C%20PtPd-C-60%2C%20Pd-2-C-60%2C%20and%20Pt-2-C-60%20complexes.%20The%20most%20stable%20structures%20feature%20the%20metal%20atom%20located%20above%20a%20high%20electron%20density%20site%2C%20namely%2C%20the%20pi%20bond%20between%20two%20adjacent%20hexagons%20%28pi-66%20bond%29.%20When%20two%20metal%20atoms%20are%20added%2C%20the%20most%20stable%20configuration%20is%20those%20in%20which%20metal%20atoms%20still%20stand%20on%20p-66%20bonds%20but%20tends%20to%20clusterize.%20The%20electronic%20structure%2C%20rationalized%20in%20terms%20of%20localized%20Wannier%20functions%2C%20provides%20a%20clear%20picture%20of%20the%20underlying%20interactions%20responsible%20for%20the%20stability%20or%20instability%20of%20the%20complexes%2C%20showing%20a%20strict%20relationship%20between%20structure%20and%20electronic%20gap.%20%28C%29%202015%20AIP%20Publishing%20LLC.%22%2C%22date%22%3A%222015%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1063%5C%2F1.4930264%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1063%5C%2F1.4930264%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220021-9606%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-04-30T14%3A44%3A20Z%22%7D%7D%2C%7B%22key%22%3A%22EZ3H65C2%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Ori%20et%20al.%22%2C%22parsedDate%22%3A%222015%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EG.%20Ori%2C%20C.%20Massobrio%2C%20A.%20Pradel%2C%20M.%20Ribes%2C%20B.%20Coasne%2C%20Structure%20and%20Dynamics%20of%20Ionic%20Liquids%20Confined%20in%20Amorphous%20Porous%20Chalcogenides%2C%20Langmuir%2031%20%282015%29%206742%26%23x2013%3B6751.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.langmuir.5b00982%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facs.langmuir.5b00982%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Structure%20and%20Dynamics%20of%20Ionic%20Liquids%20Confined%20in%20Amorphous%20Porous%20Chalcogenides%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Annie%22%2C%22lastName%22%3A%22Pradel%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Michel%22%2C%22lastName%22%3A%22Ribes%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Benoit%22%2C%22lastName%22%3A%22Coasne%22%7D%5D%2C%22abstractNote%22%3A%22Besides%20the%20abundant%20literature%20on%20ionic%20liquids%20in%20porous%20silica%20and%20carbon%2C%20the%20confinement%20of%20such%20intriguing%20liquids%20in%20porous%20chalcogenides%20has%20received%20very%20little%20attention.%20Here%2C%20molecular%20simulation%20is%20employed%20to%20study%20the%20structural%20and%20dynamical%20properties%20of%20a%20typical%20ionic%20liquid%20confined%20in%20a%20realistic%20molecular%20model%20of%20amorphous%20chalcogenide%20with%20various%20pore%20sizes%20and%20surface%20chemistries.%20Using%20molecular%20dynamics%20in%20the%20isobaric-isothermal%20%28NPT%29%20ensemble%2C%20we%20consider%20confinement%20conditions%20relevant%20to%20real%20samples.%20Both%20the%20structure%20and%20self-dynamics%20of%20the%20confined%20phase%20are%20found%20to%20depend%20on%20the%20surface-to-volume%20ratio%20of%20the%20host%20confining%20material.%20Consequently%2C%20most%20properties%20of%20the%20confined%20ionic%20liquid%20can%20be%20written%20as%20a%20linear%20combination%20of%20surface%20and%20bulk-like%20contributions%2C%20arising%20from%20the%20ions%20in%20contact%20with%20the%20surface%20and%20the%20ions%20in%20the%20pore%20center%2C%20respectively.%20On%20the%20other%20hand%2C%20collective%20dynamical%20properties%20such%20as%20the%20ionic%20conductivity%20remain%20close%20to%20their%20bulk%20counterpart%20and%20almost%20insensitive%20to%20pore%20size%20and%20surface%20chemistry.%20These%20results%2C%20which%20are%20in%20fair%20agreement%20with%20available%20experimental%20data%2C%20provide%20a%20basis%20for%20the%20development%20of%20novel%20applications%20using%20hybrid%20organic%20inorganic%20solids%20consisting%20of%20ionic%20liquids%20confined%20in%20porous%20chalcogenides.%22%2C%22date%22%3A%222015%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Facs.langmuir.5b00982%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Facs.langmuir.5b00982%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220743-7463%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-07-31T08%3A56%3A01Z%22%7D%7D%2C%7B%22key%22%3A%22VLFLQNAJ%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Ori%20et%20al.%22%2C%22parsedDate%22%3A%222015%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EG.%20Ori%2C%20C.%20Massobrio%2C%20A.%20Bouzid%2C%20B.%20Coasne%2C%20Molecular%20Modeling%20of%20Glassy%20Surfaces%2C%20in%3A%20Molecular%20Dynamics%20Simulations%20of%20Disordered%20Materials%26%23x202F%3B%3A%20From%20Network%20Glasses%20to%20Phase-Change%20Memory%20Alloys%2C%202015%3A%20pp.%20345%26%23x2013%3B365.%20%3Ca%20class%3D%27zp-ItemURL%27%20href%3D%27https%3A%5C%2F%5C%2Flink.springer.com%5C%2Fchapter%5C%2F10.1007%5C%2F978-3-319-15675-0_13%27%3Ehttps%3A%5C%2F%5C%2Flink.springer.com%5C%2Fchapter%5C%2F10.1007%5C%2F978-3-319-15675-0_13%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22bookSection%22%2C%22title%22%3A%22Molecular%20Modeling%20of%20Glassy%20Surfaces%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22B.%22%2C%22lastName%22%3A%22Coasne%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22bookTitle%22%3A%22Molecular%20dynamics%20simulations%20of%20disordered%20materials%20%3A%20from%20network%20glasses%20to%20phase-change%20memory%20alloys%22%2C%22date%22%3A%222015%22%2C%22originalDate%22%3A%22%22%2C%22originalPublisher%22%3A%22%22%2C%22originalPlace%22%3A%22%22%2C%22format%22%3A%22%22%2C%22ISBN%22%3A%22978-3-319-15674-3%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Flink.springer.com%5C%2Fchapter%5C%2F10.1007%5C%2F978-3-319-15675-0_13%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222023-02-24T14%3A30%3A37Z%22%7D%7D%5D%7D
[1]
T. Baba, M. Boero, K. Kamiya, H. Ando, S. Negoro, M. Nakano, Y. Shigeta, Unraveling the degradation of artificial amide bonds in nylon oligomer hydrolase: from induced-fit to acylation processes, Physical Chemistry Chemical Physics 17 (2015) 4492–4504.
https://doi.org/10.1039/c4cp04419c.
[1]
M. Boero, A. Bouzid, S. Le Roux, B. Ozdamar, C. Massobrio, Firts-principles molecular dynamics methods: an overview, in: Molecular Dynamics Simulations of Disordered Materials : From Network Glasses to Phase-Change Memory Alloys, 2015: pp. 33–55.
https://link.springer.com/chapter/10.1007/978-3-319-15675-0_2.
[1]
A. Bouzid, S. Gabardi, C. Massobrio, M. Boero, M. Bernasconi, First-principles study of amorphous Ga4Sb6Te3 phase-change alloys, Physical Review B 91 (2015) 184201 /p.1–9.
https://doi.org/10.1103/PhysRevB.91.184201.
[1]
A. Bouzid, C. Massobrio, M. Boero, G. Ori, K. Sykina, E. Furet, Role of the van der Waals interactions and impact of the exchange-correlation functional in determining the structure of glassy GeTe4, Physical Review B 92 (2015) 134208.
https://doi.org/10.1103/PhysRevB.92.134208.
[1]
A. Bouzid, S. Le Roux, G. Ori, M. Boero, C. Massobrio, Origin of structural analogies and differences between the atomic structures of GeSe4 and GeS4 glasses: A first principles study, Journal of Chemical Physics 143 (2015) 034504 /p. 1–12.
https://doi.org/10.1063/1.4926830.
[1]
A. Bouzid, S. Le Roux, G. Ori, C. Tugène, M. Boero, C. Massobrio, First-Principles Modeling of Binary Chalcogenides: Recent Accomplishments and New Achievements, in: Molecular Dynamics Simulations of Disordered Materials : From Network Glasses to Phase-Change Memory Alloys, 2015.
https://link.springer.com/chapter/10.1007/978-3-319-15675-0_12.
[1]
T. Ikeda, M. Boero, Role of van der Waals corrections in first principles simulations of alkali metal ions in aqueous solutions, Journal of Chemical Physics 143 (2015) 194510.
https://doi.org/10.1063/1.4935932.
[1]
K. Koizumi, K. Nobusada, M. Boero, Reaction Pathway and Free Energy Landscape of Catalytic Oxidation of Carbon Monoxide Operated by a Novel Supported Gold-Copper Alloy Cluster, Journal of Physical Chemistry C 119 (2015) 15421–15427.
https://doi.org/10.1021/acs.jpcc.5b04223.
[1]
K. Koizumi, K. Nobusada, M. Boero, Theoretical Design of a Novel Copper Doped Gold Cluster supported on Graphene Utilizing ab initio Molecular Dynamics Simulations, in: Simos, TE and Kalogiratou, Z and Monovasilis, T (Ed.), AIP Conference Proceedings, AMER INST PHYSICS, 2 HUNTINGTON QUADRANGLE, STE 1NO1, MELVILLE, NY 11747-4501 USA, 2015: p. 090055.
https://doi.org/10.1063/1.4938863.
[1]
B. Ozdamar, M. Boero, C. Massobrio, D. Felder-Flesch, S. Le Roux, Exohedral M-C-60 and M-2-C-60 (M = Pt, Pd) systems as tunable-gap building blocks for nanoarchitecture and nanocatalysis, Journal of Chemical Physics 143 (2015) 114308.
https://doi.org/10.1063/1.4930264.
1839302
NZSFH59F
2014
1
surface-science-reports
50
creator
asc
year
7054
https://www.ipcms.unistra.fr/plugins/zotpress/
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3Afalse%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%223GJ8QRVQ%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Djimbi%20et%20al.%22%2C%22parsedDate%22%3A%222014%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ED.M.%20Djimbi%2C%20S.%20Le%20Roux%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20Metal-organic%20molecule-metal%20nano-junctions%3A%20a%20close%20contact%20between%20first-principles%20simulations%20and%20experiments%2C%20Journal%20of%20Physics-Condensed%20Matter%2026%20%282014%29%20104206.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1088%5C%2F0953-8984%5C%2F26%5C%2F10%5C%2F104206%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1088%5C%2F0953-8984%5C%2F26%5C%2F10%5C%2F104206%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Metal-organic%20molecule-metal%20nano-junctions%3A%20a%20close%20contact%20between%20first-principles%20simulations%20and%20experiments%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Duval%20Mbongo%22%2C%22lastName%22%3A%22Djimbi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22The%20realization%20of%20metal-molecule%20junctions%20for%20future%20electronic%20devices%20relies%20on%20our%20ability%20to%20assemble%20these%20heterogeneous%20objects%20at%20a%20molecular%20level%20and%20understand%20their%20structure%20and%20the%20behavior%20of%20the%20electronic%20states%20at%20the%20interface.%20Delocalized%20interface%20states%20near%20the%20metal%20Fermi%20level%20are%20a%20key%20ingredient%20for%20tailoring%20charge%20injection%2C%20and%20such%20a%20delocalization%20depends%20on%20a%20large%20number%20of%20chemical%2C%20structural%20and%20morphological%20parameters%2C%20all%20influencing%20the%20spatial%20extension%20of%20the%20electron%20wavefunctions.%20Our%20large-scale%20dynamical%20simulations%2C%20combined%20with%20experiments%2C%20show%20that%20a%20double-decker%20organometallic%20compound%20%28ferrocene%29%20can%20be%20deposited%20on%20a%20Cu%28111%29%20surface%2C%20providing%20an%20ideal%20system%20to%20investigate%20the%20adsorption%2C%20the%20interface%20states%20and%20localized%20spin%20states%20at%20a%20metal-organometallic%20interface.%20Adsorbed%20ferrocene%20is%20shown%20to%20have%20a%20peculiar%20pattern%20and%20realizes%20a%202D-like%20interface%20state%20strongly%20resembling%20Shockley%27s%20surface%20state%20of%20Cu.%20By%20a%20subsequent%20deposition%20of%20single%20metal%20atoms%20on%20the%20adsorbed%20ferrocene%2C%20we%20analyze%20the%20sensitivity%20of%20the%20interface%20state%20to%20local%20modifications%20of%20the%20interface%20potential.%20This%20provides%20an%20insight%20into%20adsorption%2C%20spin%20configuration%20and%20charge%20redistribution%20processes%2C%20showing%20how%20to%20tune%20the%20electron%20behavior%20at%20a%20metal-molecule%20interface.%22%2C%22date%22%3A%222014%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1088%5C%2F0953-8984%5C%2F26%5C%2F10%5C%2F104206%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1088%5C%2F0953-8984%5C%2F26%5C%2F10%5C%2F104206%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220953-8984%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-04-30T14%3A44%3A37Z%22%7D%7D%2C%7B%22key%22%3A%22U53GK94W%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Kamiya%20et%20al.%22%2C%22parsedDate%22%3A%222014%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Kamiya%2C%20T.%20Baba%2C%20M.%20Boero%2C%20T.%20Matsui%2C%20S.%20Negoro%2C%20Y.%20Shigeta%2C%20Nylon-Oligomer%20Hydrolase%20Promoting%20Cleavage%20Reactions%20in%20Unnatural%20Amide%20Compounds%2C%20Journal%20of%20Physical%20Chemistry%20Letters%205%20%282014%29%201210%26%23x2013%3B1216.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjz500323y%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjz500323y%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Nylon-Oligomer%20Hydrolase%20Promoting%20Cleavage%20Reactions%20in%20Unnatural%20Amide%20Compounds%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Katsumasa%22%2C%22lastName%22%3A%22Kamiya%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Takeshi%22%2C%22lastName%22%3A%22Baba%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Toru%22%2C%22lastName%22%3A%22Matsui%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Seiji%22%2C%22lastName%22%3A%22Negoro%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yasuteru%22%2C%22lastName%22%3A%22Shigeta%22%7D%5D%2C%22abstractNote%22%3A%22The%20active%20site%20of%206-aminohexanoate-dimer%20hydrolase%2C%20a%20nylon-6%20byproduct-degrading%20enzyme%20with%20a%20beta-lactamase%20fold%2C%20possesses%20a%20Ser112%5C%2FLys115%5C%2FTyr215%20catalytic%20triad%20similar%20to%20the%20one%20of%20penicillin-recognizing%20family%20of%20serine-reactive%20hydrolases%20but%20includes%20a%20unique%20Tyr170%20residue.%20By%20using%20a%20reactive%20quantum%20mechanics%5C%2Fmolecular%20mechanics%20%28QM%5C%2FMM%29%20approach%2C%20we%20work%20out%20its%20catalytic%20mechanism%20and%20related%20functional%5C%2Fstructural%20specificities.%20At%20variance%20with%20other%20peptidases%2C%20we%20show%20that%20the%20involvement%20of%20Tyr170%20in%20the%20enzyme-substrate%20interactions%20is%20responsible%20for%20a%20structural%20variation%20in%20the%20substrate-binding%20state.%20The%20acylation%20via%20a%20tetrahedral%20intermediate%20is%20the%20rate-limiting%20step%2C%20with%20a%20free-energy%20barrier%20of%20similar%20to%2021%20kcal%5C%2Fmol%2C%20driven%20by%20the%20catalytic%20triad%20Ser112%2C%20Lys115%2C%20and%20Tyr215%2C%20acting%20as%20a%20nucleophile%2C%20general%20base%2C%20and%20general%20acid%2C%20respectively.%20The%20functional%20interaction%20of%20Tyr170%20with%20this%20triad%20leads%20to%20an%20efficient%20disruption%20of%20the%20tetrahedral%20intermediate%2C%20promoting%20a%20conformational%20change%20of%20the%20substrate%20favorable%20for%20proton%20donation%20from%20the%20general%20acid.%22%2C%22date%22%3A%222014%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Fjz500323y%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Fjz500323y%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221948-7185%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-06-24T12%3A44%3A36Z%22%7D%7D%2C%7B%22key%22%3A%223ZCGNND7%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Kezilebieke%20et%20al.%22%2C%22parsedDate%22%3A%222014%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ES.%20Kezilebieke%2C%20A.%20Amokrane%2C%20M.%20Boero%2C%20S.%20Clair%2C%20M.%20Abel%2C%20J.-P.%20Bucher%2C%20Steric%20and%20electronic%20selectivity%20in%20the%20synthesis%20of%20Fe-1%2C2%2C4%2C5-tetracyanobenzene%20%28TCNB%29%20complexes%20on%20Au%28111%29%3A%20From%20topological%20confinement%20to%20bond%20formation%2C%20Nano%20Research%207%20%282014%29%20888%26%23x2013%3B897.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1007%5C%2Fs12274-014-0450-y%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1007%5C%2Fs12274-014-0450-y%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Steric%20and%20electronic%20selectivity%20in%20the%20synthesis%20of%20Fe-1%2C2%2C4%2C5-tetracyanobenzene%20%28TCNB%29%20complexes%20on%20Au%28111%29%3A%20From%20topological%20confinement%20to%20bond%20formation%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Shawulienu%22%2C%22lastName%22%3A%22Kezilebieke%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Anis%22%2C%22lastName%22%3A%22Amokrane%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sylvain%22%2C%22lastName%22%3A%22Clair%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mathieu%22%2C%22lastName%22%3A%22Abel%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jean-Pierre%22%2C%22lastName%22%3A%22Bucher%22%7D%5D%2C%22abstractNote%22%3A%22A%20study%20of%20the%20surface%20assisted%20self-assembly%20of%201%2C2%2C4%2C5-tetracyanobenzene%20%28TCNB%29%20acceptor%20molecules%20and%20Fe%20atoms%20on%20an%20Au%28111%29%20surface%20is%20presented.%20While%20conditions%20to%20get%20the%20two-dimensional%20arrays%20of%20stable%20Fe%28TCNB%29%284%29%20complexes%20are%20clearly%20identified%2C%20ultrahigh%20vacuum%20scanning%20tunneling%20microscopy%20and%20spectroscopy%20%28STM%5C%2FSTS%29%20coupled%20with%20first-principles%20calculations%20reveals%20that%20situations%20may%20occur%20where%20Fe%20and%20TCNB%20survive%20on%20the%20surface%20%28as%20Fe-4TCNB%20entities%29%20at%20a%20higher%20density%20than%20the%20original%20molecular%20monolayer%20without%20forming%20coordination%20bonds%20with%20each%20other.%20It%20is%20found%20that%20the%20square%20planar%20coordination%20of%20the%20Fe%28TCNB%29%284%29%20monomer%20complexes%20cannot%20fully%20develop%20in%20the%20presence%20of%20lateral%20strain%20due%20to%20growth-induced%20confinement.%20A%20phenomenon%20similar%20to%20steric%20hindrance%20involving%20a%20strongly%20modified%20chirality%20with%20a%20Fe-N-C%20bond%20angle%20of%20120%20degrees%20compared%20to%20the%20180%20degrees%20for%20the%20stable%20complex%20may%20then%20explain%20why%20the%20Fe%20atom%20keeps%20its%20metallic%20bond%20with%20the%20surface.%20The%20competition%20between%20steric%20and%20electronic%20effects%2C%20not%20reported%20before%2C%20may%20arise%20elsewhere%20in%20surface%20chemistry%20involved%20in%20the%20synthesis%20of%20new%20and%20potentially%20useful%20organic%20nanomaterials.%22%2C%22date%22%3A%222014%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1007%5C%2Fs12274-014-0450-y%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1007%5C%2Fs12274-014-0450-y%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221998-0124%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22RZX792Q8%22%5D%2C%22dateModified%22%3A%222016-04-13T15%3A35%3A06Z%22%7D%7D%2C%7B%22key%22%3A%22CSI736AS%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Ori%20et%20al.%22%2C%22parsedDate%22%3A%222014%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EG.%20Ori%2C%20C.%20Massobrio%2C%20A.%20Bouzid%2C%20M.%20Boero%2C%20B.%20Coasne%2C%20Surface%20of%20glassy%20GeS2%3A%20A%20model%20based%20on%20a%20first-principles%20approach%2C%20Physical%20Review%20B%2090%20%282014%29%20045423%20%5C%2Fp.1%26%23x2013%3B10.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.90.045423%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.90.045423%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Surface%20of%20glassy%20GeS2%3A%20A%20model%20based%20on%20a%20first-principles%20approach%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22G.%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22C.%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A.%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22B.%22%2C%22lastName%22%3A%22Coasne%22%7D%5D%2C%22abstractNote%22%3A%22First-principles%20calculations%20within%20the%20framework%20of%20the%20density%20functional%20theory%20are%20used%20to%20construct%20realistic%20models%20for%20the%20surface%20of%20glassy%20GeS2%20%28g-GeS2%29.%20Both%20calculations%20at%20T%20%3D%200%20K%20and%20at%20finite%20temperature%20%28T%20%3D%20300%20K%29%20are%20considered.%20This%20allows%20for%20a%20comparison%20between%20the%20structural%20and%20electronic%20properties%20of%20surface%20and%20bulk%20g-GeS2.%20Although%20the%20g-GeS2%20surface%20recovers%20the%20main%20tetrahedral%20structural%20motif%20of%20bulk%20g-GeS2%2C%20the%20number%20of%20fourfold%20coordinated%20Ge%20atoms%20and%20twofold%20coordinated%20S%20atoms%20is%20smaller%20than%20in%20the%20bulk.%20On%20the%20contrary%2C%20the%20surface%20system%20features%20a%20larger%20content%20of%20overcoordinated%20S%20atoms%20and%20threefold%20coordinated%20Ge%20atoms.%20This%20effect%20is%20more%20important%20for%20the%20g-GeS2%20surface%20relaxed%20at%200%20K.%20Maximally%20localized%20Wannier%20functions%20%28WF%29%20are%20used%20to%20inspect%20the%20nature%20of%20the%20chemical%20bonds%20of%20the%20structural%20units%20present%20at%20the%20g-GeS2%20surface.%20We%20compare%20the%20ability%20of%20several%20charge%20derivation%20methods%20to%20capture%20the%20atomic%20charge%20variations%20induced%20by%20a%20coordination%20change.%20Our%20estimate%20for%20the%20charges%20allows%20exploiting%20the%20first-principles%20results%20as%20a%20data%20base%20to%20construct%20a%20reliable%20interatomic%20force%20field.%22%2C%22date%22%3A%222014%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevB.90.045423%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevB.90.045423%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221098-0121%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-06-25T15%3A01%3A55Z%22%7D%7D%2C%7B%22key%22%3A%22FHGKVCMU%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Oshikiri%20et%20al.%22%2C%22parsedDate%22%3A%222014%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Oshikiri%2C%20J.%20Ye%2C%20M.%20Boero%2C%20The%20Role%20of%20Ni-Based%20Cocatalyst%20in%20Inhomogeneous%20RVO4%20Photocatalyst%20Systems%20%28R%20%3D%20Y%2C%20Gd%29%2C%20Journal%20of%20Physical%20Chemistry%20C%20118%20%282014%29%2012845%26%23x2013%3B12854.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjp502099v%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjp502099v%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22The%20Role%20of%20Ni-Based%20Cocatalyst%20in%20Inhomogeneous%20RVO4%20Photocatalyst%20Systems%20%28R%20%3D%20Y%2C%20Gd%29%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mitsutake%22%2C%22lastName%22%3A%22Oshikiri%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jinhua%22%2C%22lastName%22%3A%22Ye%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22We%20extend%20here%20our%20inspection%2C%20via%20first-principles%20dynamical%20simulations%20supported%20by%20experiments%2C%20to%20the%20inhomogeneous%20photocatalytic%20RVO4%20systems.%20At%20variance%20with%20our%20former%20paper%2C%20we%20limit%20the%20present%20analysis%20to%20the%20most%20promising%20photo%20catalysts%20%28R%20%3D%20Y%2C%20Gd%29%20for%20which%20the%20addition%20of%20a%20Ni-based%20cocatalyst%20provides%20the%20best%20activities.%20The%20specific%20role%20of%20Ni-based%20cocatalysts%20is%20elucidated%2C%20showing%20that%20a%20surface%20Ni%20doping%2C%20as%20opposed%20to%20a%20bulk%20one%2C%20is%20the%20most%20efficient%20way%20to%20enhance%20the%20catalytic%20activities%20of%20these%20compounds.%20This%20is%20a%20key%20step%20in%20the%20improvement%20of%20the%20photocatalysts%20and%20a%20roadmap%20to%20optimize%20the%20amount%20of%20Ni%20dopant%20needed%20for%20both%20hydrogen%20and%20oxygen%20generation.%22%2C%22date%22%3A%222014%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Fjp502099v%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Fjp502099v%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221932-7447%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-06-24T12%3A54%3A14Z%22%7D%7D%2C%7B%22key%22%3A%22HQXTM4D8%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Oshikiri%20et%20al.%22%2C%22parsedDate%22%3A%222014%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Oshikiri%2C%20J.%20Ye%2C%20M.%20Boero%2C%20Inhomogeneous%20RVO4%20Photocatalyst%20Systems%20%28R%20%3D%20V.%20Ce%2C%20Pr%2C%20Nd%2C%20Sm%2C%20Eu%2C%20Gd%2C%20Tb%2C%20Dy%2C%20Ho%2C%20Er%2C%20Tm%2C%20Yb%2C%20Lu%29%2C%20Journal%20of%20Physical%20Chemistry%20C%20118%20%282014%29%208331%26%23x2013%3B8341.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjp410565e%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjp410565e%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Inhomogeneous%20RVO4%20Photocatalyst%20Systems%20%28R%20%3D%20V.%20Ce%2C%20Pr%2C%20Nd%2C%20Sm%2C%20Eu%2C%20Gd%2C%20Tb%2C%20Dy%2C%20Ho%2C%20Er%2C%20Tm%2C%20Yb%2C%20Lu%29%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mitsutake%22%2C%22lastName%22%3A%22Oshikiri%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jinhua%22%2C%22lastName%22%3A%22Ye%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22By%20resorting%20on%20first-principles%20dynamical%20simulations%20and%20supporting%20experiments%2C%20we%20present%20a%20systematic%20and%20detailed%20inspection%20of%20a%20new%20series%20of%20inhomogeneous%20photocatalytic%20RVO4%20systems%20%28R%20%3D%20Y%2C%20Ce%2C%20Pr%2C%20Nd%2C%20Sm%2C%20Eu%2C%20Gd%2C%20Tb%2C%20Dy%2C%20Ho%2C%20Er%2C%20Tm%2C%20Yb%2C%20Lu%29%2C%20where%20YVO%2C%20is%20the%20most%20representative%20member.%20We%20evidenced%20a%20carrier%20separation%2C%20promoted%20by%20Pt%20acting%20as%20a%20cocatalyst%2C%20which%20allows%20for%20a%20clear%20understanding%20of%20the%20electronic%20properties%20of%20these%20inhomogeneous%20systems.%20These%2C%20in%20turn%2C%20are%20shown%20to%20be%20of%20crucial%20importance%20in%20enhancing%20the%20photocatalytic%20activity%20in%20the%20cases%20of%20water%20or%20methanol%20aqueous%20solutions.%20The%20presence%20of%20f%20electrons%2C%20acknowledged%20as%20essential%20for%20an%20optimal%20photocatalytic%20performance%2C%20is%20clearly%20rationalized%20and%20the%20possibility%20of%20using%20Lu%2C%20in%20view%20of%20its%20smaller%20ionic%20radius%2C%20to%20enhance%20hydrogen%20generation%20is%20disclosed.%20Experiments%20confirm%20that%20YVO4%2C%20GdVO4%2C%20and%20LuVO4%20have%20a%20potentially%20much%20higher%20efficiency%20than%20compounds%20containing%20other%20rare%20earth%20lanthanides.%20Hence%2C%20we%20provide%20a%20comprehensive%20guideline%20for%20designing%20a%20new%20generation%20of%20photocatalysts%20possessing%20unprecedented%20efficiencies.%22%2C%22date%22%3A%222014%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Fjp410565e%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Fjp410565e%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221932-7447%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-06-24T12%3A54%3A09Z%22%7D%7D%2C%7B%22key%22%3A%22ASIW4TVF%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Wezka%20et%20al.%22%2C%22parsedDate%22%3A%222014%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Wezka%2C%20A.%20Bouzid%2C%20K.J.%20Pizzey%2C%20P.S.%20Salmon%2C%20A.%20Zeidler%2C%20S.%20Klotz%2C%20H.E.%20Fischer%2C%20C.L.%20Bull%2C%20M.G.%20Tucker%2C%20M.%20Boero%2C%20S.%20Le%20Roux%2C%20C.%20Tug%26%23xE8%3Bne%2C%20C.%20Massobrio%2C%20Density-driven%20defect-mediated%20network%20collapse%20of%20GeSe2%20glass%2C%20Physical%20Review%20B%2090%20%282014%29%20054206%20%5C%2Fp.1%26%23x2013%3B9.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.90.054206%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.90.054206%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Density-driven%20defect-mediated%20network%20collapse%20of%20GeSe2%20glass%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kamil%22%2C%22lastName%22%3A%22Wezka%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Keiron%20J.%22%2C%22lastName%22%3A%22Pizzey%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Philip%20S.%22%2C%22lastName%22%3A%22Salmon%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Anita%22%2C%22lastName%22%3A%22Zeidler%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Stefan%22%2C%22lastName%22%3A%22Klotz%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Henry%20E.%22%2C%22lastName%22%3A%22Fischer%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Craig%20L.%22%2C%22lastName%22%3A%22Bull%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Matthew%20G.%22%2C%22lastName%22%3A%22Tucker%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Christine%22%2C%22lastName%22%3A%22Tug%5Cu00e8ne%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22The%20evolution%20in%20structure%20of%20the%20prototypical%20network-forming%20glass%20GeSe2%20is%20investigated%20at%20pressures%20up%20to%20similar%20to%2016%20GPa%20by%20using%20a%20combination%20of%20neutron%20diffraction%20and%20first-principles%20molecular%20dynamics.%20The%20neutron%20diffraction%20work%20at%20pressures%20%3C%3D%208.2%20GPa%20employed%20the%20method%20of%20isotope%20substitution%2C%20and%20the%20molecular%20dynamics%20simulations%20were%20performed%20with%20two%20different%20exchange-correlation%20functionals%2C%20the%20Becke-Lee-Yang-Parr%20%28BLYP%29%20and%20the%20hybrid%20Heyd-Scuseria-Ernzerhof%20HSE06.%20The%20results%20show%20density-driven%20structural%20transformations%20that%20differ%20substantially%20from%20those%20observed%20in%20common%20oxide%20glasses%20such%20as%20SiO2%20and%20GeO2.%20Edge-sharing%20tetrahedra%20persist%20as%20important%20structural%20motifs%20until%20a%20threshold%20pressure%20of%20similar%20to%208.5%20GPa%20is%20attained%2C%20whereupon%20a%20mediating%20role%20is%20found%20for%20homopolar%20bonds%20in%20the%20appearance%20of%20higher%20coordinated%20Ge-centered%20polyhedra.%20These%20mechanisms%20of%20network%20transformation%20are%20likely%20to%20be%20generic%20for%20the%20class%20of%20glass-forming%20materials%20where%20homopolar%20bonds%20and%20fragility-promoting%20edge-sharing%20motifs%20are%20prevalent%20in%20the%20ambient-pressure%20network.%22%2C%22date%22%3A%222014%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevB.90.054206%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevB.90.054206%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221098-0121%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-11-17T15%3A08%3A19Z%22%7D%7D%2C%7B%22key%22%3A%223XAACKFD%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Yang%20et%20al.%22%2C%22parsedDate%22%3A%222014%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EF.%20Yang%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20Tuning%20Magnetic%20Properties%20with%20Pressure%20in%20Hybrid%20Organic-Inorganic%20Materials%3A%20The%20Case%20of%20Copper%20Hydroxide%20Acetate%2C%20Journal%20of%20Physical%20Chemistry%20C%20118%20%282014%29%2018700%26%23x2013%3B18705.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjp503489g%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjp503489g%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Tuning%20Magnetic%20Properties%20with%20Pressure%20in%20Hybrid%20Organic-Inorganic%20Materials%3A%20The%20Case%20of%20Copper%20Hydroxide%20Acetate%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fan%22%2C%22lastName%22%3A%22Yang%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22The%20magnetic%20response%20and%20related%20spin%20topology%20of%20the%20hybrid%20organic%20inorganic%20material%20copper%20hydroxide%20acetate%20Cu-2%28OH%29%283%29%28CH3COO%29center%20dot%20H2O%20are%20studied%20as%20a%20function%20of%20an%20applied%20external%20pressure%20within%20first-principles%20approaches.%20We%20show%20that%20structural%20changes%20induced%20by%20high%20pressure%20affect%20the%20Cu-O-Cu%20angles%2C%20particularly%20when%20the%20bridging%20O%20atom%20belongs%20to%20the%20CH3COO%20chains%2C%20with%20a%20sharp%20transition%20occurring%20above%202.75%20GPa.%20These%20geometrical%20modifications%20are%20responsible%20for%20a%20transition%20from%20an%20antiferromagnetic%20%28at%20the%20ambient%20pressure%20ground%20state%29%20to%20a%20ferromagnetic%20state%20at%20high%20pressure%20%28similar%20to%207%20GPa%29.%20Our%20results%20are%20in%20agreement%20with%20the%20experimental%20outcome.%20They%20provide%20a%20guideline%20for%20applications%20of%20these%20composite%20systems%20in%20nanoelectronics%20and%20disclose%20new%20frontiers%20in%20the%20design%20of%20memory%20devices%20based%20on%20this%20family%20of%20hybrid%20lamellar%20materials.%22%2C%22date%22%3A%222014%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Fjp503489g%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Fjp503489g%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221932-7447%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-06-24T12%3A57%3A42Z%22%7D%7D%5D%7D
[1]
D.M. Djimbi, S. Le Roux, C. Massobrio, M. Boero, Metal-organic molecule-metal nano-junctions: a close contact between first-principles simulations and experiments, Journal of Physics-Condensed Matter 26 (2014) 104206.
https://doi.org/10.1088/0953-8984/26/10/104206.
[1]
K. Kamiya, T. Baba, M. Boero, T. Matsui, S. Negoro, Y. Shigeta, Nylon-Oligomer Hydrolase Promoting Cleavage Reactions in Unnatural Amide Compounds, Journal of Physical Chemistry Letters 5 (2014) 1210–1216.
https://doi.org/10.1021/jz500323y.
[1]
S. Kezilebieke, A. Amokrane, M. Boero, S. Clair, M. Abel, J.-P. Bucher, Steric and electronic selectivity in the synthesis of Fe-1,2,4,5-tetracyanobenzene (TCNB) complexes on Au(111): From topological confinement to bond formation, Nano Research 7 (2014) 888–897.
https://doi.org/10.1007/s12274-014-0450-y.
[1]
G. Ori, C. Massobrio, A. Bouzid, M. Boero, B. Coasne, Surface of glassy GeS2: A model based on a first-principles approach, Physical Review B 90 (2014) 045423 /p.1–10.
https://doi.org/10.1103/PhysRevB.90.045423.
[1]
M. Oshikiri, J. Ye, M. Boero, The Role of Ni-Based Cocatalyst in Inhomogeneous RVO4 Photocatalyst Systems (R = Y, Gd), Journal of Physical Chemistry C 118 (2014) 12845–12854.
https://doi.org/10.1021/jp502099v.
[1]
M. Oshikiri, J. Ye, M. Boero, Inhomogeneous RVO4 Photocatalyst Systems (R = V. Ce, Pr, Nd, Sm, Eu, Gd, Tb, Dy, Ho, Er, Tm, Yb, Lu), Journal of Physical Chemistry C 118 (2014) 8331–8341.
https://doi.org/10.1021/jp410565e.
[1]
K. Wezka, A. Bouzid, K.J. Pizzey, P.S. Salmon, A. Zeidler, S. Klotz, H.E. Fischer, C.L. Bull, M.G. Tucker, M. Boero, S. Le Roux, C. Tugène, C. Massobrio, Density-driven defect-mediated network collapse of GeSe2 glass, Physical Review B 90 (2014) 054206 /p.1–9.
https://doi.org/10.1103/PhysRevB.90.054206.
[1]
F. Yang, C. Massobrio, M. Boero, Tuning Magnetic Properties with Pressure in Hybrid Organic-Inorganic Materials: The Case of Copper Hydroxide Acetate, Journal of Physical Chemistry C 118 (2014) 18700–18705.
https://doi.org/10.1021/jp503489g.
1839302
NZSFH59F
2013
1
surface-science-reports
50
creator
asc
year
7054
https://www.ipcms.unistra.fr/plugins/zotpress/
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3Afalse%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%2238I9AZXX%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Bauchy%20et%20al.%22%2C%22parsedDate%22%3A%222013%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Bauchy%2C%20M.%20Micoulaut%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20Compositional%20Thresholds%20and%20Anomalies%20in%20Connection%20with%20Stiffness%20Transitions%20in%20Network%20Glasses%2C%20Physical%20Review%20Letters%20110%20%282013%29%20165501.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevLett.110.165501%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevLett.110.165501%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Compositional%20Thresholds%20and%20Anomalies%20in%20Connection%20with%20Stiffness%20Transitions%20in%20Network%20Glasses%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Bauchy%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Micoulaut%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22C.%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22The%20structural%20and%20dynamical%20properties%20of%20amorphous%20and%20liquid%20AsxSe1-x%20%280.2%20%3C%20x%20%3C%200.4%29%20are%20studied%20by%20first%20principles%20molecular%20dynamics.%20Within%20the%20above%20range%20of%20compositions%2C%20thresholds%20and%20anomalies%20are%20found%20in%20the%20behavior%20of%20reciprocal%20space%20properties%20that%20can%20be%20correlated%20to%20the%20experimental%20location%20of%20the%20so-called%20Boolchand%20intermediate%20phase%20in%20these%20glassy%20networks.%20These%20findings%20are%20associated%20with%20diffusion%20anomalies%20for%20the%20parent%20liquid%20phase%2C%20thereby%20linking%20structural%20and%20dynamical%20atomic-scale%20fingerprints%20for%20the%20onset%20of%20rigidity%20within%20the%20network%2C%20while%20also%20providing%20a%20much%20more%20complex%20picture%20than%20the%20one%20derived%20from%20mean-field%20approaches%20of%20stiffness%20transitions.%20DOI%3A%2010.1103%5C%2FPhysRevLett.110.165501%22%2C%22date%22%3A%222013%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevLett.110.165501%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevLett.110.165501%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220031-9007%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-06-24T11%3A47%3A03Z%22%7D%7D%2C%7B%22key%22%3A%22N887B7IN%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Celino%20et%20al.%22%2C%22parsedDate%22%3A%222013%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Celino%2C%20S.%20Le%20Roux%2C%20G.%20Ori%2C%20B.%20Coasne%2C%20A.%20Bouzid%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20First-principles%20molecular%20dynamics%20study%20of%20glassy%20GeS2%3A%20Atomic%20structure%20and%20bonding%20properties%2C%20Physical%20Review%20B%2088%20%282013%29%20174201.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.88.174201%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.88.174201%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22First-principles%20molecular%20dynamics%20study%20of%20glassy%20GeS2%3A%20Atomic%20structure%20and%20bonding%20properties%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Celino%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22S.%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22G.%22%2C%22lastName%22%3A%22Ori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22B.%22%2C%22lastName%22%3A%22Coasne%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A.%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22C.%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22The%20structure%20of%20glassy%20GeS2%20is%20studied%20in%20the%20framework%20of%20density%20functional%20theory%2C%20by%20using%20a%20fully%20self-consistent%20first-principles%20molecular%20dynamics%20%28FPMD%29%20scheme.%20A%20comparative%20analysis%20is%20performed%20with%20previous%20molecular%20dynamics%20data%20obtained%20within%20the%20Harris%20functional%20%28HFMD%29%20total%20energy%20approach.%20The%20calculated%20total%20neutron%20structure%20factor%20exhibits%20an%20unprecedented%20agreement%20with%20the%20experimental%20counterpart.%20In%20particular%2C%20the%20height%20of%20the%20first%20sharp%20diffraction%20peak%20%28FSDP%29%20improves%20considerably%20upon%20the%20HFMD%20results.%20Both%20the%20Ge%20and%20the%20S%20subnetworks%20are%20affected%20by%20a%20consistent%20number%20of%20miscoordinations%2C%20coexisting%20with%20the%20main%20tetrahedral%20structural%20motif.%20Glassy%20GeS2%20features%20a%20short-range%20order%20quite%20similar%20to%20the%20one%20found%20in%20glassy%20GeSe2%2C%20a%20notable%20exception%20being%20the%20larger%20number%20of%20edge-sharing%20connections.%20An%20electronic%20structure%20localization%20analysis%2C%20based%20on%20the%20Wannier%20functions%20formalism%2C%20provides%20evidence%20of%20a%20more%20enhanced%20ionic%20character%20in%20glassy%20GeS2%20when%20compared%20to%20glassy%20GeS2.%22%2C%22date%22%3A%222013%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevB.88.174201%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevB.88.174201%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221098-0121%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-04-30T14%3A44%3A56Z%22%7D%7D%2C%7B%22key%22%3A%22MZGMJF4Q%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Koizumi%20et%20al.%22%2C%22parsedDate%22%3A%222013%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Koizumi%2C%20M.%20Boero%2C%20Y.%20Shigeta%2C%20A.%20Oshiyama%2C%20Atom-Scale%20Reaction%20Pathways%20and%20Free-Energy%20Landscapes%20in%20Oxygen%20Plasma%20Etching%20of%20Graphene%2C%20Journal%20of%20Physical%20Chemistry%20Letters%204%20%282013%29%201592%26%23x2013%3B1596.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjz400666h%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjz400666h%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Atom-Scale%20Reaction%20Pathways%20and%20Free-Energy%20Landscapes%20in%20Oxygen%20Plasma%20Etching%20of%20Graphene%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22K.%22%2C%22lastName%22%3A%22Koizumi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Y.%22%2C%22lastName%22%3A%22Shigeta%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A.%22%2C%22lastName%22%3A%22Oshiyama%22%7D%5D%2C%22abstractNote%22%3A%22We%20report%20first-principles%20molecular%20dynamics%20calculations%20combined%20with%20rare%20events%20sampling%20techniques%20that%20clarify%20atom-scale%20mechanisms%20of%20oxygen%20plasma%20etching%20of%20graphene.%20The%20obtained%20reaction%20pathways%20and%20associated%20free-energy%20landscapes%20show%20that%20the%20etching%20proceeds%20near%20vacancies%20via%20a%20two-step%20mechanism%2C%20formation%20of%20precursor%20lactone%20structures%20and%20the%20subsequent%20exclusive%20CO2%20desorption.%20We%20find%20that%20atomic%20oxygen%20among%20the%20plasma%20components%20is%20most%20efficient%20for%20etching%2C%20providing%20a%20guidline%20in%20tuning%20the%20plasma%20conditions.%22%2C%22date%22%3A%222013%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Fjz400666h%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Fjz400666h%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221948-7185%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-06-24T11%3A59%3A11Z%22%7D%7D%2C%7B%22key%22%3A%22RRS2HBDA%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Le%20Roux%20et%20al.%22%2C%22parsedDate%22%3A%222013%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ES.%20Le%20Roux%2C%20A.%20Bouzid%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20The%20structure%20of%20liquid%20GeSe%20revisited%3A%20A%20first%20principles%20molecular%20dynamics%20study%2C%20Journal%20of%20Chemical%20Physics%20138%20%282013%29%20174505.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4803115%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4803115%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22The%20structure%20of%20liquid%20GeSe%20revisited%3A%20A%20first%20principles%20molecular%20dynamics%20study%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22S.%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A.%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22C.%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22Early%20first-principles%20molecular%20dynamics%20results%20on%20liquid%20GeSe%20were%20characterized%20by%20shortcomings%20in%20the%20description%20of%20Ge-Ge%20%28and%20to%20a%20lesser%20extent%20Se-Se%29%20short%20range%20correlations.%20In%20that%20case%20the%20exchange-correlation%20functional%20adopted%20was%20the%20one%20devised%20by%20Perdew%20and%20Wang%20%28PW91%29.%20In%20the%20search%20of%20improvements%20in%20the%20atomic-scale%20modelling%20of%20this%20liquid%2C%20we%20have%20produced%20new%20sets%20of%20data%20by%20employing%20two%20different%20schemes%20for%20the%20exchange-correlation%20part%20within%20the%20density%20functional%20theory%20approach.%20The%20two%20functionals%20selected%20are%20those%20proposed%20by%20Becke%2C%20Lee%2C%20Yang%2C%20and%20Parr%20%28BLYP%29%20and%20by%20Perdew%2C%20Burke%2C%20and%20Ernzerhof%20%28PBE%29.%20The%20PBE%20results%20turned%20out%20to%20be%20quite%20similar%20to%20the%20PW91%20ones.%20The%20BLYP%20results%20feature%20instead%20a%20better%20account%20of%20the%20Ge-Ge%20first%20shell%20of%20neighbors%2C%20correctly%20exhibiting%20two%20clear%20maxima%20separated%20by%20a%20deep%20minimum.%20Due%20to%20the%20increase%20in%20the%20number%20of%20the%20tetrahedral%20structural%20units%2C%20the%20atomic%20mobility%20of%20Ge%20and%20Se%20atoms%20in%20the%20network%20is%20reduced%20with%20respect%20to%20the%20PW91%20case.%20This%20brings%20the%20diffusion%20coefficients%20of%20the%20two%20species%20down%20to%20values%20close%20to%20those%20of%20liquid%20Ge2Se3%20and%20liquid%20GeSe2.%20%28C%29%202013%20AIP%20Publishing%20LLC.%22%2C%22date%22%3A%222013%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1063%5C%2F1.4803115%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1063%5C%2F1.4803115%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220021-9606%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-04-30T14%3A44%3A50Z%22%7D%7D%2C%7B%22key%22%3A%22TP658RBD%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Massobrio%20et%20al.%22%2C%22parsedDate%22%3A%222013%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EC.%20Massobrio%2C%20D.M.%20Djimbi%2C%20M.%20Matsubara%2C%20R.%20Scipioni%2C%20M.%20Boero%2C%20Stability%20of%20Ge12C48%20and%20Ge20C40%20heterofullerenes%3A%20A%20first%20principles%20molecular%20dynamics%20study%2C%20Chemical%20Physics%20Letters%20556%20%282013%29%20163%26%23x2013%3B167.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cplett.2012.11.033%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cplett.2012.11.033%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Stability%20of%20Ge12C48%20and%20Ge20C40%20heterofullerenes%3A%20A%20first%20principles%20molecular%20dynamics%20study%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22C.%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22D.%20M.%22%2C%22lastName%22%3A%22Djimbi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Matsubara%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22R.%22%2C%22lastName%22%3A%22Scipioni%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22By%20using%20first-principles%20molecular%20dynamics%2C%20we%20address%20the%20issue%20of%20structural%20stability%20for%20the%20C-60%20Ge-m%28m%29%20family%20of%20doped%20heterofullerenes%20through%20a%20set%20of%20calculations%20targeting%20C48Ge12%20and%20C40Ge20.%20Three%20kinds%20of%20theoretical%20tools%20are%20employed%3A%20%28a%29%20static%20structural%20optimization%2C%20%28b%29%20a%20bonding%20analysis%20based%20on%20localized%20orbitals%20%28Wannier%20wavefunctions%20and%20centers%29%20and%20%28c%29%20first-principles%20molecular%20dynamics%20at%20finite%20temperature.%20This%20latter%20tool%20allows%20concluding%20that%20the%20segregated%20form%20of%20C40Ge20%20is%20less%20stable%20than%20its%20Si-based%20counterpart.%20However%2C%20the%20non-segregated%20forms%20of%20C40Ge20%20and%20C40Si20%20have%20comparable%20stabilities%20at%20finite%20temperatures.%20%28C%29%202012%20Elsevier%20B.%20V.%20All%20rights%20reserved.%22%2C%22date%22%3A%222013%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.cplett.2012.11.033%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.cplett.2012.11.033%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220009-2614%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222016-04-07T07%3A57%3A31Z%22%7D%7D%2C%7B%22key%22%3A%22QXECCA2C%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Micoulaut%20et%20al.%22%2C%22parsedDate%22%3A%222013%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Micoulaut%2C%20A.%20Kachmar%2C%20M.%20Bauchy%2C%20S.%20Le%20Roux%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20Structure%2C%20topology%2C%20rings%2C%20and%20vibrational%20and%20electronic%20properties%20of%20GexSe1-x%20glasses%20across%20the%20rigidity%20transition%3A%20A%20numerical%20study%2C%20Physical%20Review%20B%2088%20%282013%29%20054203.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.88.054203%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.88.054203%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Structure%2C%20topology%2C%20rings%2C%20and%20vibrational%20and%20electronic%20properties%20of%20GexSe1-x%20glasses%20across%20the%20rigidity%20transition%3A%20A%20numerical%20study%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Micoulaut%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A.%22%2C%22lastName%22%3A%22Kachmar%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Bauchy%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22S.%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22C.%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22The%20structural%2C%20electronic%2C%20and%20vibrational%20properties%20of%20glassy%20GexSe1-x%20are%20studied%20using%20density-functional-based%20molecular%20dynamics.%20The%20focus%20is%20on%20four%20compositions%20%28x%20%3D%2010%25%2C%2020%25%2C%2025%25%2C%2033%25%29%20spanning%20the%20rigidity%20transitions%20and%20representing%20typical%20compositions%20of%20flexible%2C%20intermediate%2C%20and%20stressed%20rigid%20systems.%20We%20investigate%20structural%20properties%20including%20structure%20factors%2C%20pair%20distribution%20functions%2C%20angular%20distributions%2C%20coordination%20numbers%2C%20and%20neighbor%20distributions%20and%20compare%20our%20results%20with%20experimental%20findings%2C%20when%20available.%20Most%20noticeable%20is%20the%20excellent%20agreement%20found%20in%20the%20reproduction%20of%20the%20structure%20in%20real%20and%20reciprocal%20space%20which%20allows%20tracking%20the%20effect%20of%20Ge%20composition%20on%20the%20structure.%20Ring%20statistics%20and%20ring%20correlations%20are%20examined%20and%20followed%20across%20the%20rigidity%20transition%2C%20and%20the%20details%20of%20typical%20small%20rings%20show%20a%20much%20more%20complex%20picture%20than%20established%20previously.%20A%20comparison%20is%20made%20with%20simple%20bond%20models%20and%20their%20validity%20is%20discussed.%20Topological%20constraint%20analysis%20is%20performed%20and%20shows%20that%20the%20onset%20of%20rigidity%20changes%20substantially%20the%20angular%20motion%20inside%20the%20Ge%20tetrahedra%2C%20which%20displays%20increased%20soft%20bending%20motions%20in%20the%20stressed%20rigid%20phase.%20We%20then%20investigate%20the%20vibrational%20properties%20via%20the%20vibrational%20density%20of%20states%20and%20the%20dielectric%20function%20%28infrared%20absorption%29%2C%20and%20discuss%20them%20with%20respect%20to%20experimental%20findings.%20Finally%2C%20the%20electronic%20properties%20are%20computed%20and%20show%20an%20excellent%20agreement%20with%20respect%20to%20previous%20first-principles%20simulations%20and%20to%20experiments.%22%2C%22date%22%3A%222013%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevB.88.054203%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevB.88.054203%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221098-0121%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-04-30T14%3A44%3A44Z%22%7D%7D%2C%7B%22key%22%3A%22FGHW9U77%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Rojas-Cervellera%20et%20al.%22%2C%22parsedDate%22%3A%222013%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EV.%20Rojas-Cervellera%2C%20A.%20Ardevol%2C%20M.%20Boero%2C%20A.%20Planas%2C%20C.%20Rovira%2C%20Formation%20of%20a%20Covalent%20Glycosyl-Enzyme%20Species%20in%20a%20Retaining%20Glycosyltransferase%2C%20Chemistry-a%20European%20Journal%2019%20%282013%29%2014018%26%23x2013%3B14023.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Fchem.201302898%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Fchem.201302898%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Formation%20of%20a%20Covalent%20Glycosyl-Enzyme%20Species%20in%20a%20Retaining%20Glycosyltransferase%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22V.%22%2C%22lastName%22%3A%22Rojas-Cervellera%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A.%22%2C%22lastName%22%3A%22Ardevol%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A.%22%2C%22lastName%22%3A%22Planas%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22C.%22%2C%22lastName%22%3A%22Rovira%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%222013%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1002%5C%2Fchem.201302898%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1002%5C%2Fchem.201302898%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220947-6539%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222016-04-07T07%3A58%3A28Z%22%7D%7D%5D%7D
[1]
M. Bauchy, M. Micoulaut, M. Boero, C. Massobrio, Compositional Thresholds and Anomalies in Connection with Stiffness Transitions in Network Glasses, Physical Review Letters 110 (2013) 165501.
https://doi.org/10.1103/PhysRevLett.110.165501.
[1]
M. Celino, S. Le Roux, G. Ori, B. Coasne, A. Bouzid, M. Boero, C. Massobrio, First-principles molecular dynamics study of glassy GeS2: Atomic structure and bonding properties, Physical Review B 88 (2013) 174201.
https://doi.org/10.1103/PhysRevB.88.174201.
[1]
K. Koizumi, M. Boero, Y. Shigeta, A. Oshiyama, Atom-Scale Reaction Pathways and Free-Energy Landscapes in Oxygen Plasma Etching of Graphene, Journal of Physical Chemistry Letters 4 (2013) 1592–1596.
https://doi.org/10.1021/jz400666h.
[1]
S. Le Roux, A. Bouzid, M. Boero, C. Massobrio, The structure of liquid GeSe revisited: A first principles molecular dynamics study, Journal of Chemical Physics 138 (2013) 174505.
https://doi.org/10.1063/1.4803115.
[1]
C. Massobrio, D.M. Djimbi, M. Matsubara, R. Scipioni, M. Boero, Stability of Ge12C48 and Ge20C40 heterofullerenes: A first principles molecular dynamics study, Chemical Physics Letters 556 (2013) 163–167.
https://doi.org/10.1016/j.cplett.2012.11.033.
[1]
M. Micoulaut, A. Kachmar, M. Bauchy, S. Le Roux, C. Massobrio, M. Boero, Structure, topology, rings, and vibrational and electronic properties of GexSe1-x glasses across the rigidity transition: A numerical study, Physical Review B 88 (2013) 054203.
https://doi.org/10.1103/PhysRevB.88.054203.
[1]
V. Rojas-Cervellera, A. Ardevol, M. Boero, A. Planas, C. Rovira, Formation of a Covalent Glycosyl-Enzyme Species in a Retaining Glycosyltransferase, Chemistry-a European Journal 19 (2013) 14018–14023.
https://doi.org/10.1002/chem.201302898.
1839302
NZSFH59F
2012
1
surface-science-reports
50
creator
asc
year
7054
https://www.ipcms.unistra.fr/plugins/zotpress/
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3Afalse%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%22I6VGREWS%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Bouzid%20and%20Massobrio%22%2C%22parsedDate%22%3A%222012%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EA.%20Bouzid%2C%20C.%20Massobrio%2C%20Note%3A%20Accounting%20for%20pressure%20effects%20on%20the%20calculated%20equilibrium%20structure%20of%20glassy%20GeSe2%2C%20Journal%20of%20Chemical%20Physics%20137%20%282012%29%20046101%20%5C%2Fp.1%26%23x2013%3B2.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4739953%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4739953%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Note%3A%20Accounting%20for%20pressure%20effects%20on%20the%20calculated%20equilibrium%20structure%20of%20glassy%20GeSe2%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%222012%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1063%5C%2F1.4739953%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1063%5C%2F1.4739953%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220021-9606%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-06-22T13%3A39%3A15Z%22%7D%7D%2C%7B%22key%22%3A%22Q3PW6TR6%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Ikeda%20and%20Boero%22%2C%22parsedDate%22%3A%222012%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ET.%20Ikeda%2C%20M.%20Boero%2C%20Communication%3A%20Hydration%20structure%20and%20polarization%20of%20heavy%20alkali%20ions%3A%20A%20first%20principles%20molecular%20dynamics%20study%20of%20Rb%2B%20and%20Cs%2B%2C%20Journal%20of%20Chemical%20Physics%20137%20%282012%29%20041101%20%5C%2Fp.1%26%23x2013%3B4.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4742151%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4742151%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Communication%3A%20Hydration%20structure%20and%20polarization%20of%20heavy%20alkali%20ions%3A%20A%20first%20principles%20molecular%20dynamics%20study%20of%20Rb%2B%20and%20Cs%2B%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Takashi%22%2C%22lastName%22%3A%22Ikeda%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22Hydration%20structure%20and%20polarization%20of%20Rb%2B%20and%20Cs%2B%20in%20liquid%20water%20at%20ambient%20conditions%20were%20studied%20by%20first%20principles%20molecular%20dynamics.%20Our%20systematic%20analysis%20of%20the%20relevant%20electronic%20structures%2C%20based%20on%20maximally%20localized%20Wannier%20functions%2C%20revealed%20that%20the%20dipole%20moment%20of%20H2O%20molecules%20in%20the%20first%20solvation%20shell%20of%20the%20ions%20slightly%20increases%20with%20increasing%20the%20atomic%20number.%20We%20also%20found%20that%20the%20polarization%20of%20heavy%20alkali%20ions%2C%20particularly%20Cs%2B%2C%20tends%20to%20stabilize%20a%20peculiar%20asymmetric%20hydration%20structure%20with%20relevant%20consequences%20in%20the%20extraction%20of%20the%20harmful%20Cs-137%20resulting%20from%20nuclear%20wastes.%20%28C%29%202012%20American%20Institute%20of%20Physics.%20%5Bhttp%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1063%5C%2F1.4742151%5D%22%2C%22date%22%3A%222012%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1063%5C%2F1.4742151%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1063%5C%2F1.4742151%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220021-9606%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-06-23T09%3A44%3A13Z%22%7D%7D%2C%7B%22key%22%3A%22VERPVDIM%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Koizumi%20et%20al.%22%2C%22parsedDate%22%3A%222012%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Koizumi%2C%20M.%20Boero%2C%20Y.%20Shigeta%2C%20A.%20Oshiyama%2C%20Microscopic%20mechanisms%20of%20initial%20oxidation%20of%20Si%28100%29%3A%20Reaction%20pathways%20and%20free-energy%20barriers%2C%20Physical%20Review%20B%2085%20%282012%29%20205314%20%5C%2Fp.1%26%23x2013%3B4.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.85.205314%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.85.205314%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Microscopic%20mechanisms%20of%20initial%20oxidation%20of%20Si%28100%29%3A%20Reaction%20pathways%20and%20free-energy%20barriers%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenichi%22%2C%22lastName%22%3A%22Koizumi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yasuteru%22%2C%22lastName%22%3A%22Shigeta%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Atsushi%22%2C%22lastName%22%3A%22Oshiyama%22%7D%5D%2C%22abstractNote%22%3A%22Various%20reaction%20pathways%20and%20corresponding%20activation%20barriers%20in%20the%20initial%20oxidation%20of%20Si%28100%29%20surfaces%20are%20clarified%20by%20free-energy%20sampling%20techniques%20combined%20with%20the%20Car-Parrinello%20molecular%20dynamics.%20We%20find%20a%20crucial%20stable%20geometry%20which%20is%20ubiquitous%20during%20the%20oxidation%20and%20links%20the%20dissociation%20of%20O-2%20molecules%20and%20the%20oxidation%20of%20subsurfaces.%20The%20calculated%20free-energy%20landscape%20provides%20a%20comprehensive%20picture%20of%20the%20various%20competing%20reaction%20pathways.%22%2C%22date%22%3A%222012%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevB.85.205314%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevB.85.205314%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221098-0121%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-06-23T09%3A47%3A01Z%22%7D%7D%2C%7B%22key%22%3A%22JIJ95ZKE%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Le%20Roux%20et%20al.%22%2C%22parsedDate%22%3A%222012%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ES.%20Le%20Roux%2C%20A.%20Bouzid%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20Structural%20properties%20of%20glassy%20Ge2Se3%20from%20first-principles%20molecular%20dynamics%2C%20Physical%20Review%20B%2086%20%282012%29%20224201.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.86.224201%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.86.224201%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Structural%20properties%20of%20glassy%20Ge2Se3%20from%20first-principles%20molecular%20dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Assil%22%2C%22lastName%22%3A%22Bouzid%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22The%20structural%20properties%20of%20glassy%20Ge2Se3%20were%20studied%20in%20the%20framework%20of%20first-principles%20molecular%20dynamics%20by%20using%20the%20Becke-Lee-Yang-Parr%20scheme%20for%20the%20treatment%20of%20the%20exchange-correlation%20functional%20in%20density%20functional%20theory.%20Our%20results%20for%20the%20total%20neutron%20structure%20factor%20and%20the%20total%20pair%20distribution%20function%20are%20in%20very%20good%20agreement%20with%20the%20experimental%20results.%20When%20compared%20to%20the%20structural%20description%20obtained%20for%20liquid%20Ge2Se3%2C%20glassy%20Ge2Se3%20is%20found%20to%20be%20characterized%20by%20a%20larger%20percentage%20of%20fourfold%20coordinated%20Ge%20atoms%20and%20a%20lower%20number%20of%20miscoordinations.%20However%2C%20Ge-Ge%20homopolar%20bonds%20inevitably%20occur%20due%20to%20the%20lack%20of%20Se%20atoms%20available%2C%20at%20this%20concentration%2C%20to%20form%20GeSe4%20tetrahedra.%20Focusing%20on%20the%20family%20of%20glasses%20GexSe1-x%2C%20the%20present%20results%20allow%20a%20comparison%20to%20be%20carried%20out%20in%20reciprocal%20and%20real%20space%20among%20three%20prototypical%20glassy%20structures.%20The%20first%20was%20obtained%20at%20the%20stoichiometric%20composition%20%28glassy%20GeSe2%29%2C%20the%20second%20at%20a%20Se-rich%20composition%20%28glassy%20GeSe4%29%20and%20the%20third%20at%20a%20Ge-rich%20composition%20%28glassy%20Ge2Se3%29.%20All%20networks%20are%20consistent%20with%20the%20%5Cu201c8%20-%20N%5Cu201d%20rule%2C%20in%20particular%2C%20glassy%20GeSe4%2C%20which%20exhibits%20the%20highest%20degree%20of%20chemical%20order.%20The%20electronic%20structure%20of%20glassy%20Ge2Se3%20has%20been%20characterized%20by%20using%20the%20Wannier%20localized%20orbital%20formalism.%20The%20analysis%20of%20the%20Ge%20environment%20shows%20the%20presence%20of%20dangling%2C%20ionocovalent%20Ge-Se%2C%20and%20covalent%20bonds%2C%20the%20latter%20related%20to%20Ge-Ge%20connections.%20DOI%3A%2010.1103%5C%2FPhysRevB.86.224201%22%2C%22date%22%3A%222012%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevB.86.224201%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevB.86.224201%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221098-0121%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-04-30T14%3A45%3A17Z%22%7D%7D%2C%7B%22key%22%3A%224WGZIIUC%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Micoulaut%20et%20al.%22%2C%22parsedDate%22%3A%222012%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Micoulaut%2C%20S.%20Le%20Roux%2C%20C.%20Massobrio%2C%20Investigation%20of%20size%20effects%20on%20the%20structure%20of%20liquid%20GeSe2%20calculated%20via%20first-principles%20molecular%20dynamics%2C%20Journal%20of%20Chemical%20Physics%20136%20%282012%29%20224504%20%5C%2Fp.1%26%23x2013%3B6.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4722101%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1063%5C%2F1.4722101%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Investigation%20of%20size%20effects%20on%20the%20structure%20of%20liquid%20GeSe2%20calculated%20via%20first-principles%20molecular%20dynamics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Matthieu%22%2C%22lastName%22%3A%22Micoulaut%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22S%5Cu00e9bastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22The%20structural%20properties%20of%20liquid%20GeSe2%20have%20been%20calculated%20by%20first-principles%20molecular%20dynamics%20by%20using%20a%20periodic%20simulation%20box%20containing%20N%20%3D%20480%20atoms.%20This%20has%20allowed%20a%20comparison%20with%20previous%20results%20obtained%20on%20a%20smaller%20system%20size%20%28N%20%3D%20120%29%20%5BM.%20Micoulaut%2C%20R.%20Vuilleumier%2C%20and%20C.%20Massobrio%2C%20Phys.%20Rev.%20B%2079%2C%20214205%20%282009%29%5D.%20In%20the%20domain%20of%20first-principles%20molecular%20dynamics%2C%20we%20obtain%20an%20assessment%20of%20system%20size%20effects%20of%20unprecedented%20quality.%20Overall%2C%20no%20drastic%20differences%20are%20found%20between%20the%20two%20sets%20of%20results%2C%20confirming%20that%20N%20%3D%20120%20is%20a%20suitable%20size%20to%20achieve%20a%20realistic%20description%20of%20this%20prototypical%20disordered%20network.%20However%2C%20for%20N%20%3D%20480%2C%20short%20range%20properties%20are%20characterized%20by%20an%20increase%20of%20chemical%20order%2C%20the%20number%20of%20Ge%20tetrahedra%20coordinated%20to%20four%20Se%20atoms%20being%20larger.%20At%20the%20intermediate%20range%20order%20level%2C%20size%20effect%20mostly%20modify%20the%20low%20wavevector%20region%20%28k%20similar%20to%201%20angstrom%28-1%29%29%20in%20the%20concentration-concentration%20partial%20structure%20factor.%20%28C%29%202012%20American%20Institute%20of%20Physics.%20%5Bhttp%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1063%5C%2F1.4722101%5D%22%2C%22date%22%3A%222012%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1063%5C%2F1.4722101%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1063%5C%2F1.4722101%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220021-9606%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-04-30T14%3A45%3A10Z%22%7D%7D%2C%7B%22key%22%3A%225F9KJMET%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Sykina%20et%20al.%22%2C%22parsedDate%22%3A%222012%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Sykina%2C%20E.%20Furet%2C%20B.%20Bureau%2C%20S.%20Le%20Roux%2C%20C.%20Massobrio%2C%20Network%20connectivity%20and%20extended%20Se%20chains%20in%20the%20atomic%20structure%20of%20glassy%20GeSe4%2C%20Chemical%20Physics%20Letters%20547%20%282012%29%2030%26%23x2013%3B34.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cplett.2012.07.077%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cplett.2012.07.077%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Network%20connectivity%20and%20extended%20Se%20chains%20in%20the%20atomic%20structure%20of%20glassy%20GeSe4%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kateryna%22%2C%22lastName%22%3A%22Sykina%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Eric%22%2C%22lastName%22%3A%22Furet%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Bruno%22%2C%22lastName%22%3A%22Bureau%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22In%20order%20to%20contribute%20to%20the%20ongoing%20debate%20on%20the%20atomic%20structure%20of%20Se-rich%20GexSe1-x%20glasses%2C%20we%20have%20studied%20the%20structure%20of%20glassy%20GeSe4%20by%20first-principles%20molecular%20dynamics.%20The%20Perdew-Burke-Ernzerhof%20generalized%20gradient%20approximation%20has%20been%20employed.%20The%20nearest-neighbor%20Se%20environment%20is%20found%20to%20be%20compatible%20with%20the%20coexistence%20of%20a%20network%20of%20GeSe4%20tetrahedra%20and%20Se-n%20chains%2C%20thereby%20confirming%20previous%20Perdew-Wang%20results.%20When%20comparing%20PW%20and%20PBE%20results%2C%20changes%20are%20found%20in%20the%20Ge-Ge%20coordination%20environment%20and%20in%20the%20number%20of%20GeSe4%20tetrahedra.%20In%20addition%2C%20very%20extended%20Se-n%20chains%20%28n%20%3E%203%2C%20up%20to%20n%20%3D%2012%29%20are%20present%20%28PBE%20case%29.%20%28C%29%202012%20Elsevier%20B.V.%20All%20rights%20reserved.%22%2C%22date%22%3A%222012%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.cplett.2012.07.077%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.cplett.2012.07.077%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220009-2614%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-04-30T14%3A45%3A03Z%22%7D%7D%2C%7B%22key%22%3A%22ME46U7QS%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Yang%20et%20al.%22%2C%22parsedDate%22%3A%222012%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EF.%20Yang%2C%20M.%20Boero%2C%20P.%20Rabu%2C%20C.%20Massobrio%2C%20First%20principles%20investigation%20of%20the%20atomic%20structure%20and%20magnetic%20properties%20of%20copper%20hydroxide%20acetate%2C%20Comptes%20Rendus%20Chimie%2015%20%282012%29%20202%26%23x2013%3B208.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.crci.2011.09.007%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.crci.2011.09.007%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22First%20principles%20investigation%20of%20the%20atomic%20structure%20and%20magnetic%20properties%20of%20copper%20hydroxide%20acetate%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fan%22%2C%22lastName%22%3A%22Yang%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Pierre%22%2C%22lastName%22%3A%22Rabu%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22Within%20the%20density%20functional%20theory%20%28DFT%29%20framework%2C%20we%20study%20the%20structural%20properties%20and%20the%20spin%20density%20distribution%20of%20copper%20hydroxide%20acetate%20Cu-2%28OH%29%283%29%28CH3COO%29center%20dot%20H2O%2C%20a%20fundamental%20compound%20in%20the%20field%20of%20hybrid%20organic-inorganic%20materials.%20In%20particular%2C%20after%20a%20careful%20comparison%20of%20the%20computed%20structure%20with%20the%20experimentally%20obtained%20X-ray%20pattern%2C%20we%20provide%20an%20insight%20into%20the%20effect%20of%20two%20exchange-correlation%20functionals%20%28BLYP%20and%20HCTH%5C%2F120%29%20on%20the%20description%20of%20the%20magnetic%20properties%20and%20on%20the%20related%20changes%20in%20the%20atomic%20structure.%20Since%20these%20compounds%20are%20known%20to%20possess%20a%20piezomagnetic%20character%2C%20for%20the%20total%20spin%20value%20S%20%3D%200%20and%20S%20%3D%204%2C%20we%20inspect%20at%20different%20pressures%2C%20and%20for%20each%20one%20of%20the%20two%20exchange-correlation%20functionals%2C%20the%20changes%20in%20the%20geometry%20and%20in%20the%20spin%20density%20distribution.%20We%20notice%20that%2C%20at%20total%20spin%20density%20S%20%3D%200%2C%20and%20for%20both%20the%20external%20pressures%20of%203%20and%205%20GPa%2C%20most%20of%20the%20local%20spin%20densities%20are%20due%20to%2042%20orbitals%20with%20about%2025%25%20contribution%20from%20d%28z%29%282%29%20orbitals.%20Such%20a%20feature%20is%20insensitive%20to%20the%20exchange-correlation%20functional%20adopted.%20Overall%2C%20our%20results%20provide%20an%20important%20first%20step%20towards%20the%20comprehension%20of%20the%20effects%20of%20an%20external%20stress%20on%20the%20magnetic%20properties%20of%20this%20class%20of%20hybrid%20materials.%20%28C%29%202011%20Academie%20des%20sciences.%20Published%20by%20Elsevier%20Masson%20SAS.%20All%20rights%20reserved.%22%2C%22date%22%3A%222012%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.crci.2011.09.007%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.crci.2011.09.007%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221631-0748%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22M244N6AF%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-06-24T10%3A46%3A37Z%22%7D%7D%5D%7D
[1]
A. Bouzid, C. Massobrio, Note: Accounting for pressure effects on the calculated equilibrium structure of glassy GeSe2, Journal of Chemical Physics 137 (2012) 046101 /p.1–2.
https://doi.org/10.1063/1.4739953.
[1]
T. Ikeda, M. Boero, Communication: Hydration structure and polarization of heavy alkali ions: A first principles molecular dynamics study of Rb+ and Cs+, Journal of Chemical Physics 137 (2012) 041101 /p.1–4.
https://doi.org/10.1063/1.4742151.
[1]
K. Koizumi, M. Boero, Y. Shigeta, A. Oshiyama, Microscopic mechanisms of initial oxidation of Si(100): Reaction pathways and free-energy barriers, Physical Review B 85 (2012) 205314 /p.1–4.
https://doi.org/10.1103/PhysRevB.85.205314.
[1]
M. Micoulaut, S. Le Roux, C. Massobrio, Investigation of size effects on the structure of liquid GeSe2 calculated via first-principles molecular dynamics, Journal of Chemical Physics 136 (2012) 224504 /p.1–6.
https://doi.org/10.1063/1.4722101.
[1]
K. Sykina, E. Furet, B. Bureau, S. Le Roux, C. Massobrio, Network connectivity and extended Se chains in the atomic structure of glassy GeSe4, Chemical Physics Letters 547 (2012) 30–34.
https://doi.org/10.1016/j.cplett.2012.07.077.
[1]
F. Yang, M. Boero, P. Rabu, C. Massobrio, First principles investigation of the atomic structure and magnetic properties of copper hydroxide acetate, Comptes Rendus Chimie 15 (2012) 202–208.
https://doi.org/10.1016/j.crci.2011.09.007.
1839302
NZSFH59F
2011
1
surface-science-reports
50
creator
asc
year
7054
https://www.ipcms.unistra.fr/plugins/zotpress/
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3Afalse%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%22BG98WNG3%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Bauchy%20et%20al.%22%2C%22parsedDate%22%3A%222011%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Bauchy%2C%20M.%20Micoulaut%2C%20M.%20Celino%2C%20S.%20Le%20Roux%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20Angular%20rigidity%20in%20tetrahedral%20network%20glasses%20with%20changing%20composition%2C%20Physical%20Review%20B%2084%20%282011%29%20054201%20%5C%2Fp.%201%26%23x2013%3B9.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.84.054201%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.84.054201%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Angular%20rigidity%20in%20tetrahedral%20network%20glasses%20with%20changing%20composition%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Bauchy%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Micoulaut%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Celino%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22S%5Cu00e9bastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22A%20set%20of%20oxide%20and%20chalcogenide%20tetrahedral%20glasses%20is%20investigated%20using%20molecular%20dynamics%20simulations.%20We%20show%20that%20the%20changes%20in%20the%20Ge%20composition%20affect%20mostly%20bending%20around%20germanium%20in%20binary%20Ge-Se%20systems%2C%20leaving%20Se-centered%20bending%20almost%20unchanged.%20In%20contrast%2C%20the%20corresponding%20Se%20twisting%20%28quantified%20by%20the%20dihedral%20angle%29%20depends%20on%20the%20Ge%20composition%20and%20is%20reduced%20when%20the%20system%20becomes%20rigid.%20It%20is%20also%20shown%20that%20angles%20involving%20the%20fourth%20neighbor%20around%20Ge%20is%20found%20to%20change%20when%20the%20system%20enters%20the%20stressed%20rigid%20phase.%20The%20same%20analysis%20reveals%20that%20unlike%20stoichiometric%20selenides%20such%20as%20GeSe2%20and%20SiSe2%2C%20germania%20and%20silica%20display%20large%20standard%20deviations%20in%20the%20bond%20angle%20distributions.%20Within%20bond-bending%20constraints%20theory%2C%20this%20pattern%20can%20be%20interpreted%20as%20a%20manifestation%20of%20broken%20%28i.e.%2C%20ineffective%29%20oxygen%20bond-bending%20constraints%2C%20whereas%20the%20silicon%20and%20germanium%20bending%20in%20oxides%20is%20found%20to%20be%20similar%20to%20the%20one%20found%20in%20flexible%20and%20intermediate%20Ge-Se%20systems.%20Our%20results%20establish%20the%20atomic-scale%20foundations%20of%20the%20phenomenological%20rigidity%20theory%2C%20thereby%20profoundly%20extending%20its%20significance%20and%20impact%20on%20the%20structural%20description%20of%20network%20glasses.%22%2C%22date%22%3A%222011%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevB.84.054201%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevB.84.054201%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221098-0121%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-04-30T14%3A45%3A44Z%22%7D%7D%2C%7B%22key%22%3A%22R546XZJ3%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Bhosle%20et%20al.%22%2C%22parsedDate%22%3A%222011%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ES.%20Bhosle%2C%20K.%20Gunasekera%2C%20P.%20Chen%2C%20P.%20Boolchand%2C%20M.%20Micoulaut%2C%20C.%20Massobrio%2C%20Meeting%20experimental%20challenges%20to%20physics%20of%20network%20glasses%3A%20Assessing%20the%20role%20of%20sample%20homogeneity%2C%20Solid%20State%20Communications%20151%20%282011%29%201851%26%23x2013%3B1855.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.ssc.2011.10.016%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.ssc.2011.10.016%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Meeting%20experimental%20challenges%20to%20physics%20of%20network%20glasses%3A%20Assessing%20the%20role%20of%20sample%20homogeneity%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22S.%22%2C%22lastName%22%3A%22Bhosle%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22K.%22%2C%22lastName%22%3A%22Gunasekera%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ping%22%2C%22lastName%22%3A%22Chen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22P.%22%2C%22lastName%22%3A%22Boolchand%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%22%2C%22lastName%22%3A%22Micoulaut%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22We%20introduce%20a%20Raman%20profiling%20method%20to%20track%20homogenization%20of%20Ge%28x%29Se%28100-x%29%20melts%20in%20real%20time%2C%20and%20show%20that%202%20g%20melts%20reacted%20at%20950%20degrees%20C%20in%20high%20vacuum%20homogenize%20in%20168%20h%20on%20a%20scale%20of%2010%20mu%20m.%20Homogenization%20of%20melts%20is%20precursive%20to%20self-organization%20of%20glasses.%20In%20the%20present%20glasses%2C%20compositional%20variation%20of%20Raman%20active%20corner-sharing%20mode%20frequency%20of%20GeSe%284%29%20units%2C%20molar%20volumes%2C%20and%20the%20enthalpy%20of%20relaxation%20at%20T%28g%29%2C%20reveal%20the%20rigidity%20%28x%28c%29%281%29%20%3D%2019.5%283%29%25%29%20and%20the%20stress%20%28x%28c%29%282%29%20%3D%2026.0%283%29%25%29%20transitions%20to%20be%20rather%20sharp%20%28Delta%20chi%20%3C%200.6%25%29.%20These%20abrupt%20elastic%20phase%20transitions%20are%20in%20harmony%20with%20rigidity%20theory%20and%20have%20a%20direct%20bearing%20on%20the%20physics%20of%20glasses.%20%28C%29%202011%20Elsevier%20Ltd.%20All%20rights%20reserved.%22%2C%22date%22%3A%222011%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.ssc.2011.10.016%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.ssc.2011.10.016%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220038-1098%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222016-04-08T07%3A06%3A13Z%22%7D%7D%2C%7B%22key%22%3A%22R66UT4NM%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Boero%22%2C%22parsedDate%22%3A%222011%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Boero%2C%20LeuRS%20Synthetase%3A%20A%20First-Principles%20Investigation%20of%20the%20Water-Mediated%20Editing%20Reaction%2C%20Journal%20of%20Physical%20Chemistry%20B%20115%20%282011%29%2012276%26%23x2013%3B12286.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjp2070024%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjp2070024%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22LeuRS%20Synthetase%3A%20A%20First-Principles%20Investigation%20of%20the%20Water-Mediated%20Editing%20Reaction%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22Starting%20from%20the%20crystallographic%20structure%20of%20the%20Therm%20us%20thermophilus%20leucyl-tRNA%20%28LeuRS%29%20synthethase%20system%20and%20recent%20theoretical%20findings%2C%20our%20combined%20hybrid%20QM%5C%2FMM%20and%20free%20energy%20metadynamics%20sampling%20approach%20shows%20that%20in%20the%20editing%20domain%20the%20enzymatic%20activity%20is%20initiated%20by%20the%20dissociation%20of%20a%20specific%20water%20molecule%20in%20proximity%20of%20the%20chemical%20bonds%20%28-C%2A-O%2A-%29%20constituting%20the%20nucleotide%20binding%20of%20the%20tRNA%20to%20the%20leucyl%20protein.%20A%20crucial%20promoter%20of%20the%20reaction%20is%20the%203%27-OH%20group%20of%20the%20cognate%20tRNA%20bound%20to%20the%20editing%20site%20of%20leucyl%2C%20which%20forms%20a%20stable%20hydrogen%20bond%20with%20this%20peculiar%20catalytic%20water%20molecule.%20We%20could%20identify%20two%20possible%20reaction%20mechanisms%20for%20the%20initial%20stage%20of%20the%20editing%20reaction%3A%20In%20one%20case%20the%203%27-OH%20group%20of%20the%20cognate%20tRNA%20acts%20as%20a%20Lewis%20acid%2C%20and%20one%20of%20the%20protons%20of%20the%20catalytic%20water%20molecule%20becomes%20a%20temporarily%20shared%20proton%20between%203%27-OH%20and%20this%20specific%20H%282%29O%2C%20thus%20helping%20its%20dissociation.%20The%20dissociation%20products%20OH%28-%29%20and%20H%28%2B%29%20attack%20the%20C%2A%20and%20O%2A%20atoms%2C%20respectively%2C%20thus%20promoting%20the%20C-O%20bond%20cleavage.%20In%20the%20second%20case%2C%20the%203%27-OH%20group%20of%20the%20cognate%20tRNA%20drives%2C%20via%20its%20hydrogen%20bond%2C%20the%20catalytic%20water%20molecule%20toward%20the%20-C%2A-O%2A-%20bond%2C%20and%20the%20unoccupied%20LUMO%20state%2C%20located%20on%20top%20of%20this%20bond%2C%20becomes%20the%20electron%20acceptor%20for%20the%20dissociating%20water%20molecule.%20This%20promotes%20the%20reaction%20toward%20the%20same%20final%20product%20found%20in%20the%20former%20pathway%2C%20but%20without%20involving%20temporary%20proton%20transfers%20to%203%27-OH.%22%2C%22date%22%3A%222011%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Fjp2070024%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Fjp2070024%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221520-6106%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-11-30T09%3A43%3A11Z%22%7D%7D%2C%7B%22key%22%3A%22NUHHKHAR%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Giacomazzi%20et%20al.%22%2C%22parsedDate%22%3A%222011%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EL.%20Giacomazzi%2C%20C.%20Massobrio%2C%20A.%20Pasquarello%2C%20Vibrational%20properties%20of%20vitreous%20GeSe2%20with%20the%20Becke-Lee-Yang-Parr%20density%20functional%2C%20Journal%20of%20Physics-Condensed%20Matter%2023%20%282011%29%20295401%20%5C%2Fp.%201%26%23x2013%3B6.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1088%5C%2F0953-8984%5C%2F23%5C%2F29%5C%2F295401%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1088%5C%2F0953-8984%5C%2F23%5C%2F29%5C%2F295401%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Vibrational%20properties%20of%20vitreous%20GeSe2%20with%20the%20Becke-Lee-Yang-Parr%20density%20functional%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Luigi%22%2C%22lastName%22%3A%22Giacomazzi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Alfredo%22%2C%22lastName%22%3A%22Pasquarello%22%7D%5D%2C%22abstractNote%22%3A%22The%20vibrational%20properties%20of%20vitreous%20GeSe2%20are%20studied%20within%20the%20Becke-Lee-Yang-Parr%20%28BLYP%29%20generalized%20gradient%20approximation%20to%20density%20functional%20theory.%20For%20this%20purpose%2C%20we%20consider%20two%20models.%20The%20first%20one%20is%20derived%20from%20a%20chemically%20ordered%20network%2C%20previously%20generated%20through%20classical%20molecular%20dynamics.%20The%20second%20one%20is%20generated%20through%20first-principles%20molecular%20dynamics%20simulations%20based%20on%20the%20BLYP%20functional%20and%20shows%20a%20larger%20variety%20of%20structural%20motifs.%20We%20describe%20the%20structural%20and%20electronic%20properties%20of%20our%20models%2C%20including%20bond%20lengths%2C%20first-neighbor%20coordinations%2C%20and%20the%20electronic%20density%20of%20states.%20The%20vibrational%20properties%20are%20addressed%20through%20the%20vibrational%20density%20of%20states%2C%20the%20infrared%20spectrum%2C%20and%20the%20Raman%20spectrum.%20For%20the%20chemically%20ordered%20model%2C%20the%20vibrational%20properties%20obtained%20with%20the%20BLYP%20functional%20are%20of%20comparable%20quality%20with%20respect%20to%20previous%20results%20obtained%20with%20the%20Perdew-Wang%20%28PW%29%20functional.%20However%2C%20a%20global%20consideration%20of%20our%20results%20indicates%20that%20the%20BLYP%20functional%20yields%20an%20overall%20improved%20description%20for%20the%20vibrational%20properties%20of%20vitreous%20GeSe2.%20The%20BLYP%20dielectric%20constants%20are%20closer%20to%20experimental%20values.%20Furthermore%2C%20we%20found%20that%20the%20Raman%20spectrum%20of%20the%20model%20generated%20through%20first-principles%20molecular%20dynamics%20within%20the%20BLYP%20scheme%20is%20largely%20improved%20with%20respect%20to%20an%20analogous%20model%20generated%20within%20the%20PW%20scheme.%22%2C%22date%22%3A%222011%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1088%5C%2F0953-8984%5C%2F23%5C%2F29%5C%2F295401%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1088%5C%2F0953-8984%5C%2F23%5C%2F29%5C%2F295401%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220953-8984%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-11-30T09%3A30%3A45Z%22%7D%7D%2C%7B%22key%22%3A%22VQ3H8SF8%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Heinrich%20et%20al.%22%2C%22parsedDate%22%3A%222011%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EB.W.%20Heinrich%2C%20L.%20Limot%2C%20M.V.%20Rastei%2C%20C.%20Iacovita%2C%20J.-P.%20Bucher%2C%20D.M.%20Djimbi%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20Dispersion%20and%20Localization%20of%20Electronic%20States%20at%20a%20Ferrocene%5C%2FCu%28111%29%20Interface%2C%20Physical%20Review%20Letters%20107%20%282011%29%20216801%20%5C%2Fp.%201%26%23x2013%3B5.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevLett.107.216801%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevLett.107.216801%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Dispersion%20and%20Localization%20of%20Electronic%20States%20at%20a%20Ferrocene%5C%2FCu%28111%29%20Interface%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Beno%5Cu00eet%20W.%22%2C%22lastName%22%3A%22Heinrich%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Laurent%22%2C%22lastName%22%3A%22Limot%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mircea%20V.%22%2C%22lastName%22%3A%22Rastei%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22C.%22%2C%22lastName%22%3A%22Iacovita%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jean-Pierre%22%2C%22lastName%22%3A%22Bucher%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Duval%20Mbongo%22%2C%22lastName%22%3A%22Djimbi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22Low-temperature%20scanning%20tunneling%20microscopy%20and%20spectroscopy%20combined%20with%20first-principles%20simulations%20reveal%20a%20nondissociative%20physisorption%20of%20ferrocene%20molecules%20on%20a%20Cu%28111%29%20surface%2C%20giving%20rise%20to%20ordered%20molecular%20layers.%20At%20the%20interface%2C%20a%202D-like%20electronic%20band%20is%20found%2C%20which%20shows%20an%20identical%20dispersion%20as%20the%20Cu%28111%29%20Shockley%20surface-state%20band.%20Subsequent%20deposition%20of%20Cu%20atoms%20forms%20charged%20organometallic%20compounds%20that%20localize%20interface-state%20electrons.%22%2C%22date%22%3A%222011%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevLett.107.216801%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevLett.107.216801%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220031-9007%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%229USMFXMV%22%2C%22CF4ZI7HM%22%2C%22DEB5KWFS%22%2C%22IEGKATUQ%22%2C%22NZSFH59F%22%2C%22RZX792Q8%22%2C%22VRM2E3H6%22%5D%2C%22dateModified%22%3A%222021-04-30T14%3A29%3A28Z%22%7D%7D%2C%7B%22key%22%3A%22X7EIVJX6%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Hou%20et%20al.%22%2C%22parsedDate%22%3A%222011%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EZ.%20Hou%2C%20X.%20Wang%2C%20T.%20Ikeda%2C%20S.-F.%20Huang%2C%20K.%20Terakura%2C%20M.%20Boero%2C%20M.%20Oshima%2C%20M.%20Kakimoto%2C%20S.%20Miyata%2C%20Effect%20of%20Hydrogen%20Termination%20on%20Carbon%20K-Edge%20X-ray%20Absorption%20Spectra%20of%20Nanographene%2C%20Journal%20of%20Physical%20Chemistry%20C%20115%20%282011%29%205392%26%23x2013%3B5403.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjp110879d%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjp110879d%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Effect%20of%20Hydrogen%20Termination%20on%20Carbon%20K-Edge%20X-ray%20Absorption%20Spectra%20of%20Nanographene%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Zhufeng%22%2C%22lastName%22%3A%22Hou%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Xianlong%22%2C%22lastName%22%3A%22Wang%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Takashi%22%2C%22lastName%22%3A%22Ikeda%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Shen-Feng%22%2C%22lastName%22%3A%22Huang%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kiyoyuki%22%2C%22lastName%22%3A%22Terakura%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masaharu%22%2C%22lastName%22%3A%22Oshima%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masa-aki%22%2C%22lastName%22%3A%22Kakimoto%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Seizo%22%2C%22lastName%22%3A%22Miyata%22%7D%5D%2C%22abstractNote%22%3A%22Carbon%20K-edge%20X-ray%20absorption%20spectra%20of%20nanographene%20have%20been%20simulated%20by%20density%20functional%20theory%20calculations%20to%20obtain%20information%20on%20the%20edge%20termination%20by%20hydrogen.%20Such%20information%20is%20crucially%20important%20to%20understand%20and%20predict%20functions%20such%20as%20transport%20and%20catalysis.%20Our%20results%20show%20that%20different%20edge%20terminations%20significantly%20affect%20the%20binding%20energy%20of%20the%20Is%20core-level%20of%20C%20atoms%20in%20the%20vicinity%20of%20edges%20because%20of%20the%20change%20in%20chemical%20bonding%20and%20the%20localized%20edge%20states.%20We%20find%20that%20a%20shoulder%20or%20a%20peak%20appears%20below%20the%20pi%2A%20peak%20at%20relatively%20different%20positions%20with%20respect%20to%20the%20pi%2A%20peak%20position%20in%20the%20theoretical%20spectra%20of%20zigzag%20graphene%20nanoribbons%2C%20depending%20on%20the%20ratio%20of%20monohydrogen-%20to%20dihydrogen-terminations.%20We%20also%20point%20out%20that%20the%20two%20additional%20features%20observed%20between%20the%20pi%2A%20and%20sigma%2A%20peaks%20of%20an%20ideal%20graphene%20originate%20from%20the%20sigma%2A%20states%20of%20C-H%20bonding%20and%20C-H-2%20bonding%20at%20the%20edges.%22%2C%22date%22%3A%222011%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Fjp110879d%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Fjp110879d%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221932-7447%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-11-30T08%3A40%3A50Z%22%7D%7D%2C%7B%22key%22%3A%22KVU3ID9V%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Kibalchenko%20et%20al.%22%2C%22parsedDate%22%3A%222011%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.%20Kibalchenko%2C%20J.R.%20Yates%2C%20C.%20Massobrio%2C%20A.%20Pasquarello%2C%20Structural%20Composition%20of%20First-Neighbor%20Shells%20in%20GeSe2%20and%20GeSe4%20Glasses%20from%20a%20First-Principles%20Analysis%20of%20NMR%20Chemical%20Shifts%2C%20Journal%20of%20Physical%20Chemistry%20C%20115%20%282011%29%207755%26%23x2013%3B7759.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjp201345e%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjp201345e%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Structural%20Composition%20of%20First-Neighbor%20Shells%20in%20GeSe2%20and%20GeSe4%20Glasses%20from%20a%20First-Principles%20Analysis%20of%20NMR%20Chemical%20Shifts%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mikhail%22%2C%22lastName%22%3A%22Kibalchenko%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jonathan%20R.%22%2C%22lastName%22%3A%22Yates%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Alfredo%22%2C%22lastName%22%3A%22Pasquarello%22%7D%5D%2C%22abstractNote%22%3A%22Isotropic%20chemical%20shifts%20and%20quadrupole%20coupling%20parameters%20of%20Se-77%20and%20Ge-73%20nuclei%20in%20GeSe2%20and%20GeSe4%20glasses%20are%20determined%20through%20density-functional%20NMR%20calculations%20on%20amorphous%20model%20structures%20generated%20by%20ab%20initio%20molecular%20dynamics.%20The%20comparison%20with%20experimental%20NMR%20spectra%20enables%20structural%20assignments%20for%20Se-77%20chemical%20shifts%2C%20pointing%20to%20fractional%20compositions%20of%20nearest-neighbor%20coordinations%20which%20are%20consistent%20with%20both%20neutron%20diffraction%20and%20NMR%20experiments.%22%2C%22date%22%3A%222011%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Fjp201345e%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Fjp201345e%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221932-7447%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-11-30T08%3A47%3A17Z%22%7D%7D%2C%7B%22key%22%3A%22UN8N78VI%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Koizumi%20et%20al.%22%2C%22parsedDate%22%3A%222011%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EK.%20Koizumi%2C%20M.%20Boero%2C%20Y.%20Shigeta%2C%20A.%20Oshiyama%2C%20Self-diffusion%20in%20crystalline%20silicon%3A%20A%20Car-Parrinello%20molecular%20dynamics%20study%2C%20Physical%20Review%20B%2084%20%282011%29%20205203.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.84.205203%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.84.205203%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Self-diffusion%20in%20crystalline%20silicon%3A%20A%20Car-Parrinello%20molecular%20dynamics%20study%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenichi%22%2C%22lastName%22%3A%22Koizumi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yasuteru%22%2C%22lastName%22%3A%22Shigeta%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Atsushi%22%2C%22lastName%22%3A%22Oshiyama%22%7D%5D%2C%22abstractNote%22%3A%22We%20report%20detailed%20atomistic%20calculations%20within%20the%20local%20density%20approximation%20%28LDA%29%20and%20the%20generalized%20gradient%20approximation%20%28GGA%29%20in%20density%20functional%20theory%20to%20clarify%20microscopic%20mechanisms%20and%20obtain%20the%20corresponding%20diffusion%20constant%20for%20self-diffusion%20in%20crystalline%20Si.%20The%20formation%20free%20energies%20of%20intrinsic%20defects%2C%20which%20mediate%20the%20self-diffusion%2C%20are%20calculated%20by%20accurate%20total-energy%20static%20calculations.%20Diffusivity%20in%20each%20mechanism%20is%20obtained%20from%20the%20mean-square%20displacements%20computed%20through%20Car-Parrinello%20molecular%20dynamics%20for%20a%20simulation%20time%20long%20enough%20to%20allow%20for%20these%20relatively%20slow%20phenomena%20to%20occur.%20We%20find%20that%20the%20interstitial%20mechanism%20dominantly%20contributes%20to%20the%20self-diffusion%3A%20The%20self-diffusion%20constant%20via%20the%20interstitial%20mechanism%20is%20found%20to%20be%20larger%20than%20that%20via%20the%20vacancy%20mechanism%20by%20about%20two%20orders%20of%20magnitude%20in%20LDA.%20We%20also%20find%20that%20the%20calculated%20formation%20free%20energies%20and%20migration%20energies%20in%20GGA%20are%20larger%20than%20the%20corresponding%20ones%20in%20LDA.%20Due%20to%20this%2C%20GGA%20substantially%20improves%20the%20free-energy%20landscape%2C%20thus%20providing%20diffusion%20constants%20in%20quantitative%20agreement%20with%20the%20experimental%20values%20over%20a%20whole%20temperature%20range.%20Atomistic%20processes%20in%20the%20self-diffusion%20are%20also%20clarified.%22%2C%22date%22%3A%222011%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevB.84.205203%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevB.84.205203%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221098-0121%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222016-03-16T16%3A01%3A26Z%22%7D%7D%2C%7B%22key%22%3A%22VCW5AJWP%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Le%20Roux%20et%20al.%22%2C%22parsedDate%22%3A%222011%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ES.%20Le%20Roux%2C%20A.%20Zeidler%2C%20P.S.%20Salmon%2C%20M.%20Boero%2C%20M.%20Micoulaut%2C%20C.%20Massobrio%2C%20Structural%20properties%20of%20liquid%20Ge2Se3%3A%20A%20first-principles%20study%2C%20Physical%20Review%20B%2084%20%282011%29%20134203%20%5C%2Fp.%201%26%23x2013%3B13.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.84.134203%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.84.134203%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Structural%20properties%20of%20liquid%20Ge2Se3%3A%20A%20first-principles%20study%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastien%22%2C%22lastName%22%3A%22Le%20Roux%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Anita%22%2C%22lastName%22%3A%22Zeidler%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Philip%20S.%22%2C%22lastName%22%3A%22Salmon%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Matthieu%22%2C%22lastName%22%3A%22Micoulaut%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%5D%2C%22abstractNote%22%3A%22The%20structural%20properties%20of%20liquid%20Ge2Se3%20were%20investigated%20by%20first-principles%20molecular%20dynamics%20using%20the%20Becke-Lee-Yang-Parr%20scheme%20for%20the%20treatment%20of%20the%20exchange-correlation%20functional%20in%20density%20functional%20theory.%20Our%20data%20for%20the%20total%20neutron%20structure%20factor%20and%20the%20total%20pair-distribution%20function%20are%20in%20excellent%20agreement%20with%20the%20experimental%20results.%20The%20structure%20is%20made%20predominantly%20%28similar%20to%2061%25%29%20from%20units%20comprising%20fourfold%20coordinated%20Ge%20atoms%20in%20the%20form%20of%20Ge-GeSe3%20or%20Ge-Se-4%20motifs%2C%20but%20there%20is%20also%20a%20large%20variety%20of%20motifs%20in%20which%20Ge%20and%20Se%20are%20not%20fourfold%20and%20twofold%20coordinated%2C%20respectively.%20The%20miscoordinated%20atoms%20and%20homopolar%20bonds%20lead%20to%20a%20highly%20perturbed%20tetrahedral%20network%2C%20as%20reflected%20by%20diffusion%20coefficients%20that%20are%20larger%20than%20in%20the%20case%20of%20liquid%20GeSe2.%20The%20network%20does%2C%20nevertheless%2C%20exhibit%20intermediate%20range%20order%20which%20is%20associated%20with%20the%20Ge-Ge%20correlations%20and%20which%20manifests%20itself%20by%20a%20first%20sharp%20diffraction%20peak%20in%20the%20total%20neutron%20structure%20factor.%20The%20evolution%20of%20the%20properties%20of%20Ge-x%20Se1-x%20liquids%20%280%20%3C%3D%20x%20%3C%3D%201%29%20with%20composition%20is%20discussed.%22%2C%22date%22%3A%222011%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevB.84.134203%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevB.84.134203%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221098-0121%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%222DH6J37C%22%2C%22BMA9GKQT%22%2C%22CF4ZI7HM%22%2C%22NZSFH59F%22%2C%22TK3HH32E%22%5D%2C%22dateModified%22%3A%222021-04-30T14%3A45%3A22Z%22%7D%7D%2C%7B%22key%22%3A%22CDZN2KK5%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Lee%20et%20al.%22%2C%22parsedDate%22%3A%222011%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EM.V.%20Lee%2C%20R.%20Scipioni%2C%20M.%20Boero%2C%20P.L.%20Silvestrelli%2C%20K.%20Ariga%2C%20The%20initiation%20mechanisms%20for%20surface%20hydrosilylation%20with%201-alkenes%2C%20Physical%20Chemistry%20Chemical%20Physics%2013%20%282011%29%204862%26%23x2013%3B4867.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc0cp01992e%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc0cp01992e%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22The%20initiation%20mechanisms%20for%20surface%20hydrosilylation%20with%201-alkenes%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Michael%20V.%22%2C%22lastName%22%3A%22Lee%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Roberto%22%2C%22lastName%22%3A%22Scipioni%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Pier%20Luigi%22%2C%22lastName%22%3A%22Silvestrelli%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Katsuhiko%22%2C%22lastName%22%3A%22Ariga%22%7D%5D%2C%22abstractNote%22%3A%22Hydrosilylation%20provides%20a%20route%20to%20form%20substituted%20silanes%20in%20solution.%20A%20similar%20reaction%20has%20been%20observed%20in%20the%20formation%20of%20covalent%20organic%20monolayers%20on%20a%20hydrogen-terminated%20silicon%20surface%20and%20is%20called%20thermal%20hydrosilylation.%20In%20solution%2C%20the%20mechanism%20requires%20a%20catalyst%20to%20add%20the%20basal%20silicon%20and%20saturating%20hydrogen%20to%20the%20C%3DC%20double%20bond.%20On%20the%20silicon%20surface%2C%20however%2C%20the%20reaction%20proceeds%20efficiently%20at%20200%20degrees%20C%2C%20initiated%20by%20visible%20light%2C%20and%20more%20slowly%20at%20room%20temperature%20in%20the%20dark.%20Such%20low%20activation%20energy%20barriers%20for%20the%20reactions%20on%20a%20surface%20relative%20to%20that%20required%20for%20solution%20hydrosilylation%20are%20remarkable%2C%20and%20although%20many%20explanations%20have%20been%20suggested%2C%20controversy%20still%20exists.%20In%20this%20work%20using%20a%20constrained%20molecular%20dynamics%20approach%20within%20the%20density%20functional%20theory%20framework%2C%20we%20show%20that%20the%20free%20energy%20activation%20barrier%20for%20abstraction%20of%20a%20hydrogen%20from%20silicon%20by%20an%20alkene%20molecule%20can%20be%20overcome%20by%20visible%20light%20or%20thermal%20excitation.%20Furthermore%2C%20we%20show%20that%20by%20concerted%20transfer%20of%20a%20hydrogen%20from%20the%20alpha-carbon%20to%20the%20beta-carbon%2C%20a%201-alkene%20can%20insert%20its%20alpha-carbon%20into%20a%20surface%20Si-H%20bond%20to%20accomplish%20hydrosilylation.%22%2C%22date%22%3A%222011%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fc0cp01992e%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fc0cp01992e%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221463-9076%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-10-22T09%3A41%3A31Z%22%7D%7D%2C%7B%22key%22%3A%22U8SMZAEB%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Rogez%20et%20al.%22%2C%22parsedDate%22%3A%222011%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EG.%20Rogez%2C%20C.%20Massobrio%2C%20P.%20Rabu%2C%20M.%20Drillon%2C%20Layered%20hydroxide%20hybrid%20nanostructures%3A%20a%20route%20to%20multifunctionality%2C%20Chemical%20Society%20Reviews%2040%20%282011%29%201031%26%23x2013%3B1058.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc0cs00159g%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc0cs00159g%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Layered%20hydroxide%20hybrid%20nanostructures%3A%20a%20route%20to%20multifunctionality%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guillaume%22%2C%22lastName%22%3A%22Rogez%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Pierre%22%2C%22lastName%22%3A%22Rabu%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Marc%22%2C%22lastName%22%3A%22Drillon%22%7D%5D%2C%22abstractNote%22%3A%22Today%2C%20seeking%20new%20materials%20for%20tailor-made%20applications%20and%20new%20devices%20leads%20to%20explore%20the%20potential%20offered%20by%20various%20kinds%20of%20functional%20building%20blocks.%20Hence%2C%20it%20concerns%20not%20only%20solid-state%20chemists%2C%20physicists%20or%20materials%20engineers%2C%20but%20also%20the%20area%20of%20%28supra%29%20molecular%20chemistry%2C%20and%20biochemistry%20as%20well.%20This%20is%20especially%20clear%20in%20the%20field%20of%20hybrid%20multifunctional%20materials.%20Indeed%2C%20their%20design%20requires%20investigating%20new%20concepts%20derived%20from%20principles%20developed%20in%20these%20different%20disciplines.%20The%20aim%20of%20this%20critical%20review%20is%20to%20present%20the%20last%20achievements%20concerning%20transition%20metal%20hydroxide%20hybrids%2C%20their%20synthesis%2C%20flexibility%20and%20functional%20properties.%20They%20often%20provide%20nice%20model%20systems%20for%20understanding%20the%20correlation%20between%20structure%20and%20physical%20properties%20brought%20by%20the%20molecular%20moieties%20grafted%20onto%20the%20metal%20hydroxide%20basis%20layers.%20The%20contribution%20of%20the%20atomic%20scale%20modelling%20to%20the%20electronic%20structure%20calculations%20and%20structural%20optimization%20is%20also%20reported%20%28216%20references%29.%22%2C%22date%22%3A%222011%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fc0cs00159g%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fc0cs00159g%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220306-0012%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22M244N6AF%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-10-22T08%3A53%3A56Z%22%7D%7D%2C%7B%22key%22%3A%22FVQZVRCT%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Scipioni%20et%20al.%22%2C%22parsedDate%22%3A%222011%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ER.%20Scipioni%2C%20J.P.%20Hill%2C%20G.J.%20Richards%2C%20M.%20Boero%2C%20T.%20Mori%2C%20K.%20Ariga%2C%20T.%20Ohno%2C%20Tautomers%20of%20extended%20reduced%20pyrazinacenes%3A%20a%20density-functional-theory%20based%20study%2C%20Physical%20Chemistry%20Chemical%20Physics%2013%20%282011%29%202145%26%23x2013%3B2150.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc0cp01245a%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1039%5C%2Fc0cp01245a%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Tautomers%20of%20extended%20reduced%20pyrazinacenes%3A%20a%20density-functional-theory%20based%20study%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Roberto%22%2C%22lastName%22%3A%22Scipioni%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jonathan%20P.%22%2C%22lastName%22%3A%22Hill%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Gary%20J.%22%2C%22lastName%22%3A%22Richards%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Toshiyuki%22%2C%22lastName%22%3A%22Mori%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Katsuhiko%22%2C%22lastName%22%3A%22Ariga%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Takahisa%22%2C%22lastName%22%3A%22Ohno%22%7D%5D%2C%22abstractNote%22%3A%22We%20present%20a%20structural%20and%20electronic%20inspection%20of%20reduced%20pyrazinacenes%20within%20the%20DFT%20framework.%20Our%20analysis%20provides%20a%20clear%20indication%20that%20compounds%20in%20which%20reduced%20pyrazine%20rings%20are%20well%20separated%20from%20each%20other%20are%20rather%20stable.%20Conversely%2C%20if%20the%20reduced%20pyrazine%20rings%20approach%20each%20other%20or%20cluster%20together%2C%20the%20compounds%20become%20increasingly%20unstable.%20The%20tautomers%20analyzed%20are%20likely%20to%20possess%20properties%20suitable%20for%20application%20as%20proton%20transport%20materials%20due%20to%20protic%20isomerism%20processes.%20On%20the%20basis%20of%20our%20findings%2C%20we%20propose%20that%20protic%20transport%20should%20occur%20through%20a%20concerted%20proton%20transfer%20without%20involving%20intramolecular%20aggregation%20of%20the%20dihydropyrazine%20groups.%20Furthermore%2C%20the%20electronic%20structure%20analysis%20shows%20that%20this%20class%20of%20compounds%20can%20be%20classified%20as%20small%20bandgap%20semiconducting%20materials%2C%20possessing%20even%20metallic%20character%20depending%20on%20the%20tautomeric%20structure%2C%20and%20with%20potential%20nanotechnological%20applications%20in%20molecular%20electronics%20and%20fuel%20cells.%22%2C%22date%22%3A%222011%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1039%5C%2Fc0cp01245a%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1039%5C%2Fc0cp01245a%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221463-9076%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-10-22T08%3A54%3A28Z%22%7D%7D%2C%7B%22key%22%3A%22ZNF2QQ6V%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Scipioni%20et%20al.%22%2C%22parsedDate%22%3A%222011%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ER.%20Scipioni%2C%20M.%20Matsubara%2C%20E.%20Ruiz%2C%20C.%20Massobrio%2C%20M.%20Boero%2C%20Thermal%20behavior%20of%20Si-doped%20fullerenes%20vs%20their%20structural%20stability%20at%20T%3D0%20K%3A%20A%20density%20functional%20study%2C%20Chemical%20Physics%20Letters%20510%20%282011%29%2014%26%23x2013%3B17.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cplett.2011.05.019%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cplett.2011.05.019%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Thermal%20behavior%20of%20Si-doped%20fullerenes%20vs%20their%20structural%20stability%20at%20T%3D0%20K%3A%20A%20density%20functional%20study%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Roberto%22%2C%22lastName%22%3A%22Scipioni%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masahiko%22%2C%22lastName%22%3A%22Matsubara%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Eliseo%22%2C%22lastName%22%3A%22Ruiz%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%5D%2C%22abstractNote%22%3A%22We%20establish%20the%20topological%20conditions%20underlying%20the%20thermal%20stability%20of%20C%283%290Si%2830%29%20clusters.%20Two%20topologies%20have%20been%20considered%3A%20a%20segregated%20one%2C%20where%20Si%20and%20C%20atoms%20lie%20on%20neighboring%20and%20yet%2C%20separated%20parts%20of%20the%20cage%2C%20and%20a%20non-segregated%20one%2C%20where%20the%20number%20of%20Si-C%20bonds%20is%20maximized.%20The%20segregated%20network%20is%20energetically%20favored%20against%20the%20non-segregated%20one%2C%20both%20structures%20being%20fully%20relaxed%20at%20T%20%3D%200%20K.%20Conversely%2C%20the%20non-segregated%20structure%20is%20the%20only%20one%20stable%20at%20finite%20temperatures%2C%20regardless%20of%20the%20nature%20of%20the%20local%20states%20%28d%20or%20p%29%20included%20in%20the%20Kleynman-Bylander%20construction.%20%28C%29%202011%20Elsevier%20B.V.%20All%20rights%20reserved.%22%2C%22date%22%3A%222011%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.cplett.2011.05.019%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1016%5C%2Fj.cplett.2011.05.019%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220009-2614%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-11-30T09%3A04%3A02Z%22%7D%7D%2C%7B%22key%22%3A%22WAINM6V2%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Wang%20et%20al.%22%2C%22parsedDate%22%3A%222011%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EX.%20Wang%2C%20Z.%20Hou%2C%20T.%20Ikeda%2C%20S.-F.%20Huang%2C%20K.%20Terakura%2C%20M.%20Boero%2C%20M.%20Oshima%2C%20M.%20Kakimoto%2C%20S.%20Miyata%2C%20Selective%20nitrogen%20doping%20in%20graphene%3A%20Enhanced%20catalytic%20activity%20for%20the%20oxygen%20reduction%20reaction%2C%20Physical%20Review%20B%2084%20%282011%29.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.84.245434%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1103%5C%2FPhysRevB.84.245434%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Selective%20nitrogen%20doping%20in%20graphene%3A%20Enhanced%20catalytic%20activity%20for%20the%20oxygen%20reduction%20reaction%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Xianlong%22%2C%22lastName%22%3A%22Wang%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Zhufeng%22%2C%22lastName%22%3A%22Hou%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Takashi%22%2C%22lastName%22%3A%22Ikeda%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sheng-Feng%22%2C%22lastName%22%3A%22Huang%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kiyoyuki%22%2C%22lastName%22%3A%22Terakura%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masaharu%22%2C%22lastName%22%3A%22Oshima%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masa-aki%22%2C%22lastName%22%3A%22Kakimoto%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Seizo%22%2C%22lastName%22%3A%22Miyata%22%7D%5D%2C%22abstractNote%22%3A%22The%20structural%20and%20electronic%20properties%20of%20N-doped%20zigzag%20graphene%20ribbons%20with%20various%20ratios%20of%20dihydrogenated%20to%20monohydrogenated%20edge%20carbons%20are%20investigated%20within%20the%20density%20functional%20theory%20framework.%20We%20find%20that%20the%20stability%20of%20graphitic%20N%20next%20to%20the%20edge%2C%20which%20is%20claimed%20to%20play%20important%20roles%20in%20the%20catalytic%20activity%20in%20our%20previous%20work%2C%20will%20be%20enhanced%20with%20increasing%20the%20concentration%20of%20dihydrogenated%20carbons.%20Furthermore%2C%20the%20dihydrogenated%20edge%20carbons%20turn%20out%20to%20be%20easily%20converted%20into%20monohydrogenated%20ones%20in%20the%20presence%20of%20oxygen%20molecules%20at%20room%20temperature.%20Based%20on%20our%20results%2C%20we%20propose%20a%20possible%20way%20to%20enhance%20the%20oxygen%20reduction%20catalytic%20activity%20of%20N-doped%20graphene%20by%20controlling%20the%20degrees%20of%20hydrogenation%20of%20edge%20carbons.%20The%20characteristic%20features%20in%20the%20x-ray%20absorption%20and%20emission%20spectra%20for%20each%20specific%20N%20site%20considered%20here%20will%20also%20be%20given.%22%2C%22date%22%3A%222011%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1103%5C%2FPhysRevB.84.245434%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1103%5C%2FPhysRevB.84.245434%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221098-0121%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-11-30T10%3A02%3A15Z%22%7D%7D%2C%7B%22key%22%3A%227PCZAMCE%22%2C%22library%22%3A%7B%22id%22%3A1839302%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Zucca%20et%20al.%22%2C%22parsedDate%22%3A%222011%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E%5B1%5D%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ER.%20Zucca%2C%20M.%20Boero%2C%20C.%20Massobrio%2C%20C.%20Molteni%2C%20F.%20Cleri%2C%20Interacting%20Lewis-X%20Carbohydrates%20in%20Condensed%20Phase%3A%20A%20First-Principles%20Molecular%20Dynamics%20Study%2C%20Journal%20of%20Physical%20Chemistry%20B%20115%20%282011%29%2012599%26%23x2013%3B12606.%20%3Ca%20class%3D%27zp-DOIURL%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjp2055816%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Fjp2055816%3C%5C%2Fa%3E.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Interacting%20Lewis-X%20Carbohydrates%20in%20Condensed%20Phase%3A%20A%20First-Principles%20Molecular%20Dynamics%20Study%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Rinaldo%22%2C%22lastName%22%3A%22Zucca%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mauro%22%2C%22lastName%22%3A%22Boero%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carlo%22%2C%22lastName%22%3A%22Massobrio%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carla%22%2C%22lastName%22%3A%22Molteni%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fabrizio%22%2C%22lastName%22%3A%22Cleri%22%7D%5D%2C%22abstractNote%22%3A%22We%20performed%20first-principles%20molecular%20dynamics%20calculations%20at%20finite%20temperature%2C%20to%20study%20the%20interacting%20conformations%20of%20Lewis-X%20%28LeX%29%20trisaccharides%20in%20the%20crystalline%20phase.%20The%20calculated%20cell%20parameters%20and%20detailed%20atomic%20structure%20of%20the%20LeX%20molecule%20compare%20well%20to%20the%20experimental%20data%20obtained%20by%20X-ray%20diffraction.%20We%20identify%20and%20characterize%20the%20hydrogen-bond%20network%2C%20responsible%20for%20the%20mutual%20interaction%20of%20the%20LeX%20pairs%2C%20whereas%20we%20find%20the%20intramolecular%20conformation%20and%20stability%20to%20be%20mainly%20assured%20by%20dispersion%20forces.%20The%20relative%20contributions%20to%20the%20crystallization%20energy%20of%20the%20hydrogen%20bonds%20and%20of%20the%20dispersion%20forces%20are%20defined%20and%20quantified.%20From%20this%20study%2C%20candidate%20configurations%20for%20the%20fully%20hydrated%2C%20in%20vivo%20structures%20of%20homotypic%20LeX%20LeX%20interactions%20at%20cell%20surfaces%20can%20be%20proposed.%20We%20discuss%20how%20these%20configurations%20could%20also%20be%20relevant%20for%20the%20adhesion%20and%20self-assembly%20of%20nanostructures.%22%2C%22date%22%3A%222011%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Fjp2055816%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fdx.doi.org%5C%2F10.1021%5C%2Fjp2055816%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%221520-6106%22%2C%22language%22%3A%22English%22%2C%22collections%22%3A%5B%22CF4ZI7HM%22%2C%22NZSFH59F%22%5D%2C%22dateModified%22%3A%222015-11-30T09%3A48%3A27Z%22%7D%7D%5D%7D
[1]
M. Bauchy, M. Micoulaut, M. Celino, S. Le Roux, M. Boero, C. Massobrio, Angular rigidity in tetrahedral network glasses with changing composition, Physical Review B 84 (2011) 054201 /p. 1–9.
https://doi.org/10.1103/PhysRevB.84.054201.
[1]
S. Bhosle, K. Gunasekera, P. Chen, P. Boolchand, M. Micoulaut, C. Massobrio, Meeting experimental challenges to physics of network glasses: Assessing the role of sample homogeneity, Solid State Communications 151 (2011) 1851–1855.
https://doi.org/10.1016/j.ssc.2011.10.016.
[1]
M. Boero, LeuRS Synthetase: A First-Principles Investigation of the Water-Mediated Editing Reaction, Journal of Physical Chemistry B 115 (2011) 12276–12286.
https://doi.org/10.1021/jp2070024.
[1]
L. Giacomazzi, C. Massobrio, A. Pasquarello, Vibrational properties of vitreous GeSe2 with the Becke-Lee-Yang-Parr density functional, Journal of Physics-Condensed Matter 23 (2011) 295401 /p. 1–6.
https://doi.org/10.1088/0953-8984/23/29/295401.
[1]
B.W. Heinrich, L. Limot, M.V. Rastei, C. Iacovita, J.-P. Bucher, D.M. Djimbi, C. Massobrio, M. Boero, Dispersion and Localization of Electronic States at a Ferrocene/Cu(111) Interface, Physical Review Letters 107 (2011) 216801 /p. 1–5.
https://doi.org/10.1103/PhysRevLett.107.216801.
[1]
Z. Hou, X. Wang, T. Ikeda, S.-F. Huang, K. Terakura, M. Boero, M. Oshima, M. Kakimoto, S. Miyata, Effect of Hydrogen Termination on Carbon K-Edge X-ray Absorption Spectra of Nanographene, Journal of Physical Chemistry C 115 (2011) 5392–5403.
https://doi.org/10.1021/jp110879d.
[1]
M. Kibalchenko, J.R. Yates, C. Massobrio, A. Pasquarello, Structural Composition of First-Neighbor Shells in GeSe2 and GeSe4 Glasses from a First-Principles Analysis of NMR Chemical Shifts, Journal of Physical Chemistry C 115 (2011) 7755–7759.
https://doi.org/10.1021/jp201345e.
[1]
S. Le Roux, A. Zeidler, P.S. Salmon, M. Boero, M. Micoulaut, C. Massobrio, Structural properties of liquid Ge2Se3: A first-principles study, Physical Review B 84 (2011) 134203 /p. 1–13.
https://doi.org/10.1103/PhysRevB.84.134203.
[1]
M.V. Lee, R. Scipioni, M. Boero, P.L. Silvestrelli, K. Ariga, The initiation mechanisms for surface hydrosilylation with 1-alkenes, Physical Chemistry Chemical Physics 13 (2011) 4862–4867.
https://doi.org/10.1039/c0cp01992e.
[1]
G. Rogez, C. Massobrio, P. Rabu, M. Drillon, Layered hydroxide hybrid nanostructures: a route to multifunctionality, Chemical Society Reviews 40 (2011) 1031–1058.
https://doi.org/10.1039/c0cs00159g.
[1]
R. Scipioni, J.P. Hill, G.J. Richards, M. Boero, T. Mori, K. Ariga, T. Ohno, Tautomers of extended reduced pyrazinacenes: a density-functional-theory based study, Physical Chemistry Chemical Physics 13 (2011) 2145–2150.
https://doi.org/10.1039/c0cp01245a.
[1]
R. Scipioni, M. Matsubara, E. Ruiz, C. Massobrio, M. Boero, Thermal behavior of Si-doped fullerenes vs their structural stability at T=0 K: A density functional study, Chemical Physics Letters 510 (2011) 14–17.
https://doi.org/10.1016/j.cplett.2011.05.019.
[1]
X. Wang, Z. Hou, T. Ikeda, S.-F. Huang, K. Terakura, M. Boero, M. Oshima, M. Kakimoto, S. Miyata, Selective nitrogen doping in graphene: Enhanced catalytic activity for the oxygen reduction reaction, Physical Review B 84 (2011).
https://doi.org/10.1103/PhysRevB.84.245434.
[1]
R. Zucca, M. Boero, C. Massobrio, C. Molteni, F. Cleri, Interacting Lewis-X Carbohydrates in Condensed Phase: A First-Principles Molecular Dynamics Study, Journal of Physical Chemistry B 115 (2011) 12599–12606.
https://doi.org/10.1021/jp2055816.